3D Bioprinting
Introduction
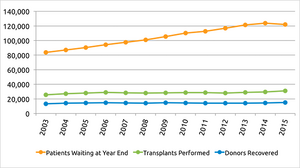
The tissue engineering field has emerged as a solution to the shortages of organ and transplantation needs. As of present, there are approximately 121,142 people in the United States awaiting an organ transplant and only 30,975 transplants were performed in 2015 [1]. The amount of people on the transplant waiting list has grown rapidly since 1991 and has greatly outpaced the amount of transplants being performed. This has provided incentive for researchers to develop methods of creating artificial organs. The traditional approach to create functioning 3D tissue is to seed cells onto biopolymer scaffolds designed to direct cell proliferation and differentiation. However, there are many challenges such as limited biopolymer availability and difficult methods for seeding of various cell types [3]. This approach has often failed to provide an efficient transportation system of growth media including oxygen, nutrients, growth factors, and water, all of which are required for the generation of thick, viable tissues or organs [4].
Recent advances in the field have made these challenges possible by way of additive manufacturing, also known as three-dimensional bioprinting (3D bioprinting). 3D bioprinting is the manufacturing of tissue or organ models by printing hydrogel seeded with live cells. Compared to non-biological printing, 3D bioprinting is more complex due to the choice of materials, cell types, growth and differentiation factors, and technical challenge related to the sensitivity of living cells and the complexities of functional tissues/organs [5]. 3D bioprinting has already been successfully in the creation of several tissues including multilayer skin, bone, vascular grafts, tracheal splints, heart tissue and cartilaginous structures [5]. Today, 3D printing is quickly becoming one of the more favored tools in the tissue engineer's arsenal. Open-source 3D printers are becoming much more commonplace, allowing researchers to make their own modifications to the original design for their research purposes [6].
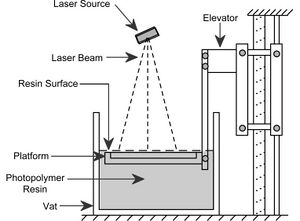
Traditional 3D printing, called stereolithography, was invented in 1984 by Charles W. Hull. His patented technique utilizes a UV activated liquid polymer that hardens when exposed to ultraviolet light. In his method, thin layers of polymer are sequentially hardened, one on top of the other, to form a solid 3D structure with instructions from computer aided design (CAD) programs. The platform upon which the object is built is lowered deeper into the liquid polymer after each layer is solidified, allowing for new layers to be constructed on top of old ones (as shown in the diagram), resulting in a 3D object. More recent 3D bioprinting techniques have borrowed this method to allowed direct printing of biomaterials into 3D scaffolds that may be used for transplantation or tissue modeling.
A Brief History
- 1976 - The Inkjet Printer is invented.
- 1984 - The 3D printer is invented and patented by physicist Charles Hull (US 4575330 A). Technically termed stereolithography, Hull’s invention allows a physical object to be printed from a 3D computer model.
- 1990s - Inkjet printers achieve a resolution of 80 µm, opening the door for bioprinter conversion [7].
- 1999 -An artificially created bladder is successfully transplanted into a beagle, regular urinary function is observed within a month [7].
- 2002 - A small, working kidney was 3D printed in the lab [7].
- 2008 - A 3D printed prosthetic leg was implanted in a human. All parts were printed together, with no assembly required.
- 2012 - Miller et al. successfully 3D print permeable blood vessels, using an open-source 3D printer and biodegradable glass channels [6].
- 2014 - Kolesky et al. successfully create an interwoven vascular network using novel Pluronic F127 fugitive ink [8].
How Do 3D Bioprinters Work?
Most bioprinters have some common, basic parts including the print head mount, elevator, platform, reservoirs, print heads or syringes, and bioink. The print heads are attached to a metal plate that is propelled by a motor along the horizontal x-axis. The elevator is driven by the z-axis motor to move the print heads vertically in order to create successive layers of biomaterial. The platform is a flat surface for the organ to rest on during the production phase. A third motor will move the platform forward and backward, along the y-axis. The reservoirs are attached to the print heads and contain the biomaterial to be used during the printing process. The print heads or syringes release the bioink through various methods. Bioink is biomaterial consisting of live cell suspension and supporting microgel, usually hydrogel that are loaded inside the reservoir of the bioprinter [9]. Currently, there are three different classes of bioprinters that are used for deposition and patterning of biological materials including inkjet, microextrusion, and laser-assisted printing. Each of these bioprinters have unique methods of depositing 3D cell structures with good resolution and viability [5].
