Nika Vafadari Week 5
From OpenWetWare
Jump to navigationJump to search
Electronic Lab Notebook Week 5
Purpose
To grasp a better understanding of the mathematics behind nutrient models and learn how to solve for the equations that characterize the non trivial equilibrium of a model.
Work Flow and Methods
Question 1
- compared model one to model two and the nutrient model found in the paper to analyze the similarities and differences in the models
Question 2
- compared the 3 Model Two equations to the solutions for y and q/R
- solved for q first using the third equation in order to plug it in and solve for y solely in the terms of R, V, u, c
- after finding q rearranged equation, dividing both sides by R, to find q/R
- plugged q into equations 1 and 2 to solve for both y solutions
Question 3
- first solved for c1 and c2 using the two equations for y
- plugged in c1 and c2 into the equation for q/R along with the u1 values from the paper u1=29, 44, 61, 66, 78, 90, 96, 114, 118
- solved for y and found two answers using quadratic equation
- plugged in both values of y into the equations for c1 and c2 to see which value gave positive values for c1 and c2
Question 4
- solved for analogous equations using the same method as explained in question 2 workflow
- except this time used the three equations from model 3 to first solve for q/R using equation 3 to find q then solving for the two solutions for y by plugging in q to the first two equations
Results
Question 1
- Explain why Model One, the additive model, may not be the best choice.
- Model One may not be the best choice since it is an "or" model, meaning that both nutrients are not necessary for the yeast to grow. If the concentration of one of the nutrients ends up going to zero, the yeast will continue to reproduce, only stopping reproduction when both of the nutrients are depleted to zero. However, this is not the best model for the journal club article since the yeast required both nutrients, ammonia and glucose, in order to reproduce. For example if one nutrient were to be depleted, such as the ammonia, the yeast would not be able to continue to reproduce continuously even if the glucose remained.
Question 2
- Work out the algebra to show that Model Two’s “non trivial” equilibrium is characterized by the equations below, for yeast, residual ammonium, and residual glucose, respectively.


Question 3
- Set q=0.15, V1=110, V2=180, K1=2.5, K2=10, R=15, u2=100. With these values, and the values of u1 in the journal club paper for ammonium feed concentrations, find the equilibrium values of yeast, residual ammonium, and residual glucose. Recreate the journal club paper Figure 1(a) plot with your findings.
- Calculations for y, c1, and c2 for the various u1 linked below.
- Part 1
- Part 2
- Part 3
- Part 4
- Part 5
- It was found that for some of the values of u1 such as u1=44, both y values gave a negative value for c1, meaning that there is most likely an algebraic error present throughout the solution for y, c1, and c2.
- However, while the algebraic error in the solutions is noted, the data was plotted rendering the following graph.
Question 4
- Are there analogous equations to the ones of Part (2) for Model Three? If so, what are they?
- Found one equation for q/R and two equations for y
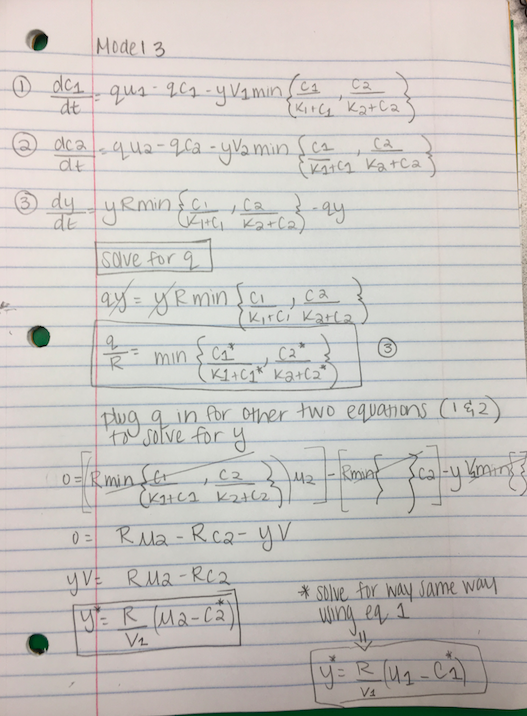
Data Files
Conclusion
- It was determined that the additive nutrient use model was not the best fit for an experiment such as the one conducted in the journal article, which introduced both glucose and ammonia as two nutrients. Comparisons between the nutrient models, such as the multiplicative nutrient model (Model 2) and the rate limited nutrient use model (Model 3,) were thoroughly observed through taking a look at and analyzing the math behind the two models. For example, three analogous equations for q/R and y that characterize the equilibrium of Model 3 were found using the same mathematical method as the one used to solve for the solutions for Model 2.
Acknowledgments
- I communicated with Margaret J. O'Neil over text several times on February, 15, 2017 in order to discuss how to complete questions 2 and 3, specifically when solving for the equilibrium, the values of c1, c2, and y for the various values of u1, and plotting the data.
- Questions were copied and pasted from the Week 5 Exercise handout.
- Except for what is noted above, this individual journal entry was completed by me and not copied from another source.
- Nika Vafadari 01:30, 16 February 2017 (EST):
References
- Dahlquist, Kam D. (2017) BIOL398-05/S17:Week 5. Retrieved from http://www.openwetware.org/wiki/BIOL398-05/S17:Week_5 on 15 February 2017.
- ter Schure, E. G., Sillje, H. H., Verkleij, A. J., Boonstra, J., & Verrips, C. T. (1995). The concentration of ammonia regulates nitrogen metabolism in Saccharomyces cerevisiae. Journal of bacteriology, 177(22), 6672-6675.
Useful Links
- Nika Vafadari
- Course Home Page
- Weekly Journal Entries
- Shared Journal Pages
- Assignment Pages
- Template:Nika Vafadari