Lab 8: Vertebrate Circulation and Respiration
Objectives
- To understand the path of blood flow through adult and fetal mammalian hearts.
- To learn the anatomy of the mammalian heart and respiratory system.
- To understand the mechanism that enables the chambers of the heart to beat in coordination.
- To learn how to record an electrocardiogram (ECG).
- To monitor changes in respiration and heart rate before and after exercise and observe the differences between designated experimental groups of students.
Introduction to Respiration and Circulation: Background
The general pattern of circulation in vertebrates involves a ventrally located heart and a series of blood vessels. Blood moves through these vessels, transporting oxygen via a pigment called hemoglobin, as well as nutrients and soluble waste products such as CO2. Specialized respiratory organs, such as lungs and gills, have evolved to provide oxygen. These organs have large exchange surfaces where a medium (air or water) containing high concentrations of oxygen can come in close contact with blood allowing O2 and CO2 to diffuse across their respective concentration gradients.
In organisms with a heart, a pressure gradient is generated by the contracting heart muscle allowing blood to move through the vessels. Arteries carry blood away from the heart and have thick, highly elastic muscular walls. The thickness of the walls is related to the amount of pressure they must bear and to their diameter. The arteries branch and decrease in diameter until they become very small vessels called capillaries. Capillary walls are only a single cell in thickness and are the site of O2, CO2, nutrient and waste exchange between the blood and the cells of the tissues. The pressure generated by the heart and transmitted to the arteries falls rapidly in the capillaries. The capillaries converge into small veins. The small veins also converge to form larger and larger veins that carry blood back to the heart. The walls of the veins are less thick than those of a similar-sized artery. Veins always have a low pressure.
Blood Flow through the Heart of Fetal and Adult Mammals
Prior to dissecting the calf heart, make sure you review the path of blood flow through the heart in adult and fetal mammals. When you dissect the heart, make sure to identify the underlined terms in this section.
In adult specimens:
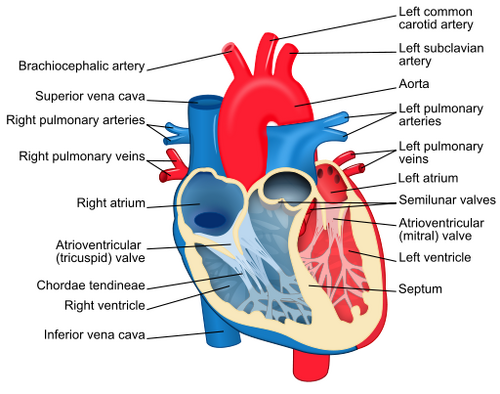
• There are four completely separated heart chambers, two atria and two ventricles.
• Deoxygenated blood returning from the body via the superior and inferior (bipedal terms) or cranial and caudal (quadrupedal terms) vena cava enters the right atrium.
• From the right atrium deoxygenated blood enters the right ventricle.
• Blood then leaves the right ventricle via the pulmonary artery (note that this is the only artery in post-fetal life to carry deoxygenated blood) and is pumped to the lungs where the blood picks up oxygen.
• Oxygenated blood returns from the lungs in the 4 pulmonary veins and enters the left atrium.
• From the left atrium, blood enters the left ventricle.
• Oxygenated blood leaves the heart in the aorta and is transported to the tissues.
In fetal specimens:
• As in adult animals there are four heart chambers: two atria and two ventricles.
• The umbilical vein carries blood rich in oxygen and nutrients and low in wastes from the placenta to the right atrium of the heart of the fetus (review the ductus venosis from lab 7). Because separation of the atria in a fetal mammal is incomplete, most of this oxygenated blood can pass directly to the left atrium, to the left ventricle, and then out the dorsal aorta to the body of the fetus. See if you can find the foramen ovale, the small opening between the right and left atria that will close after birth.
• Some of the blood from the right atrium enters the right ventricle. In the adult, blood in the right ventricle would pass through the pulmonary arteries to the lungs for oxygenation before returning to the left side of the heart. The fetal circulation can by-pass the lungs because any blood that reaches the right ventricle and pulmonary artery is shunted via the ductus arteriosus to the dorsal aorta. The spincter in this portion of the pulmonary artery will tighten and close shortly after birth. You should be able to find this opening from the pulmonary artery to the aortic arch in your specimen.
