Todd:The Lewis Acid-Catalysed Pictet-Spengler Formation of Substituted 1,2,3,4-Tetrahydroisoquinolines
This is an undergraduate project running from March-June 2012. Current plan of action here. Final document due: 17:00 +10 GMT (AEST), 15 June 2012.
Abstract
The Pictet-Spengler (PS) reaction is a useful carbon-carbon bond forming reaction. It is efficiently used in the synthesis of tetrahydroisoquinoline- (THIQ), tetrahydro-β-carboline- and more recently, quinoxaline-derived moieties, which are present in a number of biologically active molecules.[3,6-8,10-11,14] While there are a number of examples for the Brønsted acid-catalysed asymmetric PS formation the tetrahydro-β-carboline scaffold, there are no known examples of the asymmetric Lewis acid-catalysed PS reaction.[12,19] Furthermore, there are no reported examples of the catalytic asymmetric PS formation of THIQ frameworks.[17] This project involved synthesis of imine precursors to the C1-substituted-THIQ scaffold followed by evaluation of Brønsted and Lewis acids to catalytically produce C1-substituted-THIQ scaffolds from the imine precursors. The acids screened (trifluoroacetic acid, Zn(OTf)2, Cu(OTf)2, AgOTf and Yb(OTf)3) were ineffective at catalysing the PS formation of the substituted THIQs, which prompted assessment of the acyl-PS reaction. The protocol described in the previously reported AuCl3/AgOTf acyl-PS reaction[13] was adapted for use with the Yb(OTf)3. After minor optimisation and with the aid of a 1H-NMR spectroscopic assay, a highly efficient acyl-PS reaction to give 1-(6,7-dimethoxy-1-(4-nitrophenyl)-3,4-dihydroisoquinolin-2(1H)-yl)ethanone was developed.
Introduction and Background
The Pictet-Spengler Reaction in Organic Synthesis and Nature
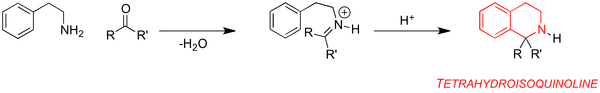
The first example of the synthesis of tetrahydroisoquinoline (THIQ) from β-phenethylamine and formaldehyde was reported by Amé Pictet and Theodor Spengler in 1911.[1] The reaction now known as the Pictet-Spengler (PS) reaction typically occurs via the condensation of a β-arylamine and an aldehyde or ketone to give an electrophilic iminium species. This is subsequently attacked by the neighbouring aryl group (Scheme 1).
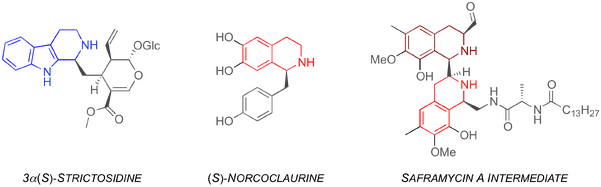
The PS reaction has been found to occur in nature. The recently discovered class of enzymes known as Pictet-Spenglerases are instrumental in the synthesis of 3α(S)-strictosidine and (S)-norcoclaurine, which are key intermediates in the biosynthesis of numerous plant alkaloids (Figure 1).[2,3] Additionally, studies of a biosynthetic pathway to the potent antibiotic saframycin A have revealed that the bacterium nonribosomal peptide synthetase SfmC constructs the scaffold via two PS reactions. Figure 1 shows the intermediate product after the two PS reactions to form the THIQ scaffolds (red).[4-5]
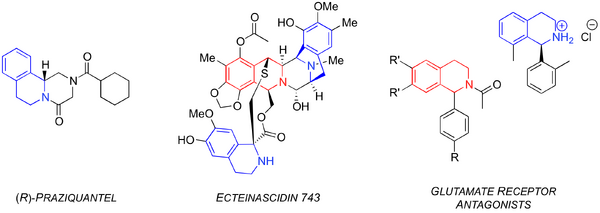
The PS reaction is a powerful tool in the synthesis of complex, biologically active polycyclic heterocycles.[3, 5-8] Three examples are ecteinascidin 743,[5,6] praziquantel[9] and the glutamate receptor antagonists (Figure 2).[7,8] The first example, Ecteinascidin 743, is an anti-tumor drug currently in phase II clinical trials.[10] The anthelmintic praziquantel is used to treat the neglected tropical disease schistosomaiasis.[11] Lastly, the aryl-THIQ have reported glutamate receptor antagonistic activity, which make them ideal for evaluation as anticonvulsants.[7,8]
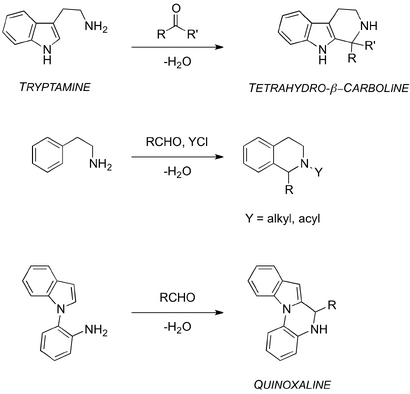
Variations of the PS reaction include the formation of the tetrahydro-β-carboline scaffold, the acyl-PS reaction and the modified Pictet-Spengler to give fused quinoxalines (Scheme 2).[3, 12-14] These variations have contributed to the ever increasing scope of the PS reaction.
The Limits of the Pictet-Spengler Reaction in Organic Synthesis
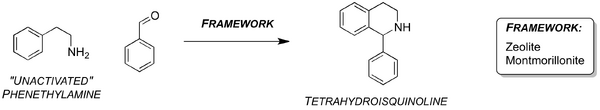
A limitation of the PS reaction is the catalytic formation of non-activated THIQs. Ironically, the first PS formation of the THIQ scaffold appears to be the most difficult to produce catalytically via the PS route, at least by traditional homogenous catalytic methods.[13,15-16] Two examples are known where the PS cyclisation of the unactivated β-phenethylamine (i.e. phenethylamine bearing no electron donating groups) with aldehydes is catalysed by heterogenous zeolite and acidic montmorillonite frameworks (Scheme 3).[15-16] Furthermore, there is yet to be found a biosynthetic example of the PS formation of the unactivated THIQ scaffold.
Recent reviews of the literature have revealed a lack of Lewis acid-catalysed asymmetric PS reactions.[3,17] Furthermore, there are no known examples of the asymmetric PS formation of the THIQ scaffold. Instead, the focus is on the organocatalysed asymmetric PS formation of tetrahydro-β-carbolines using catalysts such as the chiral thioureas[12,18] and binapthyl-derived phosphoric acids.[19]
Still, there is precedent for the development of a catalytic asymmetric PS reaction to yield the THIQ scaffold. Rather than using chiral organocatalysts, as is the case with the synthesis of the tetrahydro-β-carboline moiety, the asymmetric formation to give the THIQ scaffold potentially lies with Lewis acid-catalysis.[20,21] The low reactivity of the imine intermediates in the PS formation of THIQ scaffolds may be counteracted by highly Lewis acidic metal catalysts. Furthermore, there are increasing examples of the use of these catalysts in combination with chiral ligands to produce asymmetric outcomes via an iminium intermediate.[20, 22-25]
Project Summary
A method for the asymmetric Lewis acid-catalysed PS reaction to give the THIQ scaffold first requires a racemic "template". In comparison to the number of reported examples of the Lewis acid-catalysed formation of the tetrahydro-β-carboline scaffold, the scope of the synthesis of THIQs using Lewis acid catalysts is limited.[17] Three examples of the Lewis acid-catalysed PS reaction to give the THIQ scaffold use ytterbium(III) triflate,[26] calcium hexafluoroisopropoxide[27] and a gold(III) chloride-silver triflate combination.[13]
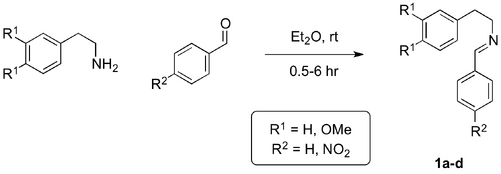
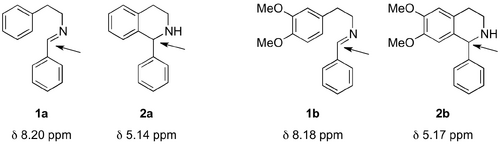
The focus of this project was the development of a method for the achiral Lewis acid-catalysed PS formation of the THIQ scaffold that would be adaptable to an asymmetric model (Scheme 4). In order to minimise complications arising from one-pot procedures using the aldehyde and arylamine starting materials, the corresponding isolatable imines were prepared for use as model substrates (Scheme 4, 1a-d).
