BIO254:Adaptation
Light adaptation
The animal eye has a remarkable ability to adapt to a vast range of light intensities. The difference between a starry night and a sunny noontime corresponds to a ~10^11-fold change in light intensity, yet we can see clearly across this range. Differences in the firing rates of RGC neurons can span only about two orders of magnitude, so additional mechanisms have evolved to mediate light adaptation. These mechanisms must solve two problems: they must be able to adapt to different mean levels of background light, and they must be able to maintain sensitivity to light and shadow when so adapted.
Several processes help generate this remarkable capability, and we will begin with a description of how light adaptation changes the visual response. Second, we will discuss the mechanism for this adaptation, first in the photoreceptor itself, and also briefly the process of light adaptation in the nervous system itself.
The phenomenon of light adaptation
The response of a photoreceptor to dim flashes of light can be measured by observing the change in current flowing into the cell. This change is generated because photoisomerized Rhodopsin activates a phosphodiesterase (PDE) to hydrolyze cGMP, and this decrease leads to the closing of cGMP-gated ion channels. Notably, photoexcitation thus decreases ion flow, and can be seen as a transient reduction in the “dark current” flowing into the cell.
This method allows us to determine the sensitivity of the cell (ie., the dimmest flash that produces a response), and also the dynamic range of the cell (ie., the brightest flash that does not saturate the response). A rod cell that is adapted to darkness is enormously sensitive to very dim flashes, and can generate a response to a single photon; however, the response of the dark-adapted cell also saturates quite readily to relatively dim flashes.
As the photoreceptor adapts to greater levels of constant light, it must maintain the “dark current” at a relatively high level – in spite of constant rhodopsin activation - in order to reliably report any further change in light levels. This process can be thought of as a sort of homeostasis. In addition, the photoreceptor must also reduce its response to further flashes, so that it can determine their relative intensity. This process, which maintains a fairly constant level of contrast across a wide range of light intensities, is called desensitization or response compresssion. Other phenomena tend to occur with light adaptation, and include a quicker termination of the light-induced signal. In general, as we shall see, all of these processes serve to reduce the normally extensive amplification of light stimuli by the photoreceptor.
Much of the work that follows relies on measurements in rod cells; this is largely because their size makes them easier to work with, and thus more is known. However, much of what we learn from light adaptation in rods is likely to hold true in cones. Further phenomena of adaptation that occur in the nervous system will be discussed below.
Adaptation in photoreceptor cells takes advantage of the endogenous signaling cascade that underlies photoexcitation of the cell. It does so in several ways, but the most prominent involves the influx of Ca2+ after illumination; this increase in [Ca2+]i leads to several biochemical changes in the signaling pathway that maintain its sensitivity in the presence of background light. Other mechanisms may also act simultaneously, including the movement of several signaling proteins into and out of the cellular compartment where the visual signal is transduced.
Calcium signaling in the photoreceptor
The mechanism for light adaptation in rod cells appears to rely primarily on a diffusible second messenger. The evidence for this takes two forms. First, it has been noted that a very low level of illumination, bleaching only tens of rhodopsins per second, can desensitize the entire rod within a few seconds – this suggests that some molecule must rapidly diffuse to allow desensitization of the hundreds to thousands of disks in the rod cell. Second, direct measurement from a rod cell that is illuminated only in a small spot suggests that disks as far as 20uM from the illumination are desensitized; this would almost certainly require the diffusion of some agent into the remainder of the phototransduction compartment.
This diffusible compound appears to be the calcium ion, Ca2+. Removing Ca2+ from the photoreceptor cell does not normally affect the initial current flow in response to light, but both prevents the termination of that current flow and also increases its maximal response. This is suggestive because adaptation to background light normally shortens the response and diminishes the maximal response; removing CA2+ from the cell has precisely the opposite effect.