• After servicing the body, the oxygen-depleted blood leaves the fetus via the umbilical arteries, and is re-oxygenated in the placenta where gas, waste and nutrient exchanges between the fetus and mother occur.
Calf Heart Dissection
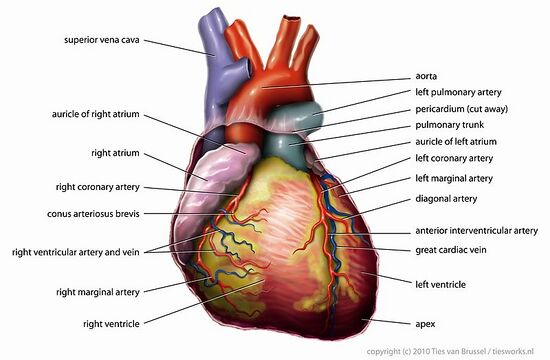
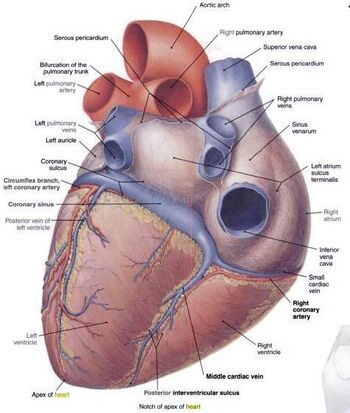
1. Examine the external anatomy of the heart (Fig. 8.1). The auricles are the ear-like structures of the atria. The ventricles are the more muscular parts of the heart located beneath the atria. In the thoracic cavity, a thin membrane called the pericardium encloses the heart. This membrane will probably have to be removed from the calf hearts.
2. Find the coronary arteries on the surface of the heart. The coronary artery that runs diagonally across the heart, on the ventral (belly) side, is a landmark that will help you identify the right and left ventricles. The left ventricle is caudal to (beneath) this coronary artery while the right ventricle is cranial to (above) it.
The function of the coronary artery is to deliver oxygenated blood to the heart. The coronary arteries leave the aorta shortly after it exits from the left ventricle. If they get blocked a heart attack, called a myocardial infarction, will follow. In a coronary by-pass operation, one or more of these arteries are replaced.
3. Start with the ventral surface of the heart in order to identify the pulmonary arteries and the aorta. The brachiocephalic artery might still be connected to the aorta. You should also be able to see the ductus arteriosus (duct between the arteries) which is not completely obliterated in the heart of young calves. Turn the heart around. Identify the two vena cavae, and the pulmonary veins. With your probe follow the passage of the blood from the vena cavae and the pulmonary veins to the atria as outlined in the section above. Some of these vessels may have been removed. Note the thicker, more muscular walls of the arteries in comparison to the walls of the veins.
4. Once you feel that you can identify all the external features of the heart, you may begin to dissect the heart. With a scalpel cut a section off the tip of the heart about one inch from the bottom. Look at the cross section of the ventricles and notice the difference in the thickness of the muscle of the two ventricles. Remember that the right heart pumps blood only to the lungs whereas the left heart pumps to the entire body.
5. With scissors longitudinally cut each ventricle up to the level of the atria. Note the difference in the thickness of the ventricular walls and find the ventricular septum. You should now be able to identify two sets of valves in the heart which prevent back flow of the blood (Fig. 8.2).
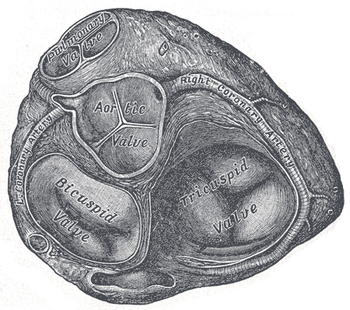
a) The atrioventricular (AV) valves are located between the atrium and the ventricle on each side of the heart. In the right heart the AV valve is called the tricuspid valve (three flaps). In the left heart the AV valve is the mitral (or bicuspid) valve (two flaps). These valves prevent back flow of blood into the atria upon contraction of the ventricles. The AV valves are anchored by the chordae tendineae that prevent the valves from being turned inside out. The chordae tendineae are attached to the papillary muscles. The papillary muscles are part of the wall of the ventricle.
