BME100 f2017:Projects2
| Home People Lab Write-Up 1 | Lab Write-Up 2 | Lab Write-Up 3 Lab Write-Up 4 | Lab Write-Up 5 | Lab Write-Up 6 Course Logistics For Instructors Photos Wiki Editing Help | ||||||
|
OUR TEAM
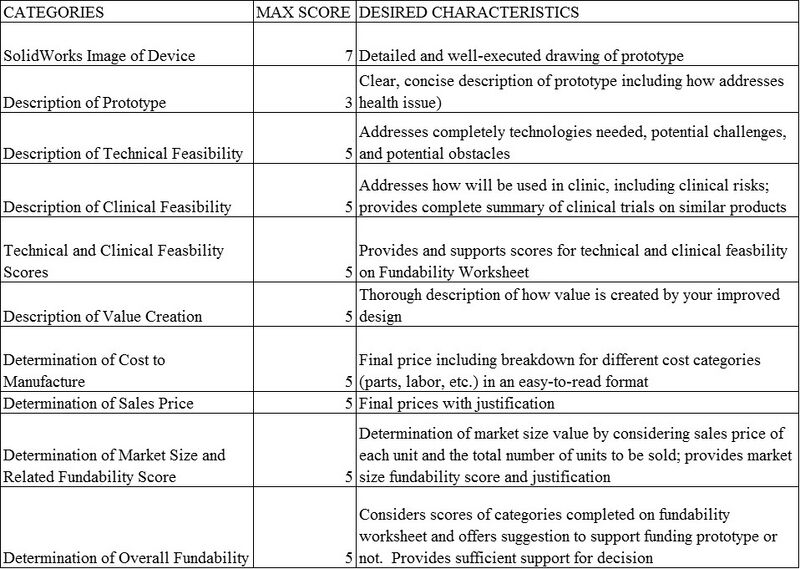
Lab 2: “Prototype Design” | ||||||
PART 1: LEARNING OBJECTIVES:
- Learn how to use SolidWorks: Leverage “Help” in SolidWorks and Custom video on Blackboard.
- Design a prototype to address the chosen health issue.
- Draw prototype in SolidWorks.
- Determine the technical feasibility.
- Determine the clinical feasibility.
PART 1: ASSIGNMENT
1. Design and draw your prototype in SolidWorks. Upload as an image and provide a brief description (including how addresses health issue) on Open Wet Ware.
This is a pain relieving thermal therapy device that is designed for comfort and portability. The device fluctuates from cooling and heating to provide maximum pain relief for people of all ages. This device is effective, safe, and it is able to easily target areas of the body that are in need of the therapy. Although the target market for this device is physical therapy, this thermal therapy product can be used for many reasons including post-surgical recovery, sports medicine, pain management, in-hospital treatment, and physiotherapy. The device comes with a screen on the front which makes changing the temperature and settings simple. An app will also be developed alongside this device so that the physician or physical therapist of the patient can set and monitor the course of treatment conveniently.
2. Determine the technical feasibility of your design. Include answers to the following questions:
a. What are the technologies needed?
The technologies needed for this device include thermoelectric heating and cooling. Thermoelectric heating/cooling uses the Peltier Effect, which is a change in temperature created through applying a voltage between two electrodes. Usually in a device that uses the Peltier Effect, the electrodes are made out of a metal with good electrical conductivity (Rouse, 2008). Using thermoelectric technologies allows the patient to cool and heat different areas of the body without the harsh temperature of ice and with therapeutic heat easily.
b. What are the challenges?
A challenge in the technical feasibility of this product is making the device alternate from heating to cooling and vice-versa with ease. The goal for the product is for the patient to be able to change the temperature of their portable device efficiently and safely without any complications. This device will also be connected to an app which the patient’s physician or physical therapist is able to monitor the progress and set the temperature as needed. This could be seen as a possible challenge in regards to technical feasibility.
c. What could go wrong?
There could be potential issues for this product when it comes to switching between heating and cooling with ease. However, with the right technologies enforced, the device should be able to function efficiently at any set temperature. There could also be technological error in regards to connecting the device to the app. This is why testing will occur in order for the device to function to its safest and most productive abilities.
