Brianna N. Samuels-Week 14
From OpenWetWare
Jump to navigationJump to search
Purpose
- to find r using Arrhenius' equation for 12, 15, 20, 25, and 30 degrees
- investigate whether E should be a function of y
Methods
- Use the Arrhenius equation (rate = A*exp(-B/(R*T)) to model the temperature dependence of the chemostat reaction.
- Figure out the constants A and B from the rate data in the TaiParamsRevised.m file.
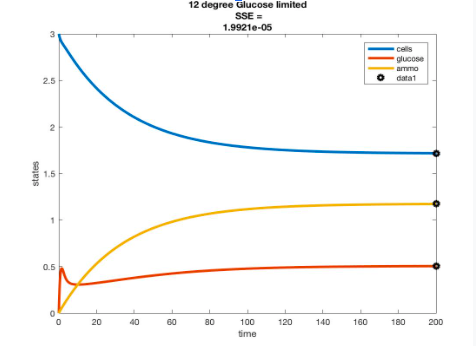
- Simulate the chemostat for T = 15,20, 25 degrees C conditions and graph the time courses of the biomass and nutrients.
- Investigate the glucose efficiency/waste constant (that is not really a constant?) for the glucose-limited and ammonium-limited conditions.
- Note the values of E for glucose-limited and ammonium-limited conditions.
- For each temperature (12, 30), find a function E(y) that matches the two points of (y,E) data.
- Modify the chemostat_2nutrient_dynamics.m file to use the functions you've created.
- Compare the resulting simulation to the previous one.
Your individual journal should detail your efforts, and your 15-minute presentation should cover these topics:
- A brief intro to the chemostat problem of Tai et al (2007).
- The data we've extracted from the paper and other sources.
- Your temperature investigation.
- Your efficiency/waste investigation.
- Discussion and reflection on your findings.
Results
Solving for r
- r=Ae^(-B/RT)
- A = 4.9 * 10^11
- B = 68258.79
- T = 12,15, 20, 25, 30 (convert to Kelvin)
- R = 8.134
- will be using Glucose limited data
- other parameters came from Tai et al. 2007
Investigating E
- determine E is a function of y
- should be modeled as a straight line on MATLAB
- replace in MATLAB wherever we see E with E(y) equation



 File:Biomathematical Modeling Final.pdf
File:Biomathematical Modeling Final.pdf
Acknowledgments
- Homework Partner: Ali texted consistently, met face to face a couple times to work on presentation, as well as attended office hours together
- Attended office hours for Dr. Fitzpatrick
- Except for what is noted above, this individual journal entry was completed by me and not copied from another source.
References
- Dahlquist, K. & Fitzpatrick, B. (2019). "BIOL388/S19: Week 14/15 Assignment Page" Week 14/15 Assignment Page
- Tai, S. L., Boer, V. M., Daran-Lapujade, P., Walsh, M. C., de Winde, J. H., Daran, J. M., and Pronk, J. T. (2005). Two-dimensional transcriptome analysis in chemostat cultures: combinatorial effects of oxygen availability and macro- nutrient limitation in Saccharomyces cerevisiae. J. Biol. Chem. 280, 437–447.
- Tai, S. L., Daran-Lapujade, P., Walsh, M. C., Pronk, J. T., & Daran, J. M. (2007). Acclimation of Saccharomyces cerevisiae to low temperature: a chemostat-based transcriptome analysis. Molecular Biology of the Cell, 18(12), 5100-5112. DOI: 10.1091/mbc.e07-02-0131
- Assignment Pages:
- Assignment Link:
- Class Journal Page:
- BIOL388 Home Page: BIOL388
- User page: Brianna N. Samuels