b) The semilunar valves (half-moon in appearance) are located at the openings of the ventricles into the aorta and pulmonary artery. They are known as the aortic and pulmonary (pulmonic) valves respectively. These valves close when the ventricles relax, just after ventricular contraction to prevent back flow of blood from the vessels leaving the heart.
c) In the right ventricle of the calf heart, you may also find a band of tissue stretching across the ventricle. This is known as the moderator band and it carries the right bundle branches to the right ventricle.
6. Use scissors to cut open the aorta. Notice the incredibly smooth inner surface. If you continue to cut until you are close to the base of the aortic arch, you will have a good view of the ductus arteriosus and the aortic semilunar valve.
Look at the heart/lung preparation on display in the lab in addition to your dissection. Before you continue to the next section of the laboratory, be sure that you understand the anatomy and function of the heart. You should be able to trace the flow of blood through the heart and to the lungs in adult and fetal mammals.
A
B
Figure 8.1A-B: A. External anatomy of the human heart, Ventral View. B: External anatomy of the human heart, Dorsal View. Picture courtesy of www.anatomytopics.wordpress.com
A
B
Figure 8.2A-B: Diagrammatic Section of the Internal Anatomy of a Mammalian Heart. A. Blood Flow through the heart chambers. B. Valves of the human heart
Coordination of Heart Contraction and The Electrocardiogram: Background
It is essential that the muscle fibers of the heart contract in a rhythmic and orderly fashion for proper working of the pump, otherwise blood would be sloshed around in the ventricles instead of being ejected efficiently into the large vessels.
Contraction of heart muscle is initiated by depolarization (change in the membrane potential also known as an action potential) of the muscle membrane. A wave of depolarization, or excitation, is initiated in the sinoatrial (SA) node, located in the right atrium. This excitation spreads throughout both atria causing their contraction. The excitation (depolarization) is picked up by the atrioventricular (AV) node, located at the base of the atria. The AV node is connected to a bundle of specialized fibers that run directly to the muscle tissue in the tip (apex) of the ventricles. This bundle of fibers is the only electrical connection between the atria and the ventricles. After leaving the AV node the excitation travels in these Purkinje fibers through the interventricular septum to the apex of the ventricles. The anatomical arrangement of these fibers guarantees that contraction begins at the tip of the ventricle thereby moving blood up and out of the heart and into the pulmonary artery and the aorta. In summary, the initiation of excitation in the SA node results in contraction of the atria, followed by contraction of the ventricles. A slight delay exists between contraction of the atria and the ventricles that allows for complete emptying of the atria before the ventricles contract (Fig. 8.3).
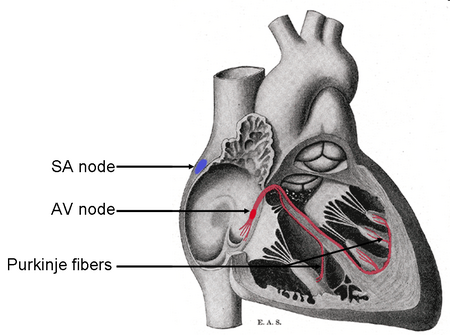
Figure 8.3: Diagram of the conducting system of the heart
The wave of depolarization or excitation may be recorded at the surface of the body by electrodes placed on the skin. It is necessary to have three electrodes: two recording electrodes, one on either side of the heart, and a third electrode that acts as a ground or reference electrode. The pattern of change of potential recorded by the electrodes is called the electrocardiogram and represents the successive depolarization of the parts of the heart.
The ECG has 3 components (Fig. 8.4):
- The P wave represents atrial depolarization.
- The QRS complex represents ventricular depolarization. Atrial repolarization is concealed by the QRS complex.
- The T wave represents ventricular repolarization.