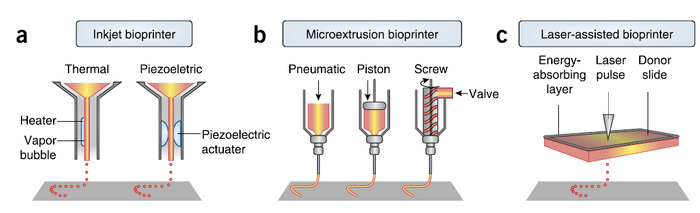
Inkjet Bioprinter
Inkjet bioprinters, also known as drop-on-demand printers, are the most commonly used type of bioprinter due to its low cost, high resolution, high speed, and compatibility with many biological materials. Inkjet printers use thermal or acoustic forces to eject drops of liquid containing cells onto a scaffold, which can support the final construct. Thermal inkjet printers use electricity to heat the print head producing a pulse of pressure that forces droplets from the nozzle. Based on several studies, the thermal temperature, which ranges from 200 C to 300 C, does not affect the stability of the biomaterial or the viability of cells [5]. The advantages of thermal inkjet printing are high print speed, low cost, and wide availability. However, there are several disadvantages to this technique as well including low droplet directionality, non-uniform droplet size, risk of exposing cells to mechanical stress, and frequent clogging of the nozzle [5].
Acoustic printers use piezoelectric crystals to create an acoustic wave inside the print head, breaking the liquid into droplets at regular intervals. Acoustic inkjet printers have the ability to create and control a uniform droplet size and high droplet directionality by adjusting the frequency and amplitude of the acoustic waves. This method generates a small enough amount of energy such that cells are not damaged and has been used with many types of cells [10]. A common drawback of inkjet bioprinting is that biomaterial must be in liquid form to enable droplet formation as well as to prevent clogging of the nozzle. Inkjet bioprinters are limited to low cell densities to avoid clogging and reduce shear stress [5]. Examples of successful applications of an inkjet bioprinter include the regeneration of functional skin and cartilage in situ [3]. High viability and function of cells after printing, as well as low cost, make inkjet bioprinters an invaluable tissue engineering tool.
Microextrusion Bioprinter

Microextrusion bioprinters usually consist of a temperature-controlled biomaterial dispensing system, a stage capable of moving in the x, y, and z directions, a fiber-optic light illuminated deposition area for photoinitiator activation, a video camera for x-y-z command and control, and a piezoelectric humidifier [5]. Unlike inkjet bioprinters, the microextrusion bioprinter generates a continuous string of bioink rather than many droplets of bioink by applying pressure, either pneumatically or mechanically, in order to force bioink from a syringe. These strings are deposited in 2-dimensional layers, as directed by the CAD-CAM software, and served as the base for the subsequent layers while the stage is moves up the z-axis, resulting in the formation of a 3D structure [3]. Microextrusion bioprinters have a wider selection of bioink including high viscosity materials such as hydrogels, bio-compatible copolymers, and cell spheroids. Yet, cell survival rate is lower in comparison with inkjet bioprinters due to substantial pressure. Microextrusion bioprinters have been used to fabricate an aortic valve, branched vessels, and tumor models [5].
Laser-Assisted Bioprinter
The laser-assisted bioprinter (LAB) is less common than inkjet or microextrusion bioprinters, but its applications engineering are increasing. A LAB usually consists of a pulsating laser, a focusing system, a ribbon support commonly made from glass covered with an energy absorbing layer (gold or titanium) and a layer of biomaterial containing cells and hydrogel [5]. The laser pulse will focus on the absorbing layer creating high pressure bubbles, propelling cells toward the substrate plate below. Droplet size was found to be directly correlated with laser pulse energy, providing precise size control [12]. Because LAB is a nozzle-free device, clogging of the nozzle is avoided. LAB is compatible with a wide range of biomaterials with various viscosities. In addition, LAB can produce high resolution 3D structures with high cell viability [5]. Despite these advantages, the high cost of a LAB device prevents its use for basic research.