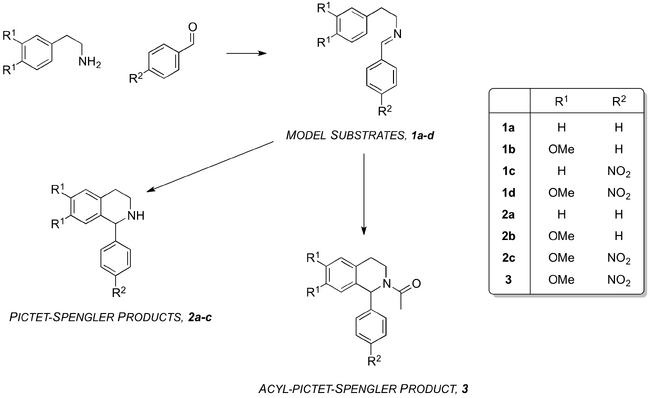
Brønsted acids only effected cyclisation of the imine substrates at high temperatures or when present in superstoichiometric amounts. This was consistent with the known literature that, in the PS-syntheses of the activated THIQ scaffold, describes the use of strong Brønsted acids in vast excess.[28]
Lewis acids revealed a similar ineffectiveness at inducing the PS formation of the THIQs 2b and 2c from the corresponding imines.
Adaptation of the model to the acyl-PS reaction produced more promising results. The ytterbium(III) triflate Lewis acid catalyst was found to be highly efficient at inducing the acyl-PS cyclisation of the activated THIQ scaffold. The reaction optimisation process was aided by a 1H nuclear magnetic resonance (NMR) spectroscopic assay that involved development of a post-reaction work-up procedure.
Results and Discussion
Synthesis of the Model Substrate Materials and the Brønsted Acid-Mediated Cyclisations
The first phase of the project required synthesis of the imine model substrates, which were easily prepared in excellent yield (Scheme 5, Table 1). The condensation reactions of the arylamines with the corresponding aldehydes was facile. The nitro-substituted imines (1c and 1d) did not require post-reaction addition of a dehydrating agent but readily crystallised from the crude reaction mixtures.[29]

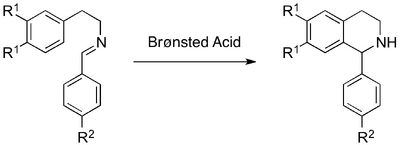
The literature procedures for the PS formation of activated-THIQs describe harsh conditions: excess strong Brønsted acids and high temperatures.[28] In contrast, there are a number of examples describing the Brønsted acid-catalysed PS formation of the tetrahydro-β-carboline scaffold from tryptamine and various aryl- and alkyl-aldehydes.[3,19] It was therefore appropriate to evaluate the reactivity of the model imine substrates under excess and catalytic Brønsted acid reaction conditions (Scheme 6).
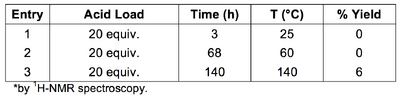
Attempts to cyclise imine 1a using neat methanesulfonic acid and elevated temperatures were largely unsuccessful (Table 2, Entries 1-3). Of the activated imine, 1b, 1H-NMR spectroscopic analysis indicated good conversion when a large excess of trifluoroacetic acid was used (Table 2, Entry 5). Lowering the acid loading and increasing the reaction temperature resulted in moderate conversion. Under catalytic conditions, no conversion of 1b was observed after 5 days (Table 2, Entry 7).
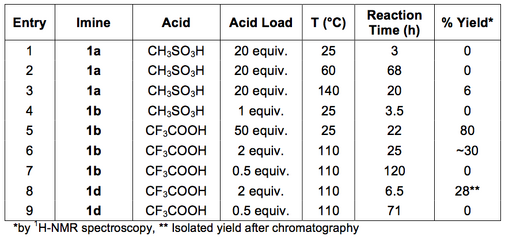
The conditions for monitoring the reaction by thin layer chromatography (TLC) required a highly polar eluent (1:5, methanol/dichloromethane, v/v). Use of a ninhydrin stain facilitated visualisation of the characteristic secondary amine, indicative of the THIQ formation. Conversion for the attempts to give 2a and 2b were determined by 1H-NMR spectroscopy of the crude products of known mass. The conversion and overall yield was calculated by the total mass of the isolated crude product and the relative peak integrals of the imine protons and the C1-THIQ protons (Figure 3).[30]
Despite the considerable conversion, by 1H-NMR spectroscopy, of imine 1b to the corresponding THIQ (Table 2, Entry 5), no product was isolated. The high polarity of the substrates and products made observation and purification of the reaction mixtures and products by chromatographic techniques difficult. The imine substrate materials did not behave as expected by TLC, despite appearing clean by 1H-NMR spectroscopy. Stambulli and co-workers made a comment on the high polarities and difficulties in handling the THIQ products.[27] They suggested, based on infrared spectroscopy, the substrate and products existed as zwitterions. The difficulties in purification of the THIQ by chromatography or recrystallisation were consistent with these literature comments.
The conversion of imine 1d to the THIQ 2c was only qualitatively assessable by TLC and 1H-NMR spectroscopy (Table 2, Entries 8-9). The 1H-NMR spectrum of the crude reaction mixture exhibited the characteristic C1-proton of the cyclised product, however comparison of the peak with the imine proton was complex (Table 2, Entry 8). The imine was situated in a region of high coincidence (~7.5-9.0 ppm) so integrations were unreliable. The product 2c was partially purified under polar chromatographic conditions in moderate yield (Table 2, Entry 8). Under a catalytic acid load, no conversion of imine 1d was observed after 3 days (Table 2, Entry 9).
The results of the Brønsted acid-catalysed reactions were not entirely unexpected given the low reactivity of the imines and lack of examples and in the literature. The inability to efficiently monitor conversion and yield for the reactions in the presence of Brønsted acids encouraged movement onto the next stage of the project.
The Lewis Acid-Catalyst Screen
The next phase of the project involved the evaluation of a number of Lewis acids with potential to effect the formation of the THIQ scaffold. Given the literature procedures for the formation of the THIQ scaffold required excess amounts of strong Brønsted acids,[28] it was expected that strong Lewis acids would be required for the PS reaction under catalytic conditions.