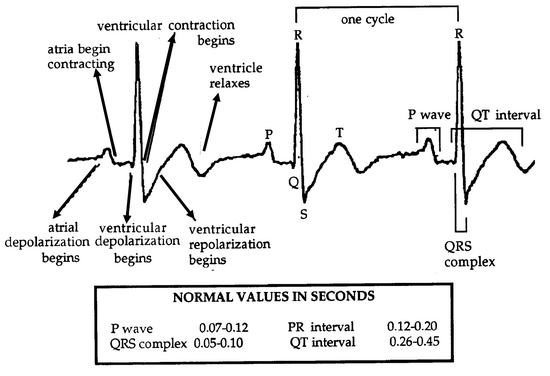
The P-Q interval is the length of time required for the excitation to travel from the SA to the AV node. The T-P segment is the time period between the end of the T wave and the beginning of the P wave when the heart is resting and refilling with blood. This period is also known as diastole, while the contraction is known as systole (Fig. 8.4).
In analyzing an ECG, time periods that include deflection waves are called intervals, and time periods that exclude deflection waves are called segments. For example, the P-T interval is the time period from the beginning of the P wave through the end of the T wave and would include all the electrical changes in one cardiac cycle. The T-P segment would begin at the end of the T wave and stop at the start of the P wave. Since this is the time period after ventricular repolarization and before the next atrial depolarization, it is the time between stimulations, the rest period. The T-P segment plus the P-T interval should equal one entire cardiac cycle, most easily measured as R-R.
Forming Hypotheses About Heart and Breathing Rates Before and After Exercise
How do the heart and lungs respond to the demands of exercise?
When you start to exercise, the muscles need more energy, so the heart and lungs need to deliver more oxygen rich blood to the exercising muscles. With each breath the lungs draw in a volume of air (tidal volume) that is only a small part of the total lung capacity. The volume of air in the lungs can be increased by breathing deeper to draw from the inspiratory reserve volume, or by increasing the frequency of breathing (increasing breaths/min), or both (Fig. 8.5).
Theoretically the heart could increase the quantity of blood delivered to the tissues by increasing the volume of blood per beat (stroke volume), increasing the heart rate, or both. In actuality, it is easier to increase heart rate than to increase stroke volume on demand.
Stroke volume, however, does vary among individuals. As people exercise and become more fit they tone their muscles, particularly in the extremities (arms and legs). These toned muscles contract more effectively and thus are more efficient at helping blood at low pressure in the veins get back to the heart. As the volume of the blood filling the heart during diastole increases, the heart itself often increases in size to accommodate the larger volume. If there is more blood in the heart during systole, the stroke volume increases, sending more blood to the lungs to pick up oxygen with each contraction and sending more oxygenated blood to the tissues of the body. Thus in fit individuals the rate of breathing and the heart rate can be lower while supplying an adequate supply of oxygen to the tissues. You can measure roughly the "degree of fitness" of your subjects by monitoring the breathing and heart rates.
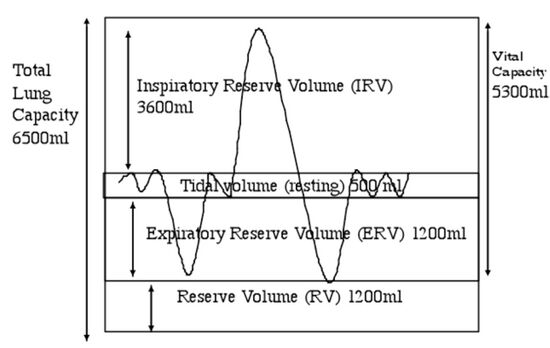
Fig. 8.5. Lung volumes in milliliters. The resting volume of air exchanged by the lungs is the tidal volume in and out of the average lung with each breath, the inspiratory reserve volume can be used in times of greater oxygen needs and represents the exchange you can force when you breath in very deeply and exhale with effort.
Measuring Heart and Breathing Rates Before Exercise
Before collecting data, the class will be divided into experimental groups by their instructor. If you choose to be a human subject, you must sign and hand in the Informed Consent form.
Draft a data table in your lab notebook in which you will record heart rate (beats/min), R-R (s), P-T interval (s), and T-P segment (s) for each individual tested, both at rest and immediately after exercise. You can also use this table to calculate the % of each total R-R represented by each subunit.

