BME100 f2017:Group10 W1030 L2
| Home People Lab Write-Up 1 | Lab Write-Up 2 | Lab Write-Up 3 Lab Write-Up 4 | Lab Write-Up 5 | Lab Write-Up 6 Course Logistics For Instructors Photos Wiki Editing Help | ||||||
OUR TEAM
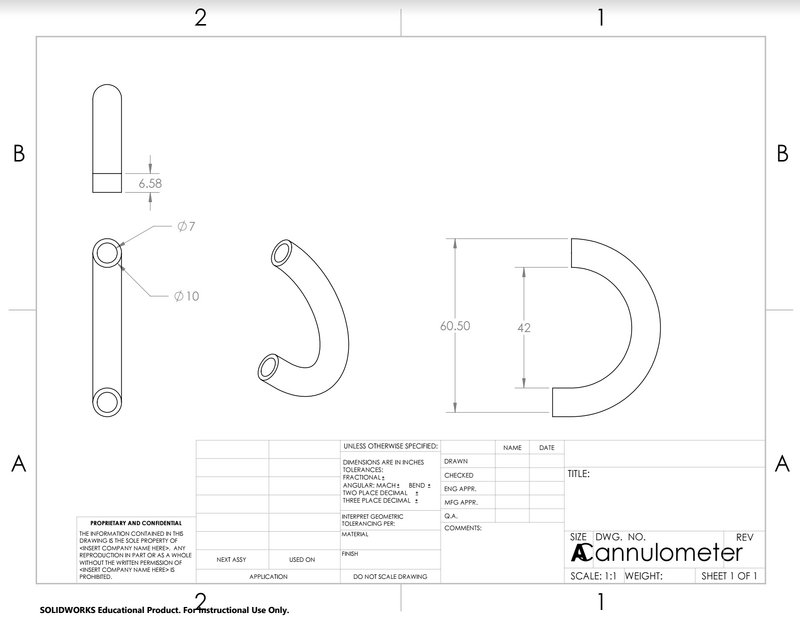
LAB 2 WRITE-UPDevice Image and DescriptionIn order to minimize discomfort and skin irritation caused by other methods of securing nasal cannulas to a patient's face such as tape or headbands, the Cannulometer instead wraps around a patient's ears. Composed of silicone-coated plastic with a micro infrared temperature sensor embedded into the surface, this device will be both compact and comfortable. In addition, the behind-the-ear thermometer instantly reads temperature and transmits it via radio waves to any device, allowing care-takers to conveniently track critical patient's temperature and potentially detect signs of infection earlier.
Technical and Clinical FeasibilityTechnical Feasibility 2. Challenges: The prototype must simultaneously secure the nasal cannula out of the way of the patient’s face and wrap around to the base of the ear where body temperature can accurately be read. In addition, the device must not be too bulky so as to put pressure on the ear and cause discomfort. Cost-effectiveness is also a challenge because the device requires a compact thermometer with its own power source and an ability to transmit data. 3. Potential Errors: Because the device is c-shaped, it might not properly fit the patient’s ear causing instability and discomfort. The temperature sensor may not line up to the position behind the ear with the most accuracy and precision on every single person. The sensor could be inaccurate, causing medical professionals to run tests or treat for infection when the patient actually does not have an elevated temperature. The sensor could fail to transmit data real-time and thus not alert nurses and doctors of a dangerously high fever. Clinical Feasibility 2. Clinical Risks: Through trial, the physical risks are rather slim. Being that the device is simply a hook that wraps around the ear, very little can go wrong. However, like all new up and coming devices, the risks should be mentioned. Certain situations that involve “pulling” or “yanking” of the nasal tubing may cause bruising or discomfort in the ears. 3. Similar Clinical Trials:
The technology of infrared temperature sensors has been patented but the sensor required for this small of a device has not yet been patented. Because it is a low-risk device, it merely needs FDA approval without clinical data. There are many clinical trials have utilized nasal cannulas when treating pulmonary and respiratory complications, but no trials have been completed testing the effectiveness of a regular nasal cannula across a wide range of patients. The Cannulometer must show that it is substantially equivalent to nasal cannula and body temperature sensors, which have already been approved by the FDA.
Market AnalysisValue Creation Manufacturing Cost Sales Price Market Size
Fundability DiscussionThe Cannulometer scored a 3 in the “technical feasibility” section because, in its simplest form, it is just a plastic hook that goes around the ear of the wearer. While there are a few bells and whistles (temperature gauge) these features require nothing more than implementation into an already spacious location which will prove to be extremely simple. The Cannulometer scored a 2 for the “clinical feasibility” due to its slightly limited user base, even though it is believed that those that do require a nasal cannula would be more than willing to test this product. However, the success of its clinical feasibility is weakened due to the fact that not everyone who is ill or needs hospitalization is in need of a nasal cannula. Frankly, the outcome according to patients is predicted to be rather positive overall.
Works CitedBLE Nano & MK20 USB Board http://redbearlab.com/blenano/ (accessed Sep 18, 2017). Faris, O.; Shuren, J. An FDA Viewpoint on Unique Considerations for Medical-Device Clinical Trials — NEJM http://www.nejm.org/doi/full/10.1056/NEJMra1512592?rss=searchAn dBrowse&#t=article (accessed Sep 18, 2017). Nasal Cannula | Completed Studies https://clinicaltrials.gov/ct2/results?term=Nasal%2BCannula &Search=Apply&recrs=e&age_v=&gndr=&type=&rslt=#wrapper (accessed Sep 19, 2017). The World's Tiniest Temperature Sensor is Powered by Radio Waves https://phys.org/ne ws/2015-12-world-tiniest-temperature-sensor-powered.html (accessed Sep 18, 2017).
|
||||||