How might Ca2+ levels affect light adaptation? As the rod cell is stimulated by light, cGMP hydrolysis causes the cGMP-gated ion channels to close, which lowers the concentration of Ca2+. This lowered [Ca2+] probably acts directly to alter three processes in the signal transduction cascade: decreasing the activity of phosphodiesterase (PDE); increasing the activity of guanylate cyclase; and altering the sensitivity of the cGMP-gated channel.
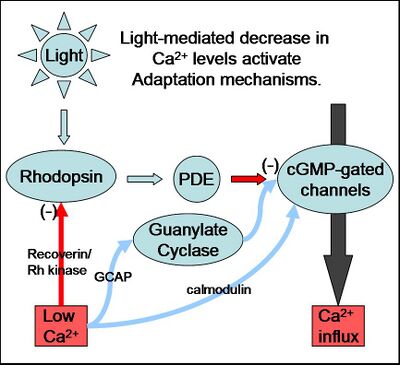
Figure 1: The various mechanisms of Ca2+ mediated adaptation are shown here, with activating steps shown by blue arrows and inhibiting steps shown by red arrows. Illumination of a photoreceptor results in a drop in intracellular calcium, which results in negative feedback onto the signalling cascade.
Ca2+ effects on Rhodopsin and PDE
The enzyme phosphodiesterase acts in the phototransduction cascade to hydrolyze cGMP into GMP when stimulated by light. Stimulation of PDE occurs through the binding of the alpha subunit of the transducin G-protein complex, and the resulting decline in [cGMP] results in a reduced current flow through the population of cGMP-gated ion channels.
PDE has been traditionally considered as one of the crucial steps in amplification, with the activation of a single Rhodopsin molecule leading to the hydrolysis of 105 – 106 cGMP molecules. The termination of this activity is crucial to limiting photoexcitation, and probably occurs through the inactivation of the Gα subunit. However, Kawamura and colleagues identified a protein, known as recoverin, that prolonged the activation of PDE in the presence of high [Ca2+].
Interestingly, recoverin is not actually associated with PDE; instead, recoverin interacts with Rhodopsin kinase to regulate the activity of photoactivated Rhodopsin. In low light levels, high [Ca2+] allows recoverin to bind to Rhodopsin kinase, which prevents the phosphorylation and inactivation of Rhodopsin – extending and amplifying the signal. When the cell is adapted to light, lower [Ca2+] levels prevent recoverin binding, and Rhodopsin kinase quickly inactivates Rhodopsin. The major effect of this is to shorten the response and maintain dynamic range when the rod is adapted to background light.
This control of PDE through the activity of Rhodopsin kinase shows that a major part of adaptation is the Ca2+-dependent regulation of the amplification steps in phototransduction. However, it should be noted that the high activity of PDE under strong background illumination diminishes the cell’s response to another flash of light in a Ca2+ independent manner – ie., the PDE machinery is simply saturated. Thus, the Ca2+-dependent modulation of PDE activity through Rhodopsin kinase serves to preserve what little sensitivity remains in that part of the phototransduction cascade.
Ca2+ effects on guanylate cyclase
A clever experiment by Matthews (1997) was able to distinguish the role of calcium in mediating an “early” and “late” component of the phototransduction cascade that acts in light adaptation. The early component has been verified to be a change in PDE activity, but it is complemented by a later change in the activity of guanylate cyclase that is similarly responsive to Ca2+ levels.
Guanylate cyclase normally antagonizes the activity of PDE; when light-induced activation of PDE leads to cGMP hydrolysis and a drop in [cGMP], guanylate cyclase is activated to replenish the molecule and ultimately to re-open cGMP-gated channels.
The activation of guanylate cyclase is regulated by two small Ca2+-binding proteins called Guanylate Cyclase Activating Proteins (GCAP) 1 and 2. These proteins are normally dissociated from guanylate cyclase in dark-adapted (and high [Ca2+]) conditions, but bind to and activate guanylate cyclase when Ca2+ levels fall in response to light.