Draft a second data table in which you will record R-R (s), Heart Rate (beats/min) and Breathing Rate (breaths/min) for each individual for each of these time periods: before exercise, immediately after exercise, and 1, 2, 5, and 10 minutes after exercise.
To record the ECG, electrodes are attached to the surface of the skin. Because the size of the potentials recorded on the skin are very small, they need to be amplified. The leads from the electrodes are connected to the PowerLab which amplifies the signal where it is displayed on the computer using ADInstruments' software called Chart.
Collecting Data Before Exercise:
1. Attach the sticky electrodes to both inner wrists and your left ankle bone. Be sure the ankle (ground) electrode is firmly attached and over the bony part of your ankle. If you need to replace an electrode for any reason, be sure to wipe off all excess gel and then replace the electrode.
2. Attach the electrode wires to the electrodes: Start with white on right wrist and the green (ground) on your left ankle bone, black to left wrist.
3. Open the ECG settings icon on the desktop. If you cannot find this icon speak to the instructor.
4. Seat the subject in a restful position without any tension in her muscles.
5. Once the subject is connected and sitting very still you can start collecting data by clicking start in the lower right and observing the ECG.
Note: If there is a lot of "noise" in your data so that it is hard to identify the peaks, be sure that the subject is absolutely still and the electrodes are firmly attached with no excess gel.
If you do not like the appearance of the ECG or cannot see some peaks, you can make further adjustments to the initial settings while collecting data.
You can adjust the Y axis by changing the range on channel 3. For instance, if you decrease the range setting you decrease the volts/division on the Y axis and the peaks will appear larger. You can adjust time on the X -axis. For instance, a larger time/division on the X -axis would cause the peaks to be less spread out. Be sure the time scale is sufficiently expanded so that the time periods will be easily measured. (You may want to change the ratio at the bottom of the recording to 2:1).
6. When you are satisfied with the ECG press STOP. If you want to get another page of ECGs so that you have more to choose from, press START again. Each recording is automatically saved and can be retrieved by clicking the numbered tabs at the left of your screen. You can add a description of the ECG by selecting COMMAND; ADD COMMENT.
7. Measure the breaths/minute of the subject at rest. Count the breaths by putting your hand lightly on the subject’s back and counting the movements of her chest up and down for 15 s and multiply by 4. Record this datum in your data table.
8. Measure R-R and the P-T interval of your selected ECG. You can measure the various time points directly from the ECG screen or you can highlight a section of the screen and magnify it by using the magnifying glass on the upper left hand side of the menu. To measure the various time periods drag the "M" marker from the lower left of the screen up to the first position. If you lose the "M" marker, you can get it back by double-clicking in its "home" box in the lower left of the screen. Then put the cursor on the next position and you will get the difference (∆ time) in the upper right. Subtract the time you recorded for the P-T interval from R-R to get the duration of the T-P segment.
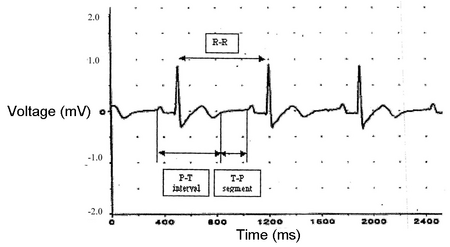
Figure 8.6. ECG Recorded using PowerLab. A three lead recording of the hearts electrical activity. A typical ECG of a healthy subject will show this pattern of waves and segments.
Measuring Heart and Breathing Rates After Exercise
1. The subject should disconnect the electrode cables, but leave the sticky pads in place.
2. Have the subject jog for five minutes (do not record on the computer while the subject is jogging as there will be too much "noise".
3. Get ready to record the subject's ECG and to measure her breathing rate as soon as the exercise period ends.
4. When the exercise period ends, reconnect swiftly the electrodes to the pads and start the LabChart ECG file. Let the recording run without interruption for 10 minutes. Be sure to save this ECG for analysis.
5. Measure the respiration rate of the subject immediately after exercise. Count the breaths for 15 s and multiply by 4. Repeat at 1, 2, 5 and 10 min after exercise. Record the values in the appropriate table.
