BME100 f2017:Group5 W0800 L2
| Home People Lab Write-Up 1 | Lab Write-Up 2 | Lab Write-Up 3 Lab Write-Up 4 | Lab Write-Up 5 | Lab Write-Up 6 Course Logistics For Instructors Photos Wiki Editing Help | ||||||
OUR TEAM
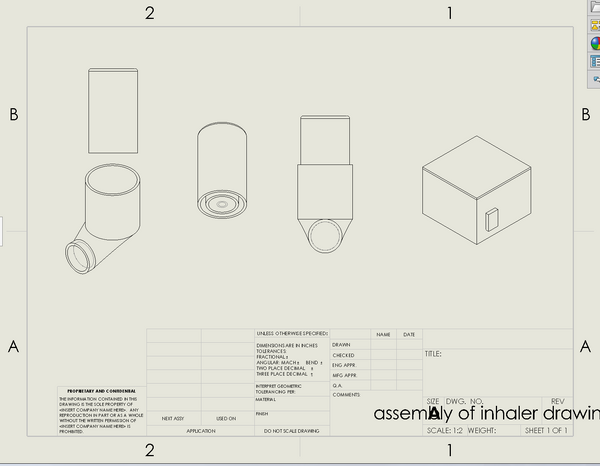
LAB 2 WRITE-UPDevice Image and DescriptionThe inhaler that we designed is 2 chambers. The center chamber will hold the medication within it, and the outer contains vegetable glycerin. Also, refrigeration units are scarce in China, so we designed a mini fridge that will contain the inhaler cartridge. Once the cartridge has been the unit for some time, it will stay cool due to the properties of the vegetable glycerin.
Technical and Clinical FeasibilityTechnical Feasibility What are the technologies needed? This design requires a bio preservation chamber to keep the organisms cooler. The cooling device will have to be electrically powered, with a battery to prolong its preservation. The chamber which contains the microorganisms need to contain an agar solution of nutrients to sustain the organisms. The aerosolized spray will contain eucalyptus oil and menthol in separate chambers. Each compartment will have to dispense a set amount of its contents into the the mixture for inhalation. The mixture will need to be aerosolized into a mist that can be inhaled. Inhalers can currently release medication in two different ways: though aerosolized spray, or through dry powder spray. “Metered dose inhalers” spray aerosolized medication with manual activation, and “breath activated inhalers” do the same automatically upon inhalation. “Dry powdered inhalers” spray out, as the name implies, a dry powder medicine [1]. We are currently planning on a metered dose inhaler.
We will have to set a realistic yet feasible expiration date; one that will allow the product to undergo transportation and reasonable time before use, but can be reasonably attained with our design. To ensure the optimal time of preservation, we will have to balance the amount of nutrients in the agar solution for the organisms. We will have to determine dosage and ensure that the amount of each substance in each dose is consistent and in the proper amount. There will have to be instructions for use and care of the inhaler given to the patient.
The microorganisms could become infectious, they could die before the planned expiration date, and the eucalyptus and menthol could harm the microorganism. If the cooling mechanism fails, the organisms will expire before the expiration date. If the dosage is not consistent it can affect the recovery of the patient and potentially harm their health.
Given the technical feasibility it work in the clinic? Our inhaler is designed to be patient-friendly and easy-to-use. Studies have shown that proper inhaler technique is correlated with receiving medical professional instruction on proper usage [2]. Demonstrating proper inhaler technique is brief and straightforward, and afterwards, the patient is able to administer his or her own inhaler, placing minimal strain on healthcare professional resources. Since our inhaler dispenses coal-degrading microorganisms, it will likely require a prescription to acquire.
The clinical risks associated with corticosteroid inhalers include increased risk of pneumonia, gastrointestinal bleeding, hyperglycemia, and hypertension [3, 4], and beta-agonist inhalers can lead to a higher risk of myocardial infarction [3, 4, 5]. Since our inhaler introduces a new kind of therapy, free of synthetic drugs, we are interested in developing our technology to avoid these clinical risks already associated with inhalers. Since we are introducing microorganisms into the lungs, we risk generating an adverse immune response. It may be essential for us to genetically engineering our microorganisms to evade detection by the host immune system, such as by altering the outer membrane components of our bacteria (to minimize recognition of LPS, if we use gram-negative bacteria, for example). Alternatively, we could disguise our organisms in microns-wide glycerin packages to minimize the time between detection by the host immune response and the initiation of coal breakdown. We may need to resort to isolating the coal-degrading compounds from our microorganisms, and having those be the sole active ingredients in our inhaler, sans microbes. If we are able to use microorganisms in our inhaler, we would likely want the organisms to die off in the lungs relatively quickly after inoculation, to avoid unwanted evolution in the lungs. Modeling off of how pathogenic bacteria in higher-BLS level labs are modified to lack an essential nutrient-synthesizing gene, and thus cannot survive in a medium without that nutrient, we could remove a similar gene if we use bacteria, or perform a similarly-goaled modification if we use fungi.