3D Bioprinting Strategies
3D Bioprinting Approaches
3D bioprinting is based upon three central approaches: biomimicry, autonomous self-assembly, and mini-tissue building blocks.
- Biomimicry involves the replication of both intracellular and extracellular components of a tissue or organ. In order to successfully mimic a specific functional component of a tissue, an understanding of microenvironment is needed. This includes the specific arrangement of functional and support cell types, gradients of soluble and insoluble factors, and composition of the ECM [5]. Biomimicry attempts to model a fully developed tissue and its environment.
- Autonomous self-assemblyuses the development of embryonic organs as a guide. During the early stage of embryo growth, cellular components produce their own ECM components that give the appropriate signal for the formation of functional biological micro-architecture. Autonomous self-assembly relies on cells to direct the composition, localization, functional, and structural properties of the tissue. A scaffold-free tissue fabrication method takes advantage of self-assembling cellular spheroids to mimic developing tissue [5]. This approach would require an in-depth understanding of the environment that drives the development of embryonic tissue genesis and organogenesis.
- Mini-tissues are the functional building blocks of tissue and organs. Mini-tissues are fabricated separately and assembled into a larger tissue through rational design, self-assembly, or a combination of both [5].
Imaging and Digital Design
In order to reproduce complex, heterogeneous functional tissues and organs found in the human body, understanding of composition and organization of their components is an essential requirement. Medical imaging tools are used to provide information on the 3-Dimensional structure and function of these systems at the cellular level [5]. The most commonly utilized methods are computed tomography (CT) and magnetic resonance imaging (MRI). Computer-aided design and computer-aided manufacturing (CAD-CAM) tools are also used to generate complex visual designs for tissue. This is just the first step in the path to creating a 3D tissue or organ model.
Material Consideration
The selection of bioink material is an important consideration when planning to bioprint a tissue model; materials must be suitable for crosslinking in order to facilitate bioprinter deposition and shape retention, as well as biocompatible for long-term transplantation goals. The table below outlines each of the key considerations in preparing an ideal bioink. There are countless possible biomaterials suitable for bioprinting including poly(ethylene glycol) (PEG), polylactic acid (PLA), polyglycolic acid (PGA), poly(lactic-co-glycolic) acid (PLGA) and gellan gum. The choice of material is dependent upon desired cell type and mechanical properties of the tissue being mimicked.

Cell Types
The choice of cell type is crucial for a functional fabricated tissue or organ. Most tissue consists of multiple cell types, each with a different function that must be replicated in the transplanted tissue. Most organs and tissues contain cell types that are necessary for supportive, structural, barrier functions, vasculature networking, and provide cell proliferation as well as differentiation. Cells chosen for bioprinting must mimic the physiological state of native cells to maintain their functional and survival roles. As with many transplants, rejection of bioprinted parts by the host immune system is a possible problem. Therefore, using analogous sources of cells may help to prevent immune rejection. Stem cells are a promising choice for fabrication of tissue due to their ability to proliferate as well as self-differentiate. They also have the ability to generate multiple tissue-specific cell phenotypes. Embryonic stem cells and induced pluripotent stem cells are the most promising cells for bioprinting applications due to their vast potential to differentiate to many different types of cells. Because cells are continuously under mechanical stress from the bioprinter, cells used in bioprinting must be robust enough to overcome those shear stress as well as nonphysiological pH of the printing environment [5].
Vascularization
Vascularization is a vitally important process in ensuring the viability and survival of any tissue. The process occurs naturally in vivo through the process of angiogenesis in which endothelial cells respond to intercellular signals by extending toward the signal. Cells surrounding these “stalk” cells proliferate, sprouting a new vessel. More vessels branch off of this one through the same process, eventually forming a vastly complex capillary network. It has been observed in vivo that almost all cells lay no more than 200 µm from a blood vessel in order to maintain proper nutrient and oxygen diffusion [13]. Advancements have been achieved in developing microvascular networks in vitro by introducing angiogenic growth factors to tissue models in the presence of endothelial cells. However, the process occurs on the order of days and is thus not a viable option for the survival of a bioprinted macrostructure pre-seeded with live cells [13].