3. Determine the clinical feasibility.
a. Will it work in the clinic?
b. What are the clinical risks?
c. Have similar products been in a clinical trial? How long was the trial?
4. Using the fundability worksheet chart, determine the scores (0-3) for technical feasibility and clinical feasibility and provide support for scores.
The Thermal Therapy device scores a 3 out of 3 for technical feasibility since a product very similar to it has already been created. Though the concept of the competing product is more or less the same as the Thermal Therapy pad, installing the wireless feature is not a significant obstacle to overcome in the manufacturing process. The device has a very straightforward design with many already known materials. For clinical feasibility, the TT pad earns a 2 out of 3 because customer approval of this specific product has yet to be conducted and collected. While devices very similar to it have been well received by customers in the home-remedy healing market. A slight risk posed by the wireless feature of the pad includes the potential for the timing of the pad to get off its set schedule. For instance, customers could have the pad timed to turn on while they are sleeping, and if it does not shut off when it should it could pose a risk to them. Since they are dependent on the pad turning on and off independently, they may go to bed without having to worry about turning it off like they would expect to with a pad that needs to be plugged in.
Resources:
- Video on the basics of SolidWorks (on Bb)
- Video on how to search for clinical trial information: See “Improving Searches with Boolean Operators” Video to learn about ClinicalTrials.gov (on Bb)
PART 2: LEARNING OBJECTIVES:
- Determine the market size.
- Determine fundability.
PART 2: ASSIGNMENT:
1. What value does your prototype create for the customer?
Customers generally evaluate a product or service based on the value vs. the price that it is being asked for. Therefore, the thermal therapy device that we will be presenting to our customers will provide a more portable and electronically convenient method for healing and cooling therapy. The device will ease the patient’s condition by retrieving specific prescriptions from their physicians/physical therapists via the mobile app. The adjustable temperature of the device provides therapeutic benefits to those physically injured while successfully increasing their range of motion.
2. Determine the cost to create your design. Justify.
The determined cost to create the thermal therapy device will be moderately more expensive than the usual heating and cooling devices since it is a more efficient health solution that combines two products into one. A generic cooling machine is worth 50 dollars compared to any higher-demand form of treatments generally worth 300 dollars. Same applies to generic thermal therapy devices. Generic heating machines are worth an average of 200 dollars, but our thermal therapy device will cost an average of 125 dollars to assemble. Thus, making it a product worth the purchase.
3. What would be the anticipated average sale price (ASP)? Justify.
The anticipated average sale price would come out to $500.00. The manufacture price of the product would cost $100. In order for the device to perform both tasks of cooling and heating in such a portable manner, the product will cost more to manufacture than a basic heating/cooling device. In order to retrieve the sale price, the manufacture price was multiplied by 5. The total cost comes out to $625.
4. Using initial market size analysis in Lab 1, determine the market size in dollars per year.
The initial market size for the physical therapy field is about 200,000,000.
$500 x 0.05 x 200,000,000 = $5,000,000,000
(sale price x penetrance x number of customers)
5. Using the fundability worksheet, determine if your prototype should be funded. Justify why or why not.
Based on the fundability worksheet, our prototype should be funded. The physical therapy market size is significantly large enough to keep the product active in the market. The price of the device is reasonably priced compared to its competitors, while still maximizing its efficiency and portability. Other products/devices that relate to this device have been very successful. Therefore, clinical feasibility is not an issue for this product. People of all age ranges and backgrounds are qualified to use this device. Whether an athlete needs relief after an injury or an elderly person needs to find comfort with aches and pain, this device will provide alleviation and comfort.
HW: Lab Report 2 (due 09/19/17 at 11:59PM through OpenWetWare)
Grading Scheme:
50 points – See Rubric
50 points – Peer Assessment
| Look for your Group's lab report link in the list of links on the right. If it is RED instead of blue, DO NOT CLICK ON IT! If the link is red, type the name of your Group's lab report link exactly as shown in the following text field and click the Create Page button. This should open up an Edit page that is pre-populated with a lab report template code. |
|}