BME100 f2017:Group12 W0800 L2
| Home People Lab Write-Up 1 | Lab Write-Up 2 | Lab Write-Up 3 Lab Write-Up 4 | Lab Write-Up 5 | Lab Write-Up 6 Course Logistics For Instructors Photos Wiki Editing Help | ||||||
OUR TEAM
LAB 2 WRITE-UPDevice Image and Description
Value CreationThe ability to rapidly detect contact with a specific bacterium in the field can save the lives of the workers using them. It can save money for the companies by avoiding paying for workers compensation and having to hire new employees to replace one's that were infected. Technical FeasibilityThe technologies that will be require include nanotechnology that interacts with a bacterium as well as a secondary chemical that causes a change in color that detects the binding of the nanotechnology and bacterium. References:(1) Michael Berger. “Nanowerk.” Nanowerk Nanotechnology portal, Nanowerk, www.nanowerk.com/spotlight/spotid=25762.php. Accessed 13 Sept. 2017. Clinical FeasibilityIn a clinic this will be applied to a surgical mask before a physician goes around patients. It will also be used in field settings where humanitarian doctors are exposed to deadly infectious bacterial diseases. a risk with this technology is that the strip is not exposed to a dose large enough to change its color and the physician is unaware. There were clinical trials conducted to detect and diagnose Tuberculosis in patients that were somewhat similar to our product. These trials were conducted for the purpose of improving the detection of Tuberculosis and although the methods were different to our own potential trials, the clinical risks would be similar. This study started in May 2016 and was completed in December 2017 and had an estimated enrollment of 1000 people. One other challenge that this study encountered that we would probably also find is that many patients who have tuberculosis are also people that are in hard-to-reach groups, meaning that these people have backgrounds that prevent them from readily coming to the hospital or participating in a study. An issue in the clinical success of our product is that our product relies on physicians using them in interactions with patients rather than something that the patients directly use. In foreign countries where tuberculosis and other airborne bacterial diseases are most common, this process will depend on doctors visiting patients where they live and this can create a lot of external factors that we cannot control in a trial. These factors could affect our research. Example Clinical Trial- Southern, J. (2015, July 27). Improving the Detection of Active Tuberculosis in Accident and Emergency Departments. Retrieved September 13, 2017, from https://clinicaltrials.gov/ct2/show/NCT02512484 Market AnalysisCost to Manufacture
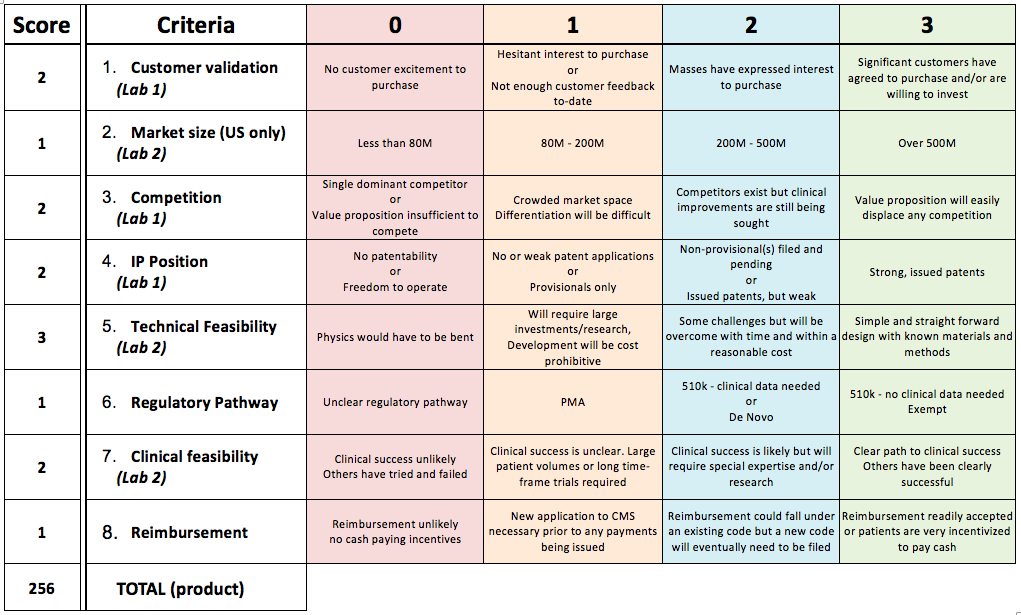
Total cost of production: $0.15 per stripSales Price DescriptionEach strip will cost $0.15 to manufacture. The product will be sold in a pack of 10, which will then cost $1.50 to produce. The sales price of this product will be five times that of production, which makes the cost of a package of ten strips to be $7.50. Sales Price: $7.50 for a pack of ten.Market SizeThe existing number of people in the world who will use this technology is estimated as 7 million. Each of these people will need a pack of the product every 5 days. This results in each person needing 73 packages a year, which will cost $547.50 a year. This multiplied by the number of people using the product per year is 3,832,000,000. Assuming 5% permanence of the market, this makes the market size equal to 191.5 million dollars. Market Size: 191.5 million dollarsMarket Size Score: 1Fundability DiscussionDiscussion:After working through the fundability worksheet, it has been decided that the product should not be funded. The overall score that was found for our product is lower than that which was proposed as a base-level fundable score. It has also been decided that there are many variables that cannot be controlled, some in the marketability and clinical feasibility of the product. The uses and benefits of the product, in theory, do not outweigh the monetary cost to hospitals and individuals.
| ||||||