Thus, obtaining proper vasculature in bioprinted tissue structures remains one of the greatest challenges facing tissue engineers today. The diameter of human blood vessels ranges from 2.5 cm for the aorta to just 20 µm for very fine capillaries [14]. The finest resolutions achievable with the current, highly expensive, laser-assisted bioprinting technology are on the order of “10s of µm.” Inkjet and microextrusion methods, more commonly used in research applications, print on the order of “100s of µm”, making the direct application of extensive vascular networks currently unattainable [15]. As a result, the latest research has focused mainly around the use of sacrificial gels and structures in order to create an artificial network of microchannels within the bioprinted model.
Perfusable Matrices

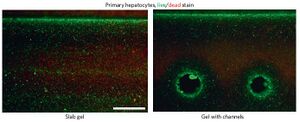
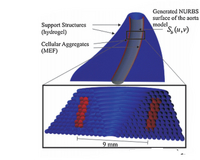
One of the most promising ways to create vasculature in 3D printed organs is through a perfusable channel system within cell-laden hydrogel matrices. Early work used molds for preparing sacrificial structures. This allows the fabrication of microfluidic networks, which then allow the transport of macromolecules into surrounding hydrogel under low pressure difference. As proposed by Miller et. al., a biocompatible carbohydrate-based glass was used as the fugitive ink for the sacrificial molding [6]. Fugitive ink is a special type of ink that is sucked out of the bioprinted structure after it is complete, creating an empty channel. The carbohydrate based glass is a stiff material, can quickly dissolve when exposed to agarose gel, and is biocompatible. To create vasculature with this system, the pre-blood-vessel network (shown right) is lowered into a solution of cells suspended in pre-ECM polymer which has yet to be crosslinked. The network was coated with a layer of polymer, poly(D-lactide-co-glycolide) (PDLGA) to force the dissolved carbohydrates to flow through the network after dissolution, rather than through the ECM, which could damage the cells. Their strategy was able to work with a variety of different ECM materials, which is groundbreaking. The team found that their technique was able to correctly seed endothelial and other cells required for creating vasculature (such as 10T1/2 cells), and even showed signs of new, smaller vessels extending from larger ones. These vessels were able to have nutrients permeate through them, and as a result cells close to the vessels had higher viability than those far away from them, or in a control without blood vessels. Miller believes this method will provide a highly effective way of vascularizing many different kinds of engineered tissues [6].
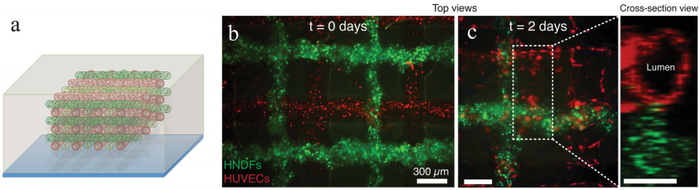
Printing of Vascular Networks With Pluronic F127 Fugitive Ink
Researchers at Wyss Institute for Biologically Inspired Engineering at Harvard University and the Harvard School of Engineering and Applied Sciences (SEAS), created a 3D bioprinting method that able to print different cells and materials at once, while fabricating embedded vasculature. The team was able to print an organ foundation from bioink consisting of gelation methacrylate seeded with fibroblasts and a fugitive ink [8]. The fugitive ink used in this study is a novel aqueous solution containing Pluronic F127, a unique copolymer that becomes more soluble in water as it is cooled and displays similar viscoelastic properties to typical bioink under normal bioprinting conditions. This copolymer is comprised of two hydrophilic polymers sandwiching a hydrophobic polymer. As temperature is decreased, copolymers coagulate to form hydrophilic micelles with dense hydrophobic cores; at 4 C, the gel experiences a dramatic drop in viscoelastic force and is easily removed from the system [8]. In their method, four separate ink reservoirs were used along with four pneumatic extruding print heads. Different nozzle types were used, including borosilicate and tapered plastic. To produce the full construct, each layer was printed separately, after which the fugitive ink was removed and the vascular network was seeded with endothelial cells. The vascularized structure was analyzed over the course of one week in order to investigate both the effectiveness of vascularization and the viability of the cell culture. After 2 days endothelial cell growth in the vascular networks become apparent, as can be seen in the figure below. Cell viability was reported to increase from 61% on the day of fabrication to 82% on day 7, providing evidence for cell proliferation and survival implying adequate nutrient and waste diffusion [8].