The lanthanoid (Ln) triflates are potent Lewis acids.[20] The triflate counterion is highly effective at augmenting the Lewis acidity of the Ln(III) metals.[31] Ytterbium(III) is particularly acidic, with its f13 configuration; it is one of the most Lewis acidic metals of the lanthanoid series.[32]
Ytterbium(III) triflate and other metal triflates, such as Cu(OTf)2 and Zn(OTf)2 are also also catalytically active in the PS reaction.[33] However, only Yb(OTf)3 has been shown to effect formation of the THIQ scaffold.[26]
Metal triflates were an attractive choice for the Lewis acid-catalysed PS reaction. They also provided a link to the broader goal of this project: asymmetric catalysis. Metal triflates coordinate to chiral ligands, such as derivatives of bisoxazoline and binapthol (Figure 4).[20,22-24] These chiral complexes have been effectively used in enantioselective cycloaddition reactions and the Friedel-Crafts reaction, which is mechanistically similar to the PS reaction.[20,22-24]
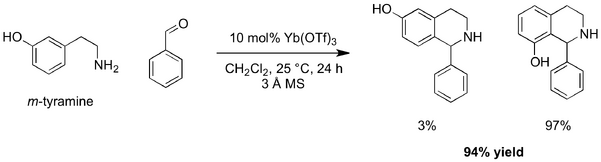
As shown previously, the electron-rich model substrates 1b and 1d were most likely to undergo the cyclisation reaction. The conditions for the Lewis acid screen for the PS reaction of these imines were adapted from Kobayashi and co-workers. They reported the PS reaction to give the THIQ scaffold using m-tyramine as a substrate (Scheme 7).[26]
Under the conditions employed, the four metal triflates were ineffective at forming the Pictet-Spengler products (2b-c) from the corresponding imines (Scheme 8). Starting material was recovered in all reactions.
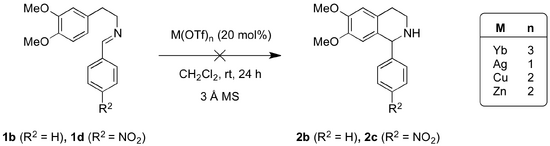
Like many examples of the PS formation of THIQ, the substrate materials in the template protocol were the aryl-amine and the aldehyde. The lack of cyclisation of the imine substrates prompted re-screening of Yb(OTf)3 in reactions from phenethylamine and benzaldehyde. This also resulted in recovery of the corresponding 1b imine (Scheme 9, Table 3).
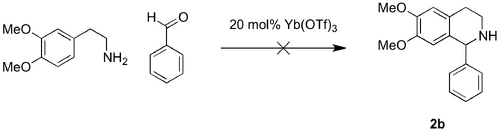
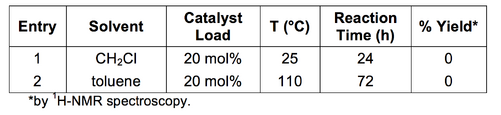
Using different conditions and m-tyramine and benzaldehyde as substrates, Stambuli and co-workers reported similar a yield to Kobayashi and co-workers for the Yb(OTf)3 catalysed formation of the THIQ scaffold.[27] The altered protocol, which involved substitution of the dichloromethane (DCM) solvent for toluene and an increase in the reaction temperature (25 °C to 110 °C) was also attempted with phenethylamine and aldehyde as substrates (Table 3: Entry 2). Again, 1b imine was recovered and zero conversion to the THIQ 2b was observed.
In all attempts, the recovered material from the attempts at the Yb(OTf)3 catalysed PS reactions from phenethylamine and benzaldehyde was identified, by 1H-NMR spectroscopy as the corresponding imine 1b. These results suggested greater substrate specificity for the Lewis acid-catalysed PS formation of the substituted THIQ.
The Lewis Acid-Catalysed Acyl-Pictet-Spengler Reaction
The lack of activity of the imines to catalytic PS conditions prompted adaptation of the model system to the acyl-PS variation. This variation involves formation of a highly electrophilic N-acylminium species. The electron withdrawing effects of the acyl group vastly increase the electrophilicity of the intermediate relative to the non-acylated iminium intermediate.[21] Nonetheless, there are limited known examples of the acyl-PS reaction effected by catalytic Lewis acids.[12-13,21]
One example of the Lewis acid-catalysed acyl-PS reaction to give the THIQ describes use of an AuCl3 and AgOTf catalyst combination.[13] This reaction was an ideal starting point for the Lewis acid catalysed acyl-PS reaction, particularly due to the identical starting materials and expected products to this project.
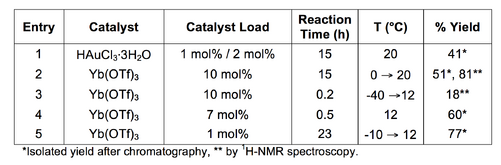
With minimal adaptation, the THIQ was produced with moderate yield (Scheme 10, Table 4: Entry 1). Presumably, the reduced yield in comparison to the reported yield of 78% was partially due to the use of HAuCl4·3H2O instead of AuCl3.[13] Following purification, the product, 3, and 4-nitrobenzaldehyde (4) and N-(3,4-dimethoxyphenethyl)acetamide (5) by-products were isolated.
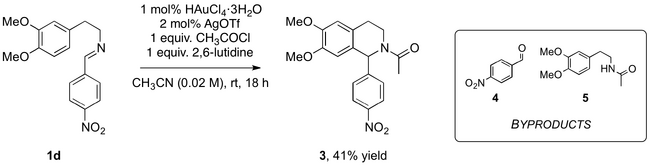
The procedure was performed in the absence of acylating conditions resulting in recovery of the imine starting material. This was consistent with the literature,[13] again suggesting the acyl group was essential for the reaction to proceed.
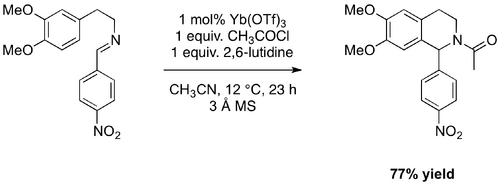
The high Lewis acidity of Yb(OTf)3 and reports of its asymmetric catalytic activity when complexed to chiral ligands encouraged adaptation of the HAuCl4·3H2O/AgOTf protocol. The reaction was carried out using Yb(OTf)3 as a Lewis acid catalyst under anhydrous conditions. After minimal optimisation, the reaction proceeded with excellent results (Scheme 11, Table 4, Entries 2-5).
1H-NMR spectroscopic analyses of the reaction product and by-products revealed sufficient separation of integrable peaks (Figure 5). Unlike monitoring of the conversion of the non-acyl-PS reactions, conversion and yield were gauged by comparison of the THIQ product and the hydrolysis by-products. Ideally, conversion was evaluated by using 1,1,2,2,-tetrachloroethane (CDCl3 δ 5.97 ppm) as an internal standard, and the peak integrals of the aldehyde proton of 4, the acyl- or CH2 environment of 5 (Figure 5, inset 2) and either the stereogenic proton or the aromatic proton of 3 (Figure 5, inset 1).
The first attempts to gauge the yield by 1H-NMR spectroscopy of the crude product (in CDCl3) were inconsistent with the isolated product yield. Spectroscopic analysis suggested a substrate to product conversion of 81%, while the isolated yield after chromatography was 51%. There were two problems associated with the spectroscopic assay: the presence of a CDCl3 insoluble white solid and peak interference by 2,6-lutidine. The nature of the assay meant that complete dissolution of the crude product into the CDCl3 solvent was crucial (see section 5.12). Incomplete dissolution of the crude product meant that the mass of the crude product assayed and the mass of the internal standard were non-comparable by 1H-NMR spectroscopy. Furthermore, the 2,6-lutidine signals were partially coincidental to the integrable peaks of interest.
A post-reaction work-up procedure was developed in order to address the problems with the 1H-NMR assay. The 2,6-lutidine peaks were minimised by including a citric acid wash of the crude product. The presence of chloride in the crude reaction mixture exacerbated the formation of a suspected hydrochloride salt in subsequent attempts to dissolve the extracted product in CDCl3. This was successfully resolved by carrying out an alkaline wash following the acid-workup.
Use of the spectroscopic assay enabled a more efficient reaction optimisation process. The reaction outcomes (i.e. product and byproduct yields) were quickly determined without chromatography. As a result, the effects of the altered reaction conditions were taken into consideration for subsequent reactions (Table 4, Entries 3-5).
