Todd:Chem3x11 ToddL8
Chem3x11 Lecture 8
This lecture is about how things add to alkenes, using bromine as an example.
(Back to the main teaching page)
Key concepts
- The stereochemical outcome of a reaction gives clues to the mechanism of that reaction
- The addition of bromine to double bonds is via a cyclic bromonium ion that is formed in a concerted process
The Addition of Bromine to a Double Bond
Alkenes are important because they occur naturally in great quantity and we can use the reactive double bond to make more interesting molecules. The formation of epoxides is one example we have seen of how alkenes can be transformed into other molecules. The addition of bromine is another widely-used reaction. The jargon for this reaction is an electrophilic 1,2-addition. The bromine adds such that one bromine is added to one carbon and one to the other (a 1,2-addition) and the reagent we add (bromine) is seeking out the electrons of the double bond, meaning we refer to this as an electrophilic addition (unlike when a nucleophile adds to a carbonyl group, which is a nucleophilic addition).
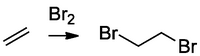
Bromine is mostly fine as a reagent, but people often use the more convenient NBS (N-bromosuccinimide), which maintains a low level of Br2 in solution. More recently Scott Snyder developed a nice bromination reagent BDSB which you can now buy.
Some Mechanistic Possibilities
How might this reaction be occurring? Perhaps a concerted mechanism like this:
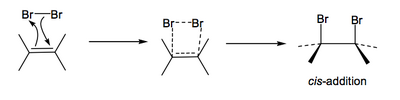
...or a stepwise polar mechanism like this:
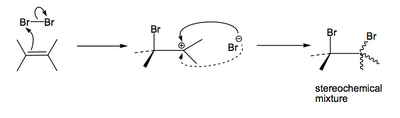
...or maybe something exotic like a stepwise radical mechanism like this:
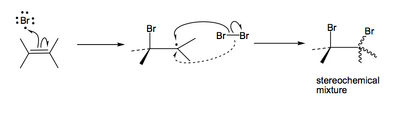
On paper there is no easy way to say which is right, so we need to go to the lab, do the reaction carefully and see what happens. If you remember back to when we looked at the opening of epoxides, we found that we learned a lot about the reaction mechanism by using Z- and E- isomers of an alkene. Let's try that trick again.
Stereochemical Clues to the Addition Mechanism
Imagine we have some double bond with things on the end (R1 and R2). Let's start with the Z-isomer, and let's add some reagent Y2 to the double bond and let's say for the moment that the addition does not go via some carbocation or radical intermediate and think about what happens if the addition is cis or trans (i.e., if both atoms of Y2 add to the same face or to opposite faces.)

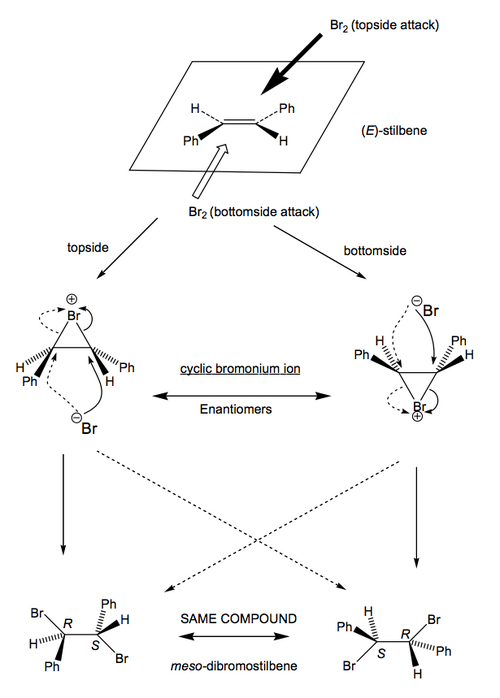
This rather complex diagram just shows that the outcomes from the different starting material isomers will be different for either cis or trans mechanisms, so we ought to be able to work out what's happened when we do the experiment. To make it simpler, imagine we go in the lab and pick the two isomers of stilbene, meaning that R1 and R2 are both phenyl groups and X = H. We do the reaction, and discover the following:
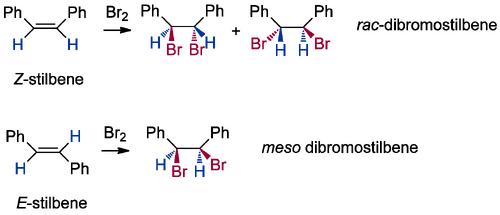
...which implies trans addition without a doubt. The question is, how? We'd need to measure more things about this reaction. We look at the same reaction in a cyclohexenyl system, and we observe selective formation of trans-diaxial products:

...which is reminiscient of what we've seen before in epoxide openings. Could it be a similar mechanism - the formation of an epoxide-like intermediate, with rear-face attack? When we measure the kinetics of the reaction we find it's first order in both alkene and bromine. We also change the alkene structure a little and find that the reaction is accelerated when there are electron-donating substituents on either carbon, and significant rate enhancements when both carbons have electron-donating substituents attached. Then you see that someone has tried the reaction with a very sterically crowded alkene shown below and trapped an intermediate bromonium ion (which was later crystallized) where one bromine is bridging the two carbons that used to form the double bond. How cool is that?