Coaxial Printing of Hollow Microchannels
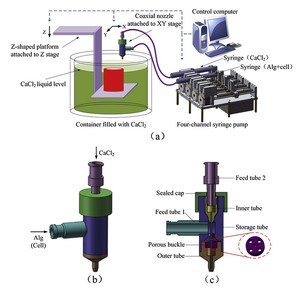
Gao et al. have proposed a novel bioprinting technique in which cell culture is printed simultaneously with hollow sodium alginate channels. The bioprinter utilizes a dual-feed syringe pump with a coaxial nozzle consisting of an inner and outer tube. Alginate and cell solution are feed to the outer tube, surrounding a feed of calcium chloride in the inner tube. The system prints onto a platform suspended in a calcium chloride bath that descends deeper into the bath with each successive layer. The sodium alginate undergoes a crosslinking reaction upon exposure to calcium chloride, resulting in a hardened, hollow microchannel. A schematic of the apparatus is shown in the image to the right [16].
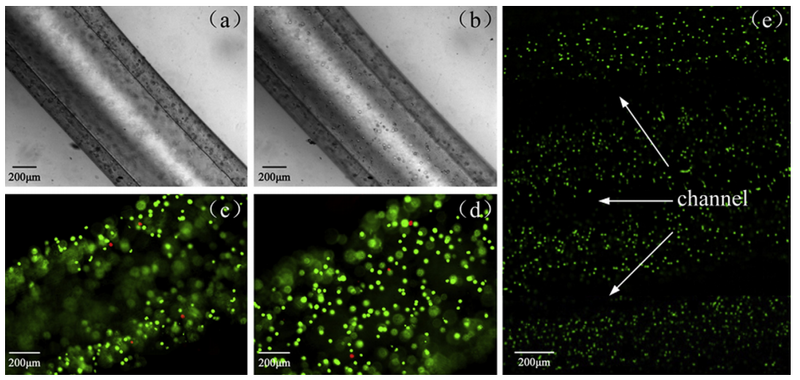
Alginate microfilaments were shown to print uniformly and continuously and diffusion tests performed with macromolecules demonstrated that the structure has viable nutrient permeability properties. A cuboid structure was later created with overlapping microchannels. Scanning electron microscopy (SEM) confirmed that the overlapping microchannels fused and become an interconnected vascular network. Viability of cells within this structure was analyzed one day after fabrication under a confocal microscope. As can be seen in the figure below, cell viability was very high in the areas surrounding the microchannels furthering endorsing the potential of this novel procedure. Future studies suggested for this method include seeding the cell culture with stem cells and consideration of various physiological stresses [16].
Modeling the Human Body
The Skull
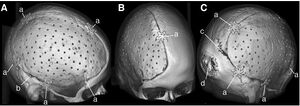
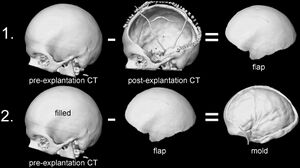
Skull reconstruction is a common procedure which can become necessary after severe head trauma or bacterial infection. Other times tumors can invade the skull, or complications can arise post cranial surgery. Traditional methods involve molding a polymer to the skull by hand, usually polymethylmethacrylate (PMMA), which often results in a poor, asymmetrical appearance, or a bad fit. To better serve patients, Stieglitz et al have devised a method of creating artificial skulls using a 3D printer, where the blueprint is reverse-engineered from patient CT scans [17].
Methods
PMMA implants, based on computer-designed molds, were implanted into twenty-eight patients--11 female, 17 male--between 2009 and 2012. The molding of the implant occurred during surgery, so that a CT scan of the patient's open skull could be used to engineer the necessary shape (see figure right). Results of the procedure were assessed using a questionnaire and quantitative geometric analysis of the implant [17].
Results
Most patients ranked the appearance of the implant to be "good", while two of the twenty-eight found their appearance to be "unfavorable." Overall, most patients said the function of the implant was "good." The quantitative analysis found that the implants had a median quality of 80 [17].