There are an abundance of clinical trials that have studied the efficacy and side-effects of inhalers. One 3 year study found that poor asthma control was associated with poor compliance, which encouraged us to pursue designs that encourage patients to use their inhaler, such as an inhaler that inflates overnight, or a refrigerator unit that glows red if it hasn’t been opened up in 24 hours [6]. Other clinical trials have looked at the success of inhaler use in respiratory distress syndrome in infants. These trials tended to range in length from a few days to a few weeks [7]. A recent 5-day clinical trial tested the combination of corticosteroid and beta-agonist medication inhalers for acute respiratory distress syndrome [8]. To investigate the effect on the type and length of our needed clinical trials from using live organisms in our inhaler, we researched clinical trial guidelines for probiotic development. The FDA essentially announced in 2010 that probiotics intended for a health improvement goal need to be classified as an “investigational new drug” (IND), and thus the FDA must be notified 30 days before clinical trials will commence, and the FDA, although not capable of “approving” an IND, oversees the process of developing the IND before clinical trials, and can halt clinical trials at any time. An overview of clinical studies of probiotics published in Gut Microbes states “It is common for this phased regulatory scheme for the collection of data to take a number of years before completion and before an agency approval of the desired drug for a given indication is achieved (if ever)” [9].
Market AnalysisValue Creation Our take on the modern inhaler was inspired by the horrible air quality in many Asian countries due to the massive amounts of coal used to supply energy. Most inhalers today are just anti-inflammatory aids that help open up the airways allowing the easy passage of oxygen to the lungs, and while useful, this doesn’t really solve the problem. Our device would take microorganisms that have been aerosolized and put into a capsule. Once inhaled, these organisms would go to work at breaking down the coal buildup in the lungs, allowing the macrophages of the body clean up the rest naturally. Manufacturing Cost Your average albuterol inhaler usually costs about 5-50 dollars with insurance depending on the brand and can range from 30-60 dollars without insurance. As our inhaler is a bit more complex but is supposed to be a daily treatment until all the medicine has been ingested. The price would only be slightly elevated due to the materials being used in the actual can being harder to find and reproduce. The price would range anywhere from 75-100 dollars [10, 11] Sales Price The average sales price would range anywhere from 75-100 dollars. This is because the same basic materials are being used to construct the actual inhaler and can. The medicine on the other hand would be made of completely different substances that are much harder to get to reproduce [10]. Market Size An earlier Global Burden of Disease study that examined health figures for 2010 found that outdoor air pollution contributed to 1.2 million premature deaths, nearly 40 percent of the global total [9]. . (75$-100$ * .05 * 1.2million) = 4.5million-6million
Fundability Discussion
Works Cited1. Asthma UK. (n.d.). Preventer inhalers. Retrieved September 13, 2017, from https://www.asthma.org.uk/advice/inhalers-medicines-treatments/inhalers-and-spacers/preventer/ 2. Elsevier. (2011, March 02). Inhaler mishandling remains common in real life and is associated with reduced disease control. Retrieved September 13, 2017, from http://www.sciencedirect.com/science/article/pii/S0954611111000096 3. Research gate. (n.d.). Clinical Trials of Postnatal Corticosteroids: Inhaled and ... Retrieved September 13, 2017, from https://www.bing.com/cr?IG=6838420C8BF64EF1BB675906106CA6C6&CID=19E5D0F918316E610EAEDA0519376F2E&rd=1&h=D34bYtOLAIZTPJOLPUpPKTQu0vGA0yJYFmZ9qHpaS1s&v=1&r=https%3a%2f%2fwww.researchgate.net%2fprofile%2fHenry_Halliday%2fpublication%2f12906620_Clinical_Trials_of_Postnatal_Corticosteroids_Inhaled_and_Systemic%2flinks%2f56544f1308ae4988a7b01b3d.pdf%3forigin%3dpublication_detail&p=DevEx,5062.1 4. Au, D. H., Curtis, J. R., Every, N. R., McDonell, M. B., & Fihn, S. D. (2002, March). Association between inhaled beta-agonists and the risk of unstable angina and myocardial infarction. Retrieved September 13, 2017, from https://www.ncbi.nlm.nih.gov/pubmed/11888971 5. Lehanneur, Mathieu. (July 2009). Science-inspired design [Video file]. Retrieved from https://www.ted.com/talks/mathieu_lehanneur_demos_science_inspired_design 6. Donohue, J. F., & Ohar, J. A. (2004, February 17). Effects of Corticosteroids on Lung Function in Asthma and Chronic Obstructive Pulmonary Disease. Retrieved September 13, 2017, from http://www.atsjournals.org/doi/full/10.1513/pats.200402-003MS 7. Festic, E., Carr, G. E., Cartin-Ceba, R., Hinds, R. F., Banner-Goodspeed, V., Bansal, V., . . . Levitt, J. E. (2017, May). Randomized Clinical Trial of a Combination of an Inhaled Corticosteroid and Beta Agonist in Patients at Risk of Developing the Acute Respiratory Distress Syndrome. Retrieved September 13, 2017, from https://www.ncbi.nlm.nih.gov/pubmed/28240689 8. Degnan, F. H. (2012, November 01). Clinical studies involving probiotics: When FDA’s investigational new drug rubric applies—and when it may not. Retrieved September 13, 2017, from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3495785/ 9. Wong, E. (2013, April 01). Air Pollution Linked to 1.2 Million Premature Deaths in China. Retrieved September 13, 2017, from http://www.nytimes.com/2013/04/02/world/asia/air-pollution-linked-to-1-2-million-deaths-in-china.html?_r=0 10. How Much Does an Albuterol Inhaler Cost? - CostHelper.com. (n.d.). Retrieved September 13, 2017, from http://health.costhelper.com/albuterol-inhaler.html 11. Drum, K. (2017, June 24). Here's why your asthma inhaler costs so damn much. Retrieved September 13, 2017, from http://www.motherjones.com/kevin-drum/2013/10/heres-why-your-asthma-inhaler-costs-so-damn-much/
|
||||||