Guanylate cyclase is normally slightly activated by a single flash of light; however, sustained illumination activates guanylate cyclase persistently, through a change in Ca2+ levels, and this activity helps keep cGMP-activated channels open and maintain sensitivity to further changes in light.
Ca2+ effects on cGMP-gated channel sensitivity
In addition to the targets above, light-induced changes in Ca2+ levels alter the sensitivity of the cGMP-gated channel. Specifically, low [Ca2+] increases that channel’s affinity for cGMP, in effect increasing current flow to replenish Ca2+ and terminate the light-induced signal.
This effect depends on the activity of calmodulin, a common mediator of intracellular calcium signaling. Calmodulin is an EF-hand protein that can bind up to four Ca2+ molecules simultaneously; in doing so, it undergoes a dramatic conformational change that can alter its interaction with other proteins. Calmodulin binds to the beta subunit of the cGMP-gated channel, but only when Ca2+ levels are high. When Ca2+ levels drop in response to photostimulation and channel closing, calmodulin loses some fraction of its bound Ca2+ and dissociates from the channel. This release increases the channels’ affinity for cGMP, and the channel opens further.
This mechanism may contribute a great deal to adaptive variations in the inward current, with changes in [Ca2+] altering the inward current by up to an order of magnitude. In rod cells, this effect probably acts most at relatively high levels of background illumination, and thus serves to preserve sensitivity and avoid saturation. However, in cone cells the effect may be important for a broad range of light levels, and may not act through calmodulin at all (Burns and Baylor, 2001).
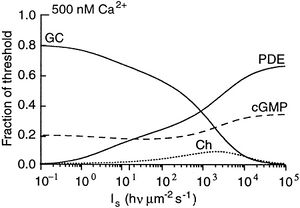
Figure 2: Fractional contribution of Ca2+-dependent mechanisms of adaptation (y axis) at a variety of light levels (x axis). GC corresponds to the modulation of guanylate cyclase; PDE, Ca2+-dependent modulation of PDE; Ch, Ca2+-dependent modulation of cGMP-gated channels; and cGMP corresponds to the non Ca2+-dependent buffering of cGMP from increased PDE activity. The x axis is mesaured in incident photons per square micrometer per second. From Fain et al., 2001.
Ca2+-independent mechanisms of adaptation
Recently, several interesting discoveries have revealed mechanisms of light adaptation that do not depend on Ca2+-sensitive changes in protein activity. Adaptation to varying light levels also appears to depend on the translocation of various signaling proteins into and out of the phototransduction compartment.
Some of these proteins migrate into the phototransduction compartment at high light levels. For example, transducin, the G-protein that is directly activated by Rhodopsin, appears to translocate into the rod outer segment in the dark, which may help the cell respond to single-photon events.
Other proteins appear to translocate to the phototransduction comparment at high light levels, and probably act in light adaptation to maintain sensitivity. Fittingly, most of these help limit or terminate the photoresponse. These include arrestin, which normally serves to terminate the photoresponse by binding activated Rhodopsin; and recoverin, which activates rhodopsin kinase to prevent the activation of PDE.
Much of the work to characterize these molecules has been performed in Drosophila melanogaster, and it is possible that some effects (such as arrestin translocation in particular) may not actually mediate light adaptation in vertebrate systems. (It is possible that the translocation simply replaces arrestin molecules that are bound to activated Rhodopsin, and simply replenishes the supply, rather than altering signal transduction more fundamentally.) Invertebrates also show another system that probably does not occur in vertebrates, which is the translocation of the light-sensitive channel itself (that is, TRPL in D. melanogaster).
In summary, it seems that while Ca2+ plays a crucial role in regulating light adaptation in the photoreceptor cell, other mechanisms actually change the stoichiometric ratio of signaling components in response to light adaptation.