The Aorta
Cardiovascular disease is a leading cause of death worldwide. Blood vessel transplants have been shown to be an effective method of battling this ailment, however, transplants are rare due to the lack of available donors in the patients’ afflicted area. Tissue engineering and bioprinting have provided a potential solution to this issue. Kucukgul et al. sought to bioprint a model human abdominal aorta using a microextrusion printer to deposit PEG hydrogel seeded with porcine aortic valve interstitial cells [18].
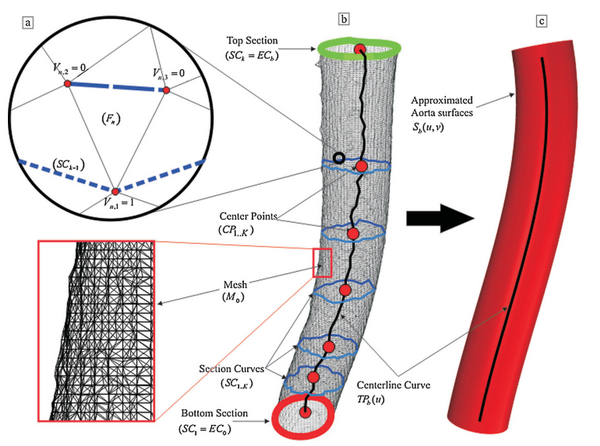
A 3D model of the human aorta was created using medical image segmentation for engineering on anatomy (Mimics) software and converted to a stereolithography file (STL) for compatibility with the 3D printing software. The mesh model created in the STL file is not smooth, but represented by a series of triangular components as seen in the figure to the right. In order to combat the loss of resolution from conversion to STL mesh format, the researchers developed a mathematical model designed to make a higher resolution model of the human aorta based upon the smoothing of the centerline curve within the aortic channel. This math model is also intended to build a structure that is self-supporting, meaning the structure will maintain its shape without the implementation of a scaffold [18].
The experiment was successful creating a self-supporting structure. The mathematical model maximized the contact area between cells on adjacent layers, facilitating the fusion of cells [18]. The success of this novel implementation of a mathematical algorithm designed to maximize printing efficacy and cellular interaction provides incentive for similar methods to be adopted in the future.
Future Direction
There are several obstacles which must be overcome in order for 3D printing to reach its potential for tissue engineering. These include more improved vascularization methods, improving the capabilities of CAD 3D modeling software, increasing resolution, and faster methods of printing. Improvement of CAD software will allow for the increased capability for the customization of 3D printed implants or prosthetics. Increasing the resolution of printed bioink layers will allow for greater precision in creating bioprinted structures, especially vascular networks which contain capillary channels with diameters as low as 20 µm. Faster printing methods will become necessary to maintain the viability of cell cultures throughout the printing process as improved methods will eventually lead the creation of larger, more complex constructs. With current printing technologies, it is estimated that the printing of an object the size of an adult human liver will take over 3 days; it is vitally important that the cells be able to survive this period [19]. In order to address this issue, Kolensky et al. implemented their novel 4-print head microextrusion printer and were able to efficiently decrease the amount of time it took to construct their model. Kolensky posits that increasing the amount of print heads is an effective method of increasing print speed and suggests that this is the direction the field should head in [19]. Another point of concern with the increasing size of bioprinted models is the requirement for more advanced imaging techniques. It is important to be able to monitor parameters such as cell density, viability, and oxygen and nutrient diffusion throughout the structure. Current, simple microscopy techniques will become nonviable when attempting to analyze macrostructures with considerable depth. MRI and ultrasound may become viable solutions in the future of imaging bioprinted tissue [13].
Video Examples
The process of 3D printing using non-biocompatible such as plastics.
<html>
<iframe width="560" height="315" src="https://www.youtube.com/embed/U-8-lu4mq8o" frameborder="0" allowfullscreen></iframe>
</html>
Printing an artificial trachea from a 3D CAD model.
<html>
<iframe width="560" height="315" src="https://www.youtube.com/embed/Ibu7ennpujM" frameborder="0" allowfullscreen></iframe>
</html>
3D bioprinting process and vasculature by Wyss Institute.