The protocol for the Yb(OTf)3 catalysed reaction, which was adapted from the HAuCl4·H2O/AgOTf co-catalysed reaction, gave highly promising results (Scheme 11, Table 4, Entry 2). Anhydrous conditions were employed to minimise competing hydrolysis reactions. The first attempts at the Yb(OTf)3 catalysed acyl-PS reaction suggested the rapid formation of the expected product and hydrolysis by-products. The extent of hydrolysis was minimised by reducing the initial reaction temperature and altering the order in which the reaction components were added (see section 5.11).
Conclusions and Future Work
The Yb(OTf)3 catalysed acyl-PS reaction is an efficient route to the dimethoxy substituted THIQ scaffold. The reaction required less preparation and was more atom efficient than the HAuCl4·H2O/AgOTf co-catalysed reaction. The best result, with minimal optimisation, yielded 1-(6,7-dimethoxy-1-(4-nitrophenyl)-3,4-dihydroisoquinolin-2(1H)-yl)ethanone (3) in 77% using 1 mol% Yb(OTf)3.
It is highly likely with further optimisation, the Yb(OTf)3 catalysed acyl-PS reaction could give greater yield of the THIQ product. Due to time constraints satisfactory optimisation of the Yb(OTf)3 catalysed acyl-PS reaction could not be performed. The effects of solvent, reaction temperature, acylating agents and duration effects were not adequately explored. The primary solvents used in catalytic asymmetric PS reactions are toluene and dichloromethane, which are notably different from the polar acetonitrile that was used in the reactions.[17] An alternative approach to optimising the reaction outcome would include carrying out the reaction in deuterated solvent and observing the progress by 1H-NMR spectroscopy.
The spectroscopic assay made assessment of reaction conditions on the results more efficient. Some improvements on the assay could involve construction of standard curves. This should minimise the dependence of the assay on a completely dry sample. The current method is dependent on the mass of crude product assayed, which produced inaccurate values if solvent was present in the 1H-NMR spectrum.
The lack of imine reactivity in both the Yb(OTf)3 catalysed and HAuCl4·H2O/AgOTf co-catalysed PS formation of the THIQ suggested the N-acyliminium intermediate was primarily responsible cyclisation. This is consistent with the increased electrophilicity of the N-acyliminium that is a direct result of the electron withdrawing acyl group.[21] As mentioned above, monitoring of the reaction by 1H-NMR spectroscopy may provide insight into the reaction mechanism.
Other appropriate avenues to explore are evaluation of the achiral Brønsted acids in the acyl-PS reaction and investigating the outcomes when different aryl- and aliphatic- aldehydes, and arylamine substitutes, such as thiophene or phenylethyl alcohol are used. Similarly, the activity of other lewis acidic Ln(III) triflates could be investigated. Comparison of the AuCl3/AgOTf co-catalysed reaction under anhydrous conditions should also be explored.
A screen of the catalytic potential of other metal triflates in the acyl-PS reaction is necessary. While Yb(OTf)3 may be one of the most Lewis acidic catalysts, there are more examples of d-block transition metal triflates participating in enantioselective reactions. Nonetheless, the number of examples of asymmetric Ln(III) triflate catalysed reactions is increasing.
Ideally, next stage after further optimisation, would involve carrying out the acyl-PS reaction in the presence of chiral ligands. Notable examples of asymmetric Ln(III) triflate catalysed reactions that should be evaluated, combine Yb(OTf)3 with chiral bisoxazoline and pyBOX ligands, and the binapthol derived ligands.[20,24-25]
Experimental
1H-NMR and 13C-NMR spectra were recorded at 300 K on a Bruker AVANCE 200 spectrometer (1H-NMR at 200.13 MHz) or a Bruker AVANCE 300 spectrometer (1H-NMR at 300.13 MHz and 13C-NMR at 75.47 MHz). 1H-NMR signals are reported with chemical shift values δ (ppm), multiplicity (s = singlet, d = doublet, t = triplet, q = quartet, and m = multiplet), coupling constants J (Hz) and relative integration. The CDCl3 and DMSO-d6 solvents used in 1H-NMR and 13C-NMR spectroscopic analyses were obtained from the Cambridge Isotope Laboratories. All melting points (m.p.) were recorded using on a Standford Research Systems OptiMelt (capillaries: ø = 1.5-1.6 mm, 90mm; ramp rate 1 °C min-1). Glassware used in anhydrous reactions were dried >2 hours at 130 °C then cooled under inert gas before use. All molecular sieves were microwave activated and cooled under nitrogen before immediate use. Column chromatography procedures were carried out using Merck silica gel (SiO2, 0.040-0.063 mm). Analytical thin layer chromatography was performed on Merck silica gel 60 F254 precoated aluminium plates (0.2 mm) and visualised with UV irradiation (254 nm or 301 nm), with ninhydrin or permangenate staining.
N-benzylidene-2-phenylethanamine (1a)
To a stirring solution of benzaldehyde (4 mL, 40 mmol, 1 equiv.) in diethyl ether (10 mL) was slowly added 2-phenylethanamine (5 mL, 40 mmol, 1 equiv.). The clear yellow solution was stirred at room temperature for 5 hours, dried (MgSO4) and concentrated under reduced pressure to yield a yellow oil that solidified on standing to give a yellow crystalline solid (8.3 g, 99%). M.p. 33-35 °C. 1H-NMR (300 MHz; DMSO-d6): δ 2.93 (t, J = 7.3 Hz, 2H), 3.81 (t, J = 7.3 Hz, 2H), 7.45-7.16 (m, 10H), 7.71 (dd, J = 6.6, 2.9 Hz, 2H), 8.26 (s, 1H). 13C-NMR (75 MHz; CDCl3): δ 37.5, 63.2, 126.1, 128.1, 128.3, 128.6, 129.0, 130.6, 136.2, 139.9, 161.5. Relevant lab book entries: KAB18-1, KAB18-2.
N-benzylidene-2-(3,4-dimethoxyphenyl)ethanamine (1b)
To a stirring solution of benzaldehyde (3.1 mL, 30 mmol, 1 equiv.) in diethyl ether (10 mL) was slowly added 2-(3,4-dimethoxyphenyl)ethanamine (5.0 mL, 30 mmol, 1 equiv.). The mixture was stirred at ambient temperature for 5 hours, diluted with diethyl ether (20 mL), dried (MgSO4) and concentrated under reduced pressure to yield a yellow oil that crystallised on standing (7.3 g, 90%). M.p. 31-33 °C. 1H-NMR (300 MHz; CDCl3): δ 2.99 (t, J = 7.2 Hz, 2H), 3.89-3.84 (m, 8H), 6.83-6.77 (m, 3H), 7.43 (dt, J = 5.3, 2.6 Hz, 3H), 7.73 (dd, J = 6.7, 3.0 Hz, 2H), 8.15 (s, 1H). 13C-NMR (75 MHz; CDCl3): δ 37.0, 55.71, 55.89, 63.3, 76.7, 77.1, 77.6, 111.2, 112.6, 120.9, 128.0, 128.6, 130.6, 132.6, 136.2, 147.4, 148.7, 161.5. Relevant lab book entries: KAB19-1, KAB19-2.
