Colin Wikholm's Individual Journal Assignment Week 3
Exploring HIV Evolution In-Class Activity
Purpose
The goal of this assignment was to learn how to work with filetypes, data of DNA sequences, and Biology Workbench. We performed this assignment to prepare to work with HIV sequences and the software that can be used to analyze it.
Methods and Results
Activity 1, Part 2: GenBank
- Go to the following paper: Patterns of HIV-1 evolution in individuals with differing rates of CD4 T cell decline
- Read through the article until you arrive at the links connecting to the nucleic acid data
- Select one of the records and view it in the full file format and the FASTA format
- Report the accession number of your selected HIV sequence.
- AF016760
- Report who your selected HIV sequence is from. What section tells you this?
- The "DEFINITION" section reports the subject as "subject 1 visit 1 clone 1 from USA"
- Select 4-6 sequences at a time in summary view to download them in FASTA format. Keep track of your file organization.
- Open the saved file with Microsoft Word to check the sequences and ensure that they are in the FASTA format. There should be a '>' followed by the sequence
- Report the accession number of your selected HIV sequence.
Activity 1, Part 3: Introduction to the Biology Workbench
- Go to Biology Workbench
- Sign into account or create one by clicking on "Set up an account"
- Go to "Nucleic Tools"
- On the list of tools, click "Add new sequence" and then select the "Run" button further down the page
- Select "Browse" to use your saved file from NCBI
- Select the file and click "Upload" and show the data on this page
- After the data field shows the labels and sequences, select "Save" to add the data to the session. Check to make sure that the sequences are listed below the analysis tools
- Select your sequences and run the group using "ClustalW"
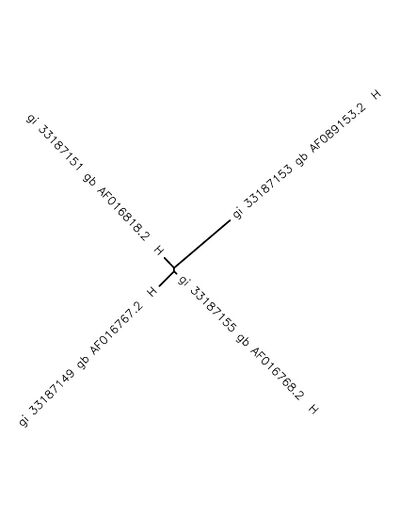
- Analyze the output to see if the sequence is related to how the tree is branched and arranged according to pairwise similarity scores. It may help to use the knowledge you used from Activity 1
The following unrooted tree was produced:
Scientific Conclusion
I believe that this assignment was successful. I learned how to use Biology Workbench (the DNA sequence analysis software) and to create phylogenetic trees for visual analysis. I believe that I will be able to apply the methods I learned in the upcoming HIV project. What is more, I practiced saving and transferring data across differing platforms, something that will be vital for the upcoming project.
Data and Files
Preparation for Week 4 Journal Club
New Vocabulary
- seroconverting (or seroconversion)
- "Development of detectable specific antibodies in the serum as a result of infection or immunization." http://medical-dictionary.thefreedictionary.com/seroconversion
- nonprogressors
- "An individual infected with the human immunodeficiency virus who does not develop worsening immune function or symptoms of active disease." http://medical-dictionary.thefreedictionary.com/nonprogressor
- coreceptors
- "A cell surface receptor, which, when bound to its respective ligand, modulates antigen receptor binding or affects cellular activation after antigen-receptor interactions." http://medical-dictionary.thefreedictionary.com/coreceptor
- nested PCR
- PCR in which "the primers used in the first round of amplification are either both replaced (nested PCR) or only one is replaced (semi-nested PCR) for the second and subsequent cycles of amplification. Increases the sensitivity and specificity of the PCR." http://medical-dictionary.thefreedictionary.com/nested+PCR
- env gene
- "a gene which encodes a protein precursor for the envelope proteins, found in the retroviral genome." http://medical-dictionary.thefreedictionary.com/env+gene
- epitopes
- "antigenic determinant a site on the surface of an antigen molecule to which a single antibody molecule binds; generally an antigen has several or many different antigenic determinants and reacts with many different antibodies. Called also epitope." http://medical-dictionary.thefreedictionary.com/epitopes
- reverse transcription–PCR
- "a reaction applied when the target sequence is RNA, such as viral RNA or messenger RNA. Reverse transcriptase that copies DNA from an RNA template is present in the first round." http://medical-dictionary.thefreedictionary.com/nested+PCR
- monophyletic viruses
- viruses that are "descended from a common ancestor." http://medical-dictionary.thefreedictionary.com/monophyletic
- chemostats
- "A fermenter for microbial growth in which the ratio of growth to synthesis of secondary products is controlled by the rate at which new medium is added to the culture." http://medical-dictionary.thefreedictionary.com/chemostat
- consensus sequence
- "a sequence of nucleotides or amino acids similar or identical between regions of homology in different but related DNA, RNA, or protein sequence." http://medical-dictionary.thefreedictionary.com/consensus+sequence
Outline of Article
- What is the importance or significance of this work?