<html>
<iframe width="560" height="315" src="https://www.youtube.com/embed/p1qyg4TbDgQ" frameborder="0" allowfullscreen></iframe>
</html>
See also
Reference
-
"Organ Procurement and Transplantation Network." U.S. Department of Health & Human Services. U.S. Department of Health & Human Services., n.d. Web.
-
Zhang, X, and Y Zhang. "Tissue Engineering Applications of Three-Dimensional Bioprinting." Cell Biochemistry and Biophysics. (2015). Print.
-
"New Findings on Tissue Engineering Described by Investigators at University of Iowa (a Hybrid Bioprinting Approach for Scale-Up Tissue Fabrication).(report)." Blood Weekly. (2014): 78. Print.
-
Murphy, SV, and A Atala. "3d Bioprinting of Tissues and Organs." Nature Biotechnology. 32.8 (2014): 773-85. Print.
-
Miller, J. S., Stevens, K. R., Yang, M. T., Baker, B. M., Nguyen, D.-H. T., Cohen, D. M., … Chen, C. S. (2012). Rapid casting of patterned vascular networks for perfusable engineered three-dimensional tissues. Nature Materials, 11(9), 768–774.
-
Lang-8. Bioprinting: Brief Timeline and Outlook. http://lang-8.com/1017887/journals/107384577998436484655804992513119667379.
-
Kolesky, D. B., Truby, R. L., Gladman, A. S., Busbee, T. A., Homan, K. A., & Lewis, J. A. (2014). 3D Bioprinting of Vascularized, Heterogeneous Cell-Laden Tissue Constructs. Advanced Materials.
-
Harris, William. "How 3-D Bioprinting Works." HowStuffWorks. HowStuffWorks, n.d. Web.
-
Gu, Q.; Hao, J.; Lu, Y.; Wang, L.; Wallace, G. G.; Zhou, Q. Three-Dimensional Bio-Printing. Science China: Life Sciences. 2015. 58. 411-419.
-
http://geeksandknots.blogspot.com/2013/04/print-what-servin-up-some-frankenstein.html
-
Fermeiro, J.B.L.; Calado, M.R.A.; Correia, I.J.S. State of the art and challenges in bioprinting technologies, contribution of the 3D bioprinting in Tissue Engineering. Bioengineering. 2015. 1-6.
-
Paulsen, S.J.; Miller, J.S. Tissue Vascularization Through 3D Printing: Will Technology Bring Us Flow?. Developmental Dynamics. 2015. 244. 629-640.
-
Hoch, E, G E. M. Tovar, and K Borchers. "Bioprinting of Artificial Blood Vessels: Current Approaches Towards a Demanding Goal." European Journal of Cardio-Thoracic Surgery. (2014). Print.
-
Bakhshinejad, A.; D’Souza, R.M. A Brief Comparison Between Available Bio-printing Methods. Great Lakes Biomedical Conference. 2015.
-
Gao, Q.; He, Y.; Fu, J.; Ma, L. Coaxial nozzle-assisted 3D bioprinting with built-in microchannels for nutrients delivery. Biomaterials. 2015. 61. 203-215.
-
Stieglitz, L. H., Gerber, N., Schmid, T., Mordasini, P., Fichtner, J., Fung, C., … Beck, J. (2014). Intraoperative fabrication of patient-specific moulded implants for skull reconstruction: single-centre experience of 28 cases. Acta Neurochirurgica, 156(4), 793–803.
-
Kucukgul, C.; Ozler, B.; Inci, I.; Karakas, E.; Irmak, S.; Gozuacik, D.; Taralp, A.; Koc, B. 3D Bioprinting of Biomimetic Aortic Vascular Constructs With Self-Supporting Cells. Biotechnology and Bioengineering. 2014. 112:4. 811-821.
-
"Findings on Tissue Engineering Detailed by Investigators at University of Iowa (bioprinting Technology: a Current State-of-the-Art Review)." Blood Weekly. (2014): 54. Print.
-
Hull, C. W. (1986, March 11). Apparatus for production of three-dimensional objects by stereolithography. Retrieved from http://www.google.com/patents/US4929402
-
Ozbolat, IT, and Y Yu. "Bioprinting Toward Organ Fabrication: Challenges and Future Trends." Ieee Transactions on Bio-Medical Engineering. 60.3 (2013): 691-9. Print.