N-(4-nitrobenzylidene)-2-phenylethanamine (1c)
To a stirring suspension of 4-nitrobenzaldehyde (6.0 g, 40 mmol, 1 equiv.) in diethyl ether (40 mL) was slowly added 2-phenylethanamine (5.0 mL, 40 mmol, 1 equiv.). The mixture was stirred at room temperature for 1 hour before a yellowish solid precipitated. The mixture was concentrated under reduced pressure affording the pure product as pale yellow needles (10 g, 99%). M.p. 71-72 °C. 1H-NMR (300 MHz; CDCl3): δ 3.04 (t, J = 7.3 Hz, 2H), 3.93 (t, J = 7.2 Hz, 2H), 7.20-7.18 (m, 1H), 7.31-7.26 (m, 2H), 7.85 (d, J = 8.7 Hz, 2H), 8.20 (s, 1H), 8.25 (d, J = 8.7 Hz, 2H). 1H-NMR (300 MHz; DMSO-d6): δ 8.41 (s, 1H), 8.41 (s, 1H), 8.41 (s, 1H), 8.41 (s, 1H), 8.41 (s, 1H), 8.41 (s, 1H), 8.41 (s, 1H), 8.28 (d, J = 8.7 Hz, 2H), 7.96 (d, J = 8.7 Hz, 2H), 7.31-7.16 (m, 5H), 3.89 (t, J = 7.2 Hz, 2H), 2.96 (t, J = 7.3 Hz, 2H). 13C-NMR (75 MHz; DMSO-d6): δ 37.1, 62.4, 124.4, 126.5, 128.7, 129.26, 129.30, 140.1, 142.1, 149.0, 160.1. Relevant lab book entry: KAB22-1.
2-(3,4-dimethoxyphenyl)-N-(4-nitrobenzylidene)ethanamine (1d)
4-Nitrobenzaldehyde (3.7 g, 24 mmol) was suspended in diethyl ether (50 mL). 2-(3,4-Dimethoxyphenyl)ethanamine (4.0 mL, 25 mmol) was added dropwise, with stirring. The mixture was left to stir at ambient temperature for 6 hours resulting in the precipitation of a fine light yellow solid. The solvent was removed under reduced pressure to give the crude product as a fine, yellow powder (8.0 g, 103%). Recrystallisation of the crude product from ethanol (~200 mL) afforded the pure product as yellow needles (7.1 g, 23 mmol, 92%). M.p. 123-124 °C. 1H-NMR (200 MHz; CDCl3): δ 2.99 (t, J = 7.1 Hz, 2H), 3.81 (s, 3H), 3.85 (s, 3H), 3.91 (td, J = 7.1, 1.1 Hz, 2H), 6.78-6.72 (m, 3H), 7.86 (d, J = 8.8 Hz, 2H), 8.19 (s, 1H), 8.29-8.23 (m, 2H). 1H-NMR (300 MHz; DMSO-d6): δ 2.90 (t, J = 7.2 Hz, 2H), 3.69 (d, J = 2.1 Hz, 6H), 3.86 (t, J = 7.1 Hz, 2H), 6.75 (dd, J = 8.2, 1.4 Hz, 1H), 6.84 (dd, J = 4.8, 3.3 Hz, 2H), 7.98 (d, J = 8.7 Hz, 2H), 8.29 (d, J = 8.7 Hz, 2H), 8.40 (s, 1H). 13C-NMR (75 MHz; DMSO-d6): δ 36.6, 55.80, 55.94, 62.7, 112.2, 113.3, 121.1, 124.4, 129.2, 132.5, 142.1, 147.6, 148.9, 160.0. Relevant lab book entries: KAB23-1, KAB23-2.
Attempts at the synthesis of 1-phenyl-1,2,3,4-tetrahydroisoquinoline (2a)
Typical procedure (e.g. Entry 2): To a stirring solution of methanesulfonic acid (3.0 mL, 46 mmol) at 0 °C was added N-benzylidene-N-phenethylamine (1a) (0.48 g, 2.3 mmol). The now yellow solution was heated to 60 °C. After 30 minutes the solution had turned dark red. Stirring was continued at 60 °C for 68 hours. The mixture was poured over an ice water slurry (~15 mL) and made alkaline by the addition of sodium hydroxide solution (5 M), resulting in the formation of a white solid. The mixture was extracted with diethyl ether (3 × 30 mL). The organic fractions were combined, dried (MgSO4) and concentrated under reduced pressure to yield a brown oil (410 mg). 1H-NMR of indicated the isolated material was the starting material, 1a.
In the case of entry 3, the yield was calculated by comparing the integrals of the 1a imine proton (CDCl3 δ 8.22 ppm) with the 2a C1 proton (CDCl3 δ 5.14 ppm).[34]
Relevant lab book entries: KAB20-2, KAB20-3, KAB20-4.
Brønsted acid synthesis of 6,7-dimethoxy-1-phenyl-1,2,3,4-tetrahydroisoquinoline (2b)
Methanesulfonic Acid
To solution of N-[2-(3,4-Dimethoxyphenyl)ethyl]-1-phenylmethanimine (1b) (418 mg, 1.55 mmol) in toluene (40 mL) was added methanesulfonic acid (0.10 mL, 1.6 mmol) at 0 °C. The mixture was stirred at ambient temperature for 3.5 hours. Ethyl acetate (50 mL) was added. The mixture was quenched with saturated sodium bicarbonate solution (50 mL). The organic layer was separated and the aqueous layer was extracted with ethyl acetate (2 × 50 mL). The organic layers were combined, dried (MgSO4) and concentrated under reduced pressure yielding a yellow oil (>100%). 1H-NMR spectroscopy of the isolated material confirmed the presence of 6,7-dimethoxy-1-phenyl-1,2,3,4-tetrahydroisoquinoline (2b).[13] Relevant lab book entry: KAB21-1.
Trifluoroacetic Acid
To a stirring solution of N-[2-(3,4-Dimethoxyphenyl)ethyl]-1-phenylmethanimine (1b) (1.9 g, 7.1 mmol) in toluene (40 mL) was added trifluoroacetic acid (30 mL, 0.36 mol). The dark yellow solution was refluxed for 22 hours. etc. etc. etc. 1H-NMR of the isolated material confirmed the presence of 6,7-Dimethoxy-1-phenyl-1,2,3,4-tetrahydroisoquinoline (2b).[13] Relevant lab book entries: KAB21-2, KAB21-3, KAB21-4.
Brønsted acid synthesis of 6,7-dimethoxy-1-(4-nitrophenyl)-1,2,3,4-tetrahydroisoquinoline (2c)
To a pale yellow suspension of partially dissolved 2-(3,4-dimethoxyphenyl)-N-(4-nitrobenzylidene)ethanamine (1d) (401 mg, 1.28 mmol) in toluene (40 mL) was slowly added trifluoroacetic acid (0.20 mL, 2.6 mmol). The now clear, orange-red solution was reflux heated to 110 °C for 6.5 h. The mixture was allowed to cool then made alkaline with sodium hydroxide solution (6 M) to pH 9-10. The organic layer was isolated and the aqueous layer was extracted with ethyl acetate (3 × 30 mL). The organic layers were combined, dried (MgSO4) and concentrated under reduced pressure to give a dark yellow oil (524 mg, 131%). Purification of the oil by silica gel column chromatography (5% methanol/dichloromethane) yielded 6,7-dimethoxy-1-(4-nitrophenyl)-1,2,3,4-tetrahydroisoquinoline (2c) as a yellow solid (112 mg, 28%). Relevant lab book entries: KAB24-1, KAB24-2, KAB24-3, KAB24-4.
Procedure for the Lewis Acid-Catalyst Screen
Substrate stock solutions of N-benzylidene-2-(3,4-dimethoxyphenyl)ethanamine (1b) and 2-(3,4-dimethoxyphenyl)-N-(4-nitrobenzylidene)ethanamine (1d) were prepared (0.20 M in dichloromethane). To a mixture of the metal triflates (15-40 mg) at 20 mol% and microwave activated 3 Å powdered molecular sieves were the substrate stock solutions added. The mixtures were stirred at ambient temperature for 24 hours. The mixtures were quenched with saturated sodium bicarbonate solution and extracted with ethyl acetate. The organic layers were combined, dried (MgSO4) and concentrated under reduced pressure. 1H-NMR spectroscopy of the crude reaction mixtures indicated the products obtained consisted mostly of the corresponding starting material. Relevant lab book entry: KAB21-5, KAB21-6, KAB21-7, KAB21-8, KAB24-5, KAB24-6, KAB24-7 & KAB24-8.