In addition, we should note that most of the Ca2+-dependent mechanisms listed above are vital for vertebrate rod cells, but may not play a role in cone cells. Instead, simple bleaching of the photopigment prevents sustained activation, and appears to be sufficient to maintain activity. While this would be problematic in rod cells, in which we would expect the activated rhodopsin to saturate the transduction machinery, that appears not to be the case in cone cells.
Light adaptation in the nervous system
The photoreceptor plays a vital role in mediating light adaptation, but other mechanisms help visual perception maintain sensitivity in a variety of light conditions.
The most obvious of these (in chordates) is the pupil. The opening and closing of this iris is controlled by retinal ganglion cells projecting to the pretectal nucleus – bypassing most of the processing circuitry normally associated with vision. This reflex closes the pupil when light levels increase, and helps maintain normal vision.
In addition, the eye itself has two populations of photoreceptors that are specialized for vision in dim and bright light: the rods and cones. Rods, in particular, are enormously sensitive to low light levels, but saturate at higher light levels once Rhodopsin begins to bleach persistently. Cones, on the other hand, lack the collecting volume of rods as well as the amplification machinery, and mediate higher light levels. Their resistance to bleaching helps contribute to their effectiveness at high light levels. Rods and cones differ in their adaptation to a dark environment; typically, cones adapt faster but relatively little, while rod adaptation provides the bulk of the ultimate change in sensitivity.
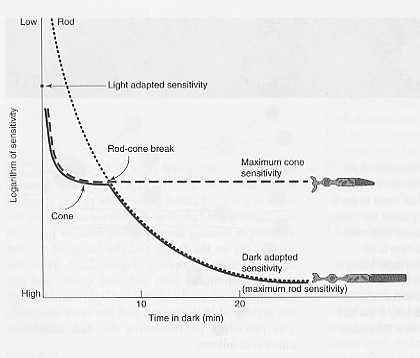
Figure 3: The relative contribution of rod and cone adaption. Cone adaptation is fairly rapid, but rod adaptation is more significant when the eyes are fully acclimatized to low light.
Part of this difference in adaptation between cones and rods may be explained by differences in their calcium homeostasis: the cGMP-gated channels in cones conduct CA2+ at a faster rate than those in rods (Frings et al, 1995), and Ca2+ efflux in cones is five to eight times faster than that seen in rods (Miller and Korenbrot, 1994). This faster Ca2+ response may underlie part of the rapid adaptation seen in cones. However, other differences between cones and rods are also responsible. Cowan et al. (1998) reported that cones have a significantly higher level of RGS9 than rods. RGS9 is a GTPase accelerating protein that increases the ability of transducin’s alpha subunit to hydrolyze bound GTP to GDP; in doing so, it increases the rate at which transducin releases the inhibitory subunit of PDE to quiet the light response. Because of their high RGS9 levels, cones are able to respond more quickly to changes in illumination, but, as seen in the figure above, they have lower peak sensitivity than rods. Interestingly, mutations in RGS9 are responsible for a human disorder in which patients are impaired in their adaptation to luminance changes (Nishiguchi et al., 1998). The patients reported being temporarily blind for up to 10 seconds after moving from a dim to bright environment, emphasizing the importance of RGS9 as a mediator of light adaptation.
Finally, it is useful to note that the higher levels of visual processing also adapt to their stimuli, including varying light levels. One mechanism to compensate for this variation is the series of small eye movement, called saccades, that generate slight shifts in the focus of the eye. These are important because they change the stimulus given to each region of the eye and also to higher neurons, and thus prevent adaptation from reducing the visual scene to a gray haze.
References
Arshavsky V (2003). Protein Translocation in Photoreceptor Light Adaptation: A common Theme in Vertebrate and Invertebrate Vision. Sci. STKE 204:pe43.
Arshavsky V, Lamb T, and Pugh E (2002). G Proteins and Phototransduction. Annu Rev Physiol. 64:153-87.
