Koch Lab:Protocols/Unzipping constructs
| Home | Research | Lab Members | Publications | Protocols | Contact | Funding |
| Principles | Data | Notebooks | Links | Meetings | Presentations | Inventory |
Principle
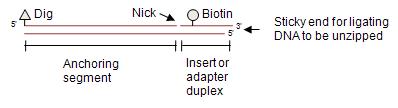
If DNA is attached to a surface (e.g. a coverglass) at one end and to another surface (e.g. a microsphere) in the middle, the DNA can be unzipped if there is a nick between the two attachments. This was first shown by Bockelmann, Essevaz-Roulet, and Heslot in the mid-1990s (PMID 9342340). We describe here a versatile adaptation first described in Koch et al. 2002 (PMID 12124289).
Compared to DNA constructs for end-to-end DNA stretching (see, e.g. labeling DNA by PCR unzipping constructs are more challenging to produce. The method we describe here has a significant stretch of double-stranded DNA (dsDNA) between the first and second attachment labels (dig and biotin). A simpler construct can be made by directly hybridizing two end-labeled DNA oligos, producing a fork construct. (See, e.g. Koch_Lab:Protocols/Fork unzipping constructs.) However, this produces shorter tethered particles, which was disadvantageous for the optical tweezers systems we were using.
The key to this method is that unzipping of a variety of downstream DNA molecules can be carried out with very little modification to the protocol. This versatility was leveraged by Jiang et al. 2004 (PMID 16337600), Shundrovsky et al. 2006 (PMID 16732285), and Johnson et al. 2007 (PMID 17604719).
General thoughts
- The anchoring segment (typically about 1 kilobase pairs (kb) in length) is convenient for producing long initial tethers. However, the structural stability of this dsDNA anchoring segment sets an upper limit of below 60 pN (to assure that the dsDNA anchoring segment does not undergo force-induced melting or "overstretching").
- As shown in figure above, the complementary overhang is provided by the biotin-labeled strand of the "insert duplex" (or "adapter duplex"). This is for a 3' overhang. For a 5' overhang, the bottom strand will be longer. Different sticky ends can be created by switching out top or bottom strand.
- Protein binding sites can be engineered directly into the insert duplex and some experiments can be carried out without need for ligation of downstream unzipping segment.