Attempts at the Yb(OTf)3 catalysed synthesis of 6,7-dimethoxy-1-(4-nitrophenyl)-1,2,3,4-tetrahydroisoquinoline (2c)
Procedure 1[26]
To a mixture of Yb(OTf)3 (121 mg, 0.194 mmol, 0.2 equiv.) and microwave activated 3 Å powdered molecular sieves (~20 mg) was added dry dichloromethane (30 mL). Benzaldehyde (0.10 mL, 0.97 mmol, 1 equiv.) and 2-(3,4-dimethoxyphenyl)ethanamine (0.16 mL, 0.97 mmol, 1 equiv.) were added. The reaction mixture was stirred under nitrogen for 24 hours. Saturated sodium bicarbonate solution (30 mL) was added to quench the reaction. The organic layer was separated and the alkaline aqueous fraction was extracted with ethyl acetate (3 × 50 mL). The organic fractions were combined, dried (MgSO4) and concentrated under reduced pressure yielding a yellow oil (390 mg, 150%). 1H-NMR spectroscopy of the oil indicated the isolated product contained water and a 1:0.15 mixture of imine 1d and 4-nitrobenzaldehyde (5). Relevant lab book entries: KAB25-1.
Procedure 2[27]
To a mixture of Yb(OTf)3 (120 mg, 0.194 mmol) and microwave activated 3 Å powdered molecular sieves (~20 mg) was added dry toluene (30 mL) under a nitrogen atmosphere. Benzaldehyde (0.10 mL, 0.97 mmol) and 2-(3,4-dimethoxyphenyl)ethanamine (0.16 mL, 0.97 mmol) were added. The reaction mixture was reflux heated to 110 °C for 96 hours. The mixture was made alkaline by the addition of saturated sodium bicarbonate solution. The organic layer was separated and the aqueous fraction was extracted with ethyl acetate (3 × 30 mL). The organic fractions were combined, dried (MgSO4) and concentrated under reduced pressure to yield a yellow solid (360 mg, 130%). 1H-NMR spectroscopy of the oil indicated the isolated product was mostly composed of imine 1b. Relevant lab book entry: KAB25-2.
HAuCl4·3H2O/AgOTf catalysed synthesis of 1-(6,7-dimethoxy-1-(4-nitrophenyl)-3,4-dihydroisoquinolin-2(1H)-yl)ethanone (3)[13]
A solution of gold(III) chloride trihydrate (31 mg, 0.079 mmol, 0.01 equiv.) and silver(I) trifluoromethanesulfonate (30 mg, 0.12 mmol, 0.02 equiv.) in acetonitrile (15 mL) was vigorously stirred at ambient temperature (~17 °C) for 1 hour. To the now yellow reaction mixture was added a pale yellow solution of 2-(3,4-dimethoxyphenyl)-N-(4-nitrobenzylidene)ethanamine (1d) (1.9 g, 6.1 mmol, 1 equiv.), acetyl chloride (0.40 mL, 6.1 mmol, 1 equiv.) and 2,6-lutidine (0.70 mL, 6.1 mmol, 1 equiv.) in acetonitrile (250 mL). The reaction mixture was stirred for 14 hours at ambient temperature (~12 °C), concentrated under reduced pressure and purified by silica gel column chromatography (50-100% ethyl acetate/hexane, v/v). The expected product (3) was isolated (0.86 g, 41%) in addition to the byproducts, 4 and 5. Relevant lab book entries: KAB26-1 & KAB26-2, KAB26-3, KAB26-10.
1-(6,7-dimethoxy-1-(4-nitrophenyl)-3,4-dihydroisoquinolin-2(1H)-yl)ethanone (3d)
M.p. 177-179 °C. Two amide rotamers (91:9). Signals corresponding to the major rotamer: 1H-NMR (200 MHz; CDCl3): δ 2.18 (s, 3H), 2.81-2.71 (m, 1H), 2.95 (ddt, J = 15.9, 10.7, 5.2 Hz, 1H), 3.42-3.27 (m, 1H), 3.74-3.70 (m, 1H), 3.89 (s, 3H), 3.76 (s, 3H), 6.48 (s, 1H), 6.69 (s, 1H), 6.90 (s, 1H), 7.42 (d, J = 8.7 Hz, 2H), 8.12 (d, J = 8.7 Hz, 2H). Signals corresponding to the minor rotamer: 1H-NMR (200 MHz; CDCl3): δ 2.32 (s, 3H), 5.94 (s, 1H), 6.60 (s, 1H), 8.17 (s, 2H). 13C-NMR (75 MHz; CDCl3): δ 21.7, 28.4, 40.6, 54.1, 55.95, 56.00, 111.0, 111.3, 123.5, 125.6, 126.5, 129.5, 147.2, 148.0, 148.6, 149.7, 169.3. Spectroscopic data matched those in the literature.[13]
4-nitrobenzaldehyde (4)
M.p. 103.2 - 104.3 °C. 1H-NMR (300 MHz; CDCl3): δ 8.10 (d, J = 8.5 Hz, 2H), 8.42 (d, J = 8.6 Hz, 2H),10.18 (s, 1H). 13C-NMR (75 MHz; CDCl3): δ 124.3, 130.5, 140.0, 190.2. Spectroscopic data matched those in the literature.[13]
N-(3,4-dimethoxyphenethyl)acetamide (5)
M.p. 77-79 °C. 1H-NMR (300 MHz; CDCl3): δ 1.94 (s, 3H), 2.76 (t, J = 7.0 Hz, 2H), 3.48 (q, J = 6.6 Hz, 2H), 3.86 (s, 3H), 3.86 (s, 3H), 6.74-6.71 (m, 2H), 5.66 (s, 1H), 6.82-6.79 (m, 1H). 13C-NMR (75 MHz; CDCl3): δ 23.3, 35.2, 40.8, 55.86, 55.91, 111.4, 111.9, 114.7, 120.6, 131.4, 147.7, 149.0, 170.1.
Yb(OTf)3 catalysed synthesis of 1-(6,7-dimethoxy-1-(4-nitrophenyl)-3,4-dihydroisoquinolin-2(1H)-yl)ethanone (3)
Acetonitrile (HPLC grade) was dried over microwave activated 3 Å molecular sieves (2.5-5.0 mm, 30 %(w/v)) for >24 hours. All glassware were ovendried (130 °C) for >2 hours and cooled under inert gas prior to use. 2,6-Lutidine was dried over microwave activated 3 Å molecular sieves (diameter, 2.5-5.0 mm, 50 %(w/v)).
Procedure 1
To a solution of 2-(3,4-dimethoxyphenyl)-N-(4-nitrobenzylidene)ethanamine (0.365 g, 1.16 mmol, 1 equiv.) dissolved in acetonitrile (60 mL) were added microwave activated 3 Å powdered molecular sieves, acetyl chloride (0.08 mL, 1 mmol, 1 equiv.) and 2,6-lutidine (0.13 mL, 1.2 mmol, 1 equiv.). Yb(OTf)3 (60 mg, 0.097 mmol, 0.10 equiv.) was added. The mixture was stirred under argon for 15 hours, filtered through a bed of celite and concentrated under reduced pressure to yield the crude product. To a sample of the crude product (5.7 mg) was added 1,1,2,2-tetrachloroethane (8.8 mg) as an internal standard. The yield calculated from the 1H-NMR spectrum of the crude product was 81%. The crude product was washed with water. The aqueous layer was extracted with ethyl acetate. The organic layers were combined, dried (MgSO4) and concentrated under reduced pressure yielding a yellow oil. The 1-(6,7-dimethoxy-1-(4-nitrophenyl)-3,4-dihydroisoquinolin-2(1H)-yl)ethanone (3) product was isolated by silica gel column chromatography (ethyl acetate) as a yellow solid (220 mg, 53%). Relevant lab book entry: KAB26-4.