6. Analyze the ECG immediately after exercise and after 1, 2, 5, and 10 minutes. For each selected time, measure R-R and P-T and subtract the interval P-T from R-R to obtain segment T-P.
Data Analysis of Heart and Breathing Rates Before and After Exercise
After all the recordings are collected, measure all the required intervals for each of the ECGs at each time point and enter data in your tables and in the class data table.
To evaluate the ways in which the electrocardiogram changes as heart rate increases, in particular, which parts of the cycle are responsible for the change in the length of a single trace and the increase in heart rate:
- Compare the baseline value of (R-R) with the value of (R-R) after exercise. By what increment does the (R-R) change? In which direction?
- What percentage of (R-R) is represented by P-T in the baseline? After exercise? By T-P? Why might these values change in the direction they do?
Perform the appropriate statistical tests to compare heart rates before and immediately after exercise among your experimental groups and to compare breathing rates before and immediately after exercise among your experimental groups.
Assignment
1A. Create a scatter graph showing the mean heart rate (HR) and respiratory rate (RR) of the subjects over time. Plot the resting value at the origin (Time 0). Convert time to minutes for the X axis. The respiratory rate should be on a second y axis and the axis scale adjusted so RR does not overlap HR. You can find instructions for adding a second axis and adjusting the scale in the stats & graphing folder. Add the caption.
1B. Compare mean heart rates at rest and immediately after exercise (do the same for mean respiratory rates) using JMP .
1C. Write a results text that describes your findings.
What patterns do you see in HR and BR during exercise?
2. Take the Pre-Lab quiz prior to next lab
Prepare to discuss the following concepts next lab.
3. Examine the class ECG table of data:
- Compare the duration of the P-T interval, and the T-P segment values at rest and immediately after exercise.
- Which parts of the cardiac cycle are most affected by exercise in your data? Least affected? Why?
4. If "Cardiac Output (the total volume of blood pumped per minute by the heart) = Stroke volume x Heart rate" can you explain what effect athletic training might have on an individual's stroke volume and heart rate.
5. Examine Figure 8.7 A-C. Which parts of the cardiac cycle are affected in each of the three examples? Be able to explain your answers.
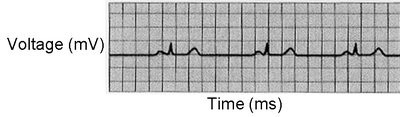
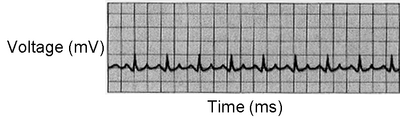
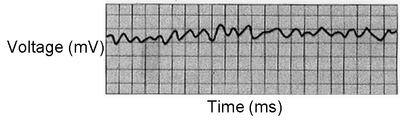
Fig. 8.7A-C. 12 electrode ECG recordings showing three different patterns of heart activity. A) A recording of a resting ECG on a 23 year old female athlete. B) A recording of the athlete in A at the peak of rigorous exercise. C) A recording of a hospitalized patient with ventricular fibrillation.
The heartbeat of an individual with bradycardia is slower than usual (46 beats/min compared to the average college student's 80 BPM heartbeat) (Fig 8.7A). It is not unusual for athletes in top shape to have discernibly slower heart rates, why?
This ECG shows an accelerated heart rate of an athlete while performing rigorous exercise (136 beats/min)(Fig. 8.7B). Tachycardia can also be caused by taking stimulants, such as caffeine or nicotine, or by some physiologic factor, like hyperthyroidism.
When ventricular contractions become completely uncoordinated, the pumping of the blood can stop and the pattern of the heart's electrical signals is lost (Fig. 8.7C). As a result of the cessation of blood flow, brain damage or death can occur. Drug overdoses and other traumas to the heart can cause fibrillation.
Note: Material from this lab will be included in the lab practical. Be sure you are familiar with the basic principles and the anatomy of the heart and how ECG deflection waves relate to cardiac events and exercise. Be sure you can graph and interpret the data and know how to perform the appropriate statistical tests.
Other Labs in This Section
Lab 7: Vertebrate Anatomy
Lab 9: Conduction Velocity of Nerves