Bähner M, Frechter S, Da Silva N, Minke B, Paulsen R, Huber A (2002). Light-regulated subcellular translocation of Drosophila TRPL channels induces long-term adaptation and modifies the light-induced current. Neuron 34(1):83-93
Burns M and Baylor D (2001). Activation, Deactivation, and Adaptation in Vertebrate Photoreceptor Cells. Annu. Rev. Neurosci 24:779-805.
Cowan CW, Fariss RN, Sokal I, Palczewski K, and Wensel, TG (1998). High expression levels in cones of RGS9, the predominant GTPase accelerating protein of rods. Proc Natl Acad Sci 95: 5351-5356.
Fain G, Matthews H, Cornwall M C, and Koutalos Y (2001). Adaptation in Vertebrate Photoreceptors. Physiological Reviews, 81:1, 117-51.
Frings S, Seifert R, Godde M, and Kaupp UB (1995). Profoundly different calcium permeation and blockage determine specific function of distinct cyclic nucleotide-gated channels.
Lee SJ, Montell C (2004). Light-dependent translocation of visual arrestin regulated by the NINAC myosin III. Neuron 43(1):95-103.
Makino CL, Dodd RL, Chen J, Burns ME, Roca A, Simon MI, Baylor DA (2004). Recoverin regulates light-dependent phosphodiesterase activity in retinal rods. J Gen Physiol 123(6):729-41
Martinez-Conde S, Macknick SL, Hubel DH (2004). The role of fixational eye movements in visual perception. Nat Rev Neurosci 229-40.
Mendez A, Burns ME, Sokal I, Dizhoor AM, Baehr W, Palczewski K, Baylor DA, Chen J (2001). Role of guanylate cyclase-activating proteins (GCAPs) in setting the flash sensitivity of rod photoreceptors. Proc Natl Acad Sci 98(17):9948-53.
Miller JL and Korenbrot JI (1994). Difference in calcium homeostasis between retinal rod and cone photoreceptors revealed by the effects of voltage on the cGMP-gated conductance in intact cells. J Gen Physiol 104: 909-940.
Nishiguchi KM, Sandberg MA, Kooijman AC, Martemyanov KA, Pott JWR, Hagstrom SA, Arshavsky VY, Berson EL, and Dryja TP (1998). Defects in RGS9 or its anchor protein R9AP in patients with slow photoreceptor deactivation. Nature 427: 74-78.
Sokolov M, Lyubarsky AL, Strissel KJ, Savchenko AB, Govardovskii VI, Pugh EN, Arshavsky VY (2002). Massive light-driven translocation of transducin between the two major compartments of rod cells: a novel mechanism of light adaptation. Neuron 34(1):95-106.
Recent updates to the site:
List of abbreviations:
- N
- This edit created a new page (also see list of new pages)
- m
- This is a minor edit
- b
- This edit was performed by a bot
- (±123)
- The page size changed by this number of bytes
5 June 2026
4 June 2026
3 June 2026
| 21:28 | CHIP:Talks diffhist +545 Gabor Balazsi talk contribs | ||||
|
|
15:53 | User:Johannes W. Dietrich 3 changes history −220 [Johannes W. Dietrich (3×)] | |||
|
|
15:53 (cur | prev) +44 Johannes W. Dietrich talk contribs (→Research interests) | ||||
|
|
15:50 (cur | prev) +83 Johannes W. Dietrich talk contribs (→Contact Info) | ||||
|
|
15:47 (cur | prev) −347 Johannes W. Dietrich talk contribs (→Contact Info) | ||||
2 June 2026
|
|
21:32 | Hu:Members 2 changes history +439 [Hugangqing (2×)] | |||
|
|
21:32 (cur | prev) +389 Hugangqing talk contribs | ||||
|
|
21:31 (cur | prev) +50 Hugangqing talk contribs | ||||
| m 16:48 | Gerber:Publications diffhist +56 Andre P. Gerber talk contribs | ||||