Procedure 2
As above, with added citric acid work-up prior to 1H-NMR spectroscopic analysis of the crude product. Relevant lab book entry: KAB26-5.
Procedure 3
As above, with cooling of the 2-(3,4-dimethoxyphenyl)-N-(4-nitrobenzylidene)ethanamine solution in an acetonitrile/liquid N2 bath before the addition of acetyl chloride, 2,6-lutidine and Yb(OTf)3. Also added alkaline wash of the organic layers following the citric acid wash. Relevant lab book entries. KAB26-6, KAB26-7 and KAB26-9.
Procedure 4
To a mixture of 3 Å molecular sieves (~30 g) in dry acetonitrile (160 mL), under nitrogen, was added 2-(3,4-dimethoxyphenyl)-N-(4-nitrobenzylidene)ethanamine (1.50 g, 4.77 mmol, 1 equiv.). Once dissolved, the mixture was cooled in a brine ice bath. Acetyl chloride (0.34 mL, 4.8 mmol, 1 equiv.) and 2,6-lutidine (0.55 mL, 4.8 mmol, 1 equiv.) were added, dropwise. Yb(OTf)3 (0.034 g, 0.048 mmol, 0.01 equiv.) was added. The reaction mixture was allowed to warm to ambient temperature (~12 °C) and stirred under argon for 23 hours. The mixture was filtered through a bed of Celite, eluting with ethyl acetate (~50 mL). The filtrate was washed with saturated sodium bicarbonate solution (40 mL). The aqueous layer was extracted with ethyl acetate (3 × 40 mL). The organic fractions were combined, dried (MgSO4) and concentrated under reduced pressure to yield a yellow oil that partially crystallised on standing (1.8 g, 106%). The crude product was dissolved in hot methanol, dry loaded onto a silica gel column (ø = 6.5 cm, 15 cm) and purified by chromatography (70-100% ethyl acetate/hexane) yielding the expected product as a yellow powder (1.3 g, 77%). Relevant lab book entry: KAB26-11.
Typical procedure for the 1H-NMR assays of the Yb(OTf)3 catalysed acyl-Pictet-Spengler reactions
Tetrachloroethane (8.0 mg, 4.8 × 10-5 mol) was added to a known amount of crude product (e.g. 10 mg). To the mixture was added CDCl3. The peaks were integrated and normalised based on the relative magnitudes of the integrated signals and their corresponding number of protons. The number of moles of product or by-product was determined by comparing the ratio of integrals with the known moles of added tetrachloroethane. The amount in moles was converted to mass, which was then divided by the amount of crude product dissolved in the CDCl3.
References
[1] Über die Bildung von Isochinolin-derivaten durch Einwirkung von Methylal auf Phenyl-äthylamin, Phenyl-alanin und Tyronsin, A. Pictet and T. Spengler, Ber. Dtsch. Chem. Ges. 1911, 44, 2030-2036. (DOI: 10.1002/cber.19110440309) Paper
[2] Strictosidine Synthase: Mechanism of a Pictet−Spengler Catalyzing Enzyme, J. J. Maresh, L.-A. Giddings, A. Friedrich, E. A. Loris, S. Panjikar, B. L. Trout, J. Stöckigt, B. Peters and S. E. O'Connor, J. Am. Chem. Soc. 2007, 130, 710-723. (DOI: 10.1021/ja077190z) Paper
[3] The Pictet–Spengler Reaction in Nature and in Organic Chemistry, J. Stöckigt, A. P. Antonchick, F. Wu and H. Waldmann, Angew. Chem., Int. Ed. 2011, 50, 8538-8564. (DOI: 10.1002/anie.201008071) Paper
[4] Reconstruction of the saframycin core scaffold defines dual Pictet-Spengler mechanisms, K. Koketsu, K. Watanabe, H. Suda, H. Oguri and H. Oikawa, Nat. Chem. Biol. 2010, 6, 408-410. (DOI: 10.1038/nchembio.365) Paper
[5] Pictet-Spenglerase involved in tetrahydroisoquinoline antibiotic biosynthesis, K. Koketsu, A. Minami, K. Watanabe, H. Oguri and H. Oikawa, Curr. Opin. Chem. Biol. 2012, 16, 142-149. (DOI: 10.1016/j.cbpa.2012.02.021) Paper
[6] Chemistry and Biology of the Tetrahydroisoquinoline Antitumor Antibiotics, J. D. Scott and R. M. Williams, Chem. Rev. 2002, 102, 1669-1730. (DOI: 10.1021/cr010212u) Paper
[7] Discovery of a Novel and Highly Potent Noncompetitive AMPA Receptor Antagonist, R. Gitto, M. L. Barreca, L. D. Luca, G. D. Sarro, G. Ferreri, S. Quartarone, E. Russo, A. Constanti and A. Chimirri, J. Med. Chem. 2003, 46, 197-200. (DOI: 10.1021/jm0210008) Paper;
[8] Affinity of 1-aryl-1,2,3,4-tetrahydroisoquinoline derivatives to the ion channel binding site of the NMDA receptor complex, M. Ludwig, C. E. Hoesl, G. Höfner and K. T. Wanner, Eur. J. Med. Chem. 2006, 41, 1003-1010. (DOI: 10.1016/j.ejmech.2006.03.005) Paper
[9] Efficient Multicomponent Reaction Synthesis of the Schistosomiasis Drug Praziquantel, H. Cao, H. Liu and A. Dömling, Chem. Eur. J. 2010, 16, 12296-12298. (DOI: 10.1002/chem.201002046) Paper
[10] (i) Phase II clinical trial of neoadjuvant trabectedin in patients with advanced localized myxoid liposarcoma, A. Gronchi, B. N. Bui, S. Bonvalot, S. Pilotti, S. Ferrari, P. Hohenberger, R. J. Hohl, G. D. Demetri, A. Le Cesne, P. Lardelli, I. Pérez, A. Nieto, J. C. Tercero, V. Alfaro, E. Tamborini and J. Y. Blay, Ann. Oncol. 2012, 23, 771-776. (DOI: 10.1093/annonc/mdr265) Paper; (ii) A retrospective pooled analysis of trabectedin safety in 1,132 patients with solid tumors treated in phase II clinical trials, A. Le Cesne, A. Yovine, J.-Y. Blay, S. Delaloge, R. G. Maki, J.-L. Misset, P. Frontelo, A. Nieto, J. J. Jiao and G. D. Demetri, Invest. New Drugs 2012, 30, 1193-1202. (DOI: 10.1007/s10637-011-9662-0) Paper
[11] Praziquantel and Schistosomiasis, A. Dömling and K. Khoury, ChemMedChem 2010, 5, 1420 – 1434. (DOI: 10.1002/cmdc.201000202) Paper
[12] Highly Enantioselective Catalytic Acyl-Pictet-Spengler Reactions, M. S. Taylor and E. N. Jacobsen, J. Am. Chem. Soc. 2004, 126, 10558-10559. (DOI: 10.1021/ja046259p) Paper
[13] Development of the Pictet-Spengler Reaction Catalyzed by AuCl3/AgOTf, S. W. Youn, J. Org. Chem 2006, 71, 2521-2523. (DOI: 10.1021/jo0524775) Paper
[14] Lewis Acid-Catalyzed Selective Synthesis of Diversely Substituted Indolo- and Pyrrolo[1,2-a]quinoxalines and Quinoxalinones by Modified Pictet–Spengler Reaction, A. K. Verma, R. R. Jha, V. K. Sankar, T. Aggarwal, R. P. Singh and R. Chandra, Eur. J. Org. Chem. 2011, 34, 6998-7070. (DOI: 10.1002/ejoc.201101013) Paper
[15] One-step preparation of 1-substituted tetrahydroisoquinolines via the Pictet–Spengler reaction using zeolite catalysts, A. Hegedüs and Z. Hell, TetLett 2004, 45, 8553–8555. (DOI: 10.1016/j.tetlet.2004.09.097) Paper
[16] Pictet-Spengler condensation reactions catalyzed by a recyclable H(+)-montmorillonite as a heterogeneous Brønsted acid, Wang, Y., Z. Song, et al. Science China-Chemistry 2010, 53(8), 562-568. (DOI: 10.1007/s11426-010-0073-4) Paper
[17] K. A. Badiola, M. N. Robertson, M. A. Tarselli and M. H. Todd, 2012, The Catalytic, Asymmetric Pictet-Spengler Reaction wiki, openwetware, 9 June 2012, http://openwetware.org/wiki/Todd:Catalytic%2C_Asymmetric_Pictet-Spengler_Reaction.