- HIV is a devastating disease worldwide, leading to AIDS if left untreated.
- Understanding the way that HIV evades host immune systems may allow us to better approach treatments.
- This work was significant because it advanced our knowledge of HIV and its leading to CD4 T cell decline (and thus to the development of AIDS).
- This study improved upon previous studies, which were limited in various ways (see below)
- A greater genetic diversity HIV strains was associated with greater virus progression and greater decline in CD4 T cells.
- This means that HIV strains that show greater genetic diversity should can be identified as a greater threat to development into AIDS.
- PERSONAL NOTE: perhaps this could be used to inform patients of the predicted severity of their infection.
- The study suggests that previous models may be correct:
- Those strains that have greater genetic diversity can produce epitopes that allow for escape of the host immune system.
- Overall, this study gave us a greater understanding of the HIV virus and factors that contribute to its detrimental effects.
- What were the limitations in previous studies that led them to perform this work?
- Previous studies were critiqued for using smaller sample sizes (6 individuals).
- However, this study did not increase the sample size substantially with only 15 individuals (see below for critiques of this study).
- Previous studies were also criticized for not analyzing genetic sequence patterns and for having limited measurements over time.
- This means that the evolution of the viruses could not be tracked with the same precision as in this study.
- Previous studies were critiqued for using smaller sample sizes (6 individuals).
- How did they overcome these limitations?
- This study somewhat overcame these limitations:
- They included 15 individuals instead of 6.
- They ensured that measurements were made over time (every 6 months).
- Lastly, they analyzed HIV genetic sequences directly, focusing on the analysis of the env gene.
- This study somewhat overcame these limitations:
- What is the main result presented in this paper? (Hint: look at the last sentence of the introduction and restate it in plain English.)
- This study found that HIV strains that progress more rapidly to AIDS development (or at least inhibit host immunity) have different genetic diversity dynamics than those that do not.
- Specifically, higher HIV genetic diversity is associated with greater decline in CD4 T cells.
- What were the methods used in the study?
- 15 HIV infected humans were selected to study.
- acquired infection from drug injections
- They divided the people into groups based on HIV progression rates and took blood samples every 6 months.
- started samples at seroconversion
- rapid progressors had fewer than 200 CD4 T cells within 2 years of seroconversion
- moderate progressors had 200-650 CD4 T cells within 4 years of seroconversion
- nonprogressors had greater than 650 CD4 T cells throughout the study
- Nested PCR was used to amply the HIV-1 env gene for analysis.
- Sequencing was performed using the Sanger chain termination method and eventually submitted online on GenBank.
- Reverse transcription-PCR was used to determine viral levels in blood plasma, and the MEGA computer package was used to create phylogenetic trees.
- Genetic diversity was analysed by correlating against CD4 T count.
- Over time, the initial genetic sequences were compared to later sequences with regard to silent/non-silent mutations.
- Subjects 9 and 15 were further analyzed for their high HIV genetic diversity.
- Using phylogenetic trees, they investigated if the individuals had been originally infected by more than one HIVs.
- 15 HIV infected humans were selected to study.
- Briefly state the result shown in each of the figures and tables.
- Figure 1: showed that greater divergence and genetic diversity of HIV sequences was associated with decline in CD4 T cells.
- Table 1: The summary of the data shows homogeneity among early tests.
- However, two individuals had greater starting heterogeneity, suggesting to the researchers that they may have each been infected more more than HIV (later shown not to be true).
- Figure 2: moderate and rapid progressors had greater HIV genetic diversity at any given visit and greater genetic divergence over time than the non-progressors.
- However, divergence was not significant between the moderate and rapid progressors.
- Figure 3: Phylogenetic tree of subject 9 shows no predominant HIV strain over time.