[18] Thiourea-Catalyzed Enantioselective Iso-Pictet–Spengler Reactions, Y. Lee, R. S. Klausen and E. N. Jacobsen, Org. Lett. 2011, 13, 5564-5567. (DOI: 10.1021/ol202300t) Paper
[19] (i) Catalytic Asymmetric Pictet–Spengler Reactions via Sulfenyliminium Ions, M. J. Wanner, R. N. S. van der Haas, K. R. de Cuba, J. H. van Maarseveen and H. Hiemstra, Angew. Chem., Int. Ed. 2007, 46, 7485-7487. (DOI: 10.1002/anie.200701808) Paper; (ii) Enantioselective Pictet–Spengler reactions of isatins for the synthesis of spiroindolones, J. J. Badillo, A. Silva-García, B. H. Shupe, J. C. Fettinger and A. K. Franz, Tetrahedron Lett. 2011, 52, 5550-5553. (DOI: 10.1016/j.tetlet.2011.08.071) Paper; (iii) Enantioselective Brønsted Acid-Catalyzed N-Acyliminium Cyclization Cascades, M. E. Muratore, C. A. Holloway, A. W. Pilling, R. I. Storer, G. Trevitt and D. J. Dixon, J. Am. Chem. Soc. 2009, 131, 10796-10797. (DOI: 10.1021/ja9024885) Paper
[20] Rare-Earth Metal Triflates in Organic Synthesis, S. Kobayashi, M. Sugiura, H. Kitagawa and W. W.-L. Lam, Chem. Rev. 2002, 102, 2227–2302. (DOI: 10.1021/cr010289i) Paper
[21] Cyclizations of N-Acyliminium Ions, B. E. M, H.-C. Zhang, J. H. Cohen, I. J. Turchi and C. A. Maryanoff, Chem. Rev. 2004, 104, 1431–1628. (DOI:10.1021/cr0306182) Paper
[22] Chiral Bis(oxazoline) Copper(II) Complexes: Versatile Catalysts for Enantioselective Cycloaddition, Aldol, Michael, and Carbonyl Ene Reactions, J. S. Johnson and D. A. Evans, Acc. Chem. Res. 2000, 33, 325-335. (DOI: 10.1021/ar960062n) Paper
[23] Trisoxazoline/Cu(II)-catalyzed asymmetric intramolecular Friedel-Crafts alkylation reaction of indoles, J.-L. Zhou, M.-C. Ye, X.-L. Sun and Y. Tang, Tetrahedron 2009 65, 6877-6881. (DOI: 10.1016/j.tet.2009.06.071) Paper
[24] Chiral 2,6-Bis(oxazolinyl)pyridine−Rare Earth Metal Complexes as Catalysts for Highly Enantioselective 1,3-Dipolar Cycloaddition Reactions of 2-Benzopyrylium-4-olates, H. Suga, K. Inoue, S. Inoue, A. Kakehi and M. S21hiro, J. Org. Chem. 2005, 70, 47-56. (DOI: 10.1021/jo049007f) Paper
[25] Self-assembled organic–inorganic hybrid silica with ionic liquid framework: a novel support for the catalytic enantioselective Strecker reaction of imines using Yb(OTf)3–pybox catalyst, B. Karimi, A. Maleki, D. Elhamifar, J. H. Clark and A. J. Hunt, Chem. Commun. 2010, 46, 6947-6949. (DOI: 10.1039/c0cc01426e) Paper
[26] Catalytic Pictet-Spengler reactions using Yb(OTf)3, K. Manabe, D. Nobutou and S. Kobayashi, Bioorg. Med. Chem. 2005, 13, 5154-5158. (DOI: 10.1016/j.bmc.2005.05.018) Paper
[27] Calcium-Promoted Pictet-Spengler Reactions of Ketones and Aldehydes, M. J. V. Eynden, K. Kunchithapatham and J. P. Stambuli, J. Org. Chem. 2010, 75, 8542-8549. (DOI: 10.1021/jo1019283) Paper
[28] (i) Synthesis of carbon-11 and fluorine-18 labeled N-acetyl-1-aryl-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline
derivatives as new potential PET AMPA receptor ligands, M. Gao, D. Kong, A. Clearfield, Q.-H. Zheng, Bioorg. Med. Chem. Lett. 2006, 16, 2229-2233. (DOI: 10.1016/j.bmcl.2006.01.042) Paper; (ii) Synthesis of stable azomethine ylides by the rearrangement of 1,3-dipolar cycloadducts of 3,4-dihydroisoquinoline-2-oxides with DMAD, N. Coşkun and S. Tunçman, Tetrahedron 2006, 62, 1345-1350. (DOI: 10.1016/j.tet.2005.11.040) Paper; (iii) Synthesis, antibacterial activity and QSAR studies of 1,2-disubstituted-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinolines, R. K. Tiwari, D. Singh, J. Singh, A. K. Chhillar, R. Chandra and A. K. Verma, Eur. J. Med. Chem. 2006, 41, 40-49. (DOI: 10.1016/j.ejmech.2005.10.010) Paper
[29] Imine 1d showed trace amounts of the 4-nitrobenzaldhyde starting material, which promted recrystallisation of the pure imine from ethanol to yield the final product. Imine 1c required no further purification.
[30] In Situ Selection of Lead Compounds by Click Chemistry: Target-Guided Optimization of Acetylcholinesterase Inhibitors, A. Krasiński, Z. Radić, R. Manetsch, J. Raushel, P. Taylor, K. B. Sharpless and H. C. Kolb, J. Am. Chem. Soc. 2005, 127, 6686-6692. (DOI: 10.1021/ja043031t) Paper
[31] Evaluation of the relative Lewis acidities of lanthanoid(III) compounds by tandem mass spectrometry, H. Tsuruta, K. Yamaguchi and T. Imamoto, Chem. Commun. 1999, 1703-1704. (DOI: 10.1039/A905569J) Paper
[32] Principal properties (PPs) for lanthanide triflates as Lewis-acid catalysts, C. G. Fortuna, G. Musumarra, M. Nardi, A. Procopio and G. Sindona, S. Scirè, J. Chemom. 2006, 20, 418-424. (DOI: 10.1002/cem.1016) Paper
[33] Highly efficient Lewis acid-catalysed Pictet-Spengler reactions discovered by parallel screening, N. Srinivasan and A. Ganesan, Chem. Commun. 2003, 916-917. (DOI: 10.1039/B212063A) Paper
[34] Isoquinoline-N-Boranes as Precursors to Substituted Tetrahydroisoquinolines, D. J. Brooks, D. S. Dowell, D. E. Minter and M. C. Villarreal, J. Org. Chem. 1984, 49, 130-133. (DOI: 10.1021/jo00175a027) Paper
Unsorted Reaction Tally
Imine Syntheses
Model PS Cyclizations
KAB21-5, KAB21-6, KAB21-7, KAB21-8, KAB24-5. KAB24-6, KAB24-7, KAB24-8
Summary of Yb(OTf)3 catalysed acyl-PS reactions
LCMS Assay Standardisation for MNR41-7 and PZQ
MNR41-15 re sieves