- Early immunity of the host selects against this, but cannot defend from such a wide variety of strains.
- Figure 4: the phylogenetic tree of multiple host HIVs confirm that no one strain predominates, and that in general multiple strains evolve throughout the study.
- How do the results of this study compare to the results of previous studies (See Discussion).
- Previous studies gave results that did not wholly support HIV divergence and diversity for progression.
- Although this previous study have similar results regarding genetic diversity, over time the rapid progressors actually showed less diversity than the slow-progressors.
- As a note, this study did not follow subjects from the first appearance of the virus.
- Although this previous study have similar results regarding genetic diversity, over time the rapid progressors actually showed less diversity than the slow-progressors.
- The other previous study had several counterexamples to correlations between diversity and progression rates.
- The exceptions may have simply had poor immune responses.
- Previous studies gave results that did not wholly support HIV divergence and diversity for progression.
- How do the results of this study support published HIV evolution models?
- Nowak's model was supported by these results:
- It proposed that greater HIV genetic diversity would allow for epitopes to reach outside host immune capabilities.
- It predicts that progressive HIV infections would have higher levels of genetic diversity and thus better destroy host immune systems (as this study seems to support).
- Nowak's model was supported by these results:
- What are the limitations in this study? (your critical evaluation of the study).
- I think the greatest limitation of this study is the small sample size.
- For a disease that infects millions of people, studying only 15 seems like it would not speak to the virus on a grander scale.
- Nonetheless, this study does improve upon the previous studies.
- As a second limitation, this study only evaluated individuals from a very specific cohort (those that acquired the disease via injection).
- Many people acquire the disease through other means such as sex, and ignoring these cohorts may fail to address other factors of HIV progression.
- This study only investigate the env gene.
- There are likely many other genes that influence HIV progression, so only studying a single genetic sequence is a rather narrow (although perhaps the most practical) approach.
- I think the greatest limitation of this study is the small sample size.
- What future work do you suggest?
- I would suggest that future research investigate cohorts such as sexually acquired HIV subjects, genes other than env, and utilize larger sample sizes.
- This would increase the meaningfulness of the paper on a grander scale, increase the dynamic understanding of the virus, and allow more definite conclusions.
- NOTE: This paper was published in 1998, so I predict that these suggestions (and much more) have already been accomplished.
- It will be interesting to see how research has advanced since this article.
- If this research group is still together, it might be enlightening for them to look back on this paper to recognize errors and to learn from their changes in thinking since 1998.
Acknowledgments
I would like to thank my partner, Matthew Oki, for his assistance on working with the Biology Workbench. We worked together face-to-face in-class as well as out of class. Thank you also to Dr. Kam D. Dahlquist for her assistance with software in-class. While I worked with the people noted above, this individual journal entry was completed by me and not copied from another source.
Colin Wikholm 01:39, 20 September 2016 (EDT)
References
- Assignment 3
- CLUSTAL W: Julie D. Thompson, Desmond G. Higgins and Toby J. Gibson, modified; any errors are due to the modifications. PHYLIP: Felsenstein, J. 1993. PHYLIP (Phylogeny Inference Package) version 3.5c. Distributed by the author. Department of Genetics, University of Washington, Seattle.
- Exploring HIV Evolution: An Opportunity for Research
- Higgins, D.G., Bleasby, A.J. and Fuchs, R. (1992) CLUSTAL V: improved software for multiple sequence alignment. Computer Applications in the Biosciences (CABIOS), 8(2):189-191. Thompson J.D., Higgins D.G., Gibson T.J. "CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap penalties and weight matrix choice." Nucleic Acids Res. 22:4673-4680(1994). Felsenstein, J. 1989. PHYLIP -- Phylogeny Inference Package (Version 3.2). Cladistics 5: 164-166.
- Markham, R. B., Wang, W.-C., Weisstein, A. E., Wang, Z., Munoz, A., Templeton, A., … Yu, X.-F. (1998). Patterns of HIV-1 evolution in individuals with differing rates of CD4 T cell decline. Proceedings of the National Academy of Sciences of the United States of America, 95(21), 12568–12573.
Important links
Bioinfomatics Lab: Fall 2016
Class Page: BIOL 368-01: Bioinfomatics Laboratory, Fall 2016
| Weekly Assignments | Individual Journal Assignments | Shared Journal Assignments |
|---|---|---|
|
|
|