(in this case the bromonium ion, which usually reacts with bromide to give the final product, is so sterically hindered that it can't)
All this, and other bits of evidence (See Anslyn and Dougherty section 10.5.3), suggest this bromonium ion is the intermediate formed (that we do not normally see) and that it's on the productive reaction pathway (and not something irrelevant). Its existence is the reason the electrophilic addition of bromine to alkenes proceeds to give the product with trans stereochemistry.
How Do We Draw The Mechanism for Formation of the Bromonium Ion?
The reaction to give the bromonium ion involves the formation of two bonds, so our mechanism needs to show that. We can do such a thing in one step if electrons come from the bromine and add to the alkene, and at the same time come from the alkene and add to the bromine. Thus the HOMO and LUMO of each interact with each other in this way:
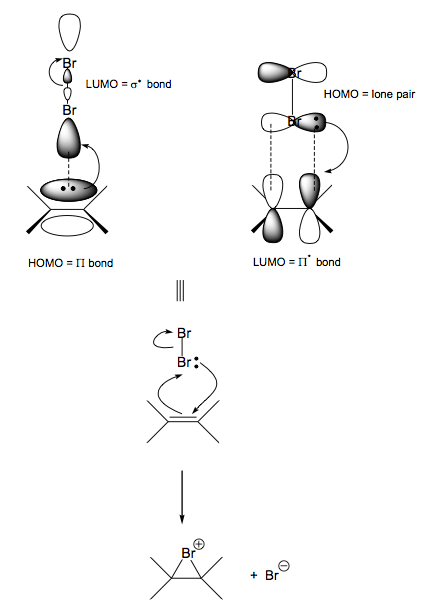
Though this drawing may at first site be rather odd, the lobes of the MOs match up perfectly for this to take place. Clearly in most cases the HOMOs and LUMOs are close in energy here and can interact productively. The reaction is exothermic. The curly arrows depict one of the bromines adding to one of the carbons, and the double bond adding "back" to the same bromine, with concerted cleavage of the Br-Br bond. The end result is a single bromine atom bridging the two carbons.
Under certain conditions, when the rate of reaction between an alkene and bromine is measured, the rate is found to be second order in Br2 (below). Can you propose a mechanism for this - i.e. a way that a second Br2 could help speed things along? You end up generating a weird-looking byproduct, Br3-.
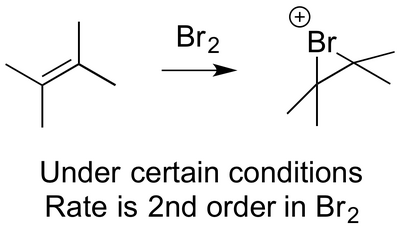
Mechanism for Addition of Bromide Ion to the Intermediate Bromonium Ion
This is rather like what we have already seen for epoxide opening. Don't learn any given case. Work it out from scratch. We're dealing with achiral starting materials (the alkene and the bromine), so there can be no preference for a certain "side" of the double bond for the bromine to interact with. You need to consider both, and see what the outcome is.
For Z-stilbene:
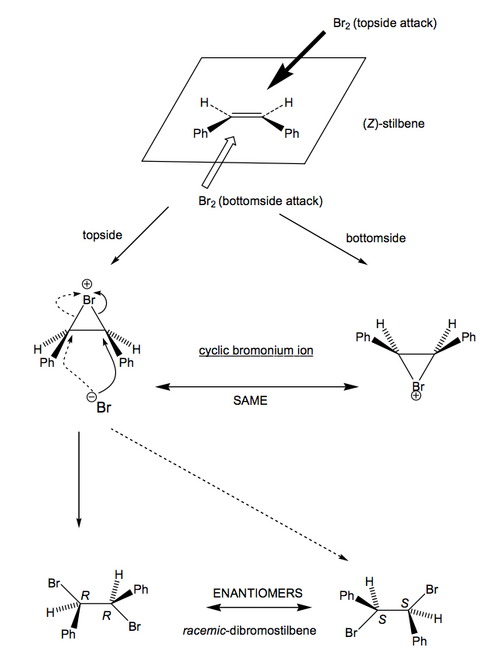
For E-stilbene:
An Easy Problem
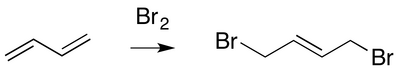
Addition of bromine to butadiene (below) can lead to the product shown. Suggest a mechanism.
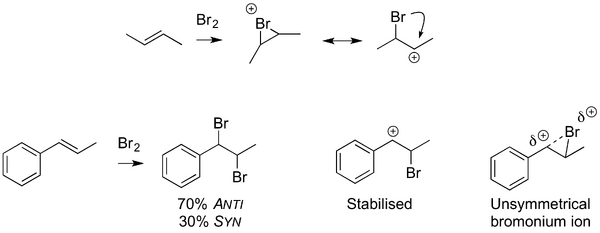
Unsymmetrically Substituted Double Bonds
We can think of bromonium ions as one resonance form of a carbocation with an adjacent bromine atom, below. In fact we're right to think more of the bromonium ion: we know they exist from all the evidence above (they've been crystallised, they lead to stereocontrol in the subsequent addition of bromide, etc). But if we employ unsymmetrical alkenes, particularly ones where carbocations can be stabilised, then we start to see a little behaviour that arises from the carbocation form. In the case of the phenyl-substituted double bond below, a benzylic carbocation can be stabilised, and we end up with imperfect anti addition of the bromine to the double bond. We can imagine that some of the time, the bromonium ion exists as the carbocation, and can react non-selectively with bromide. We can represent this by an unsymmetrical bromonium ion, rather like the unsymmetrical protonated epoxides we saw in an earlier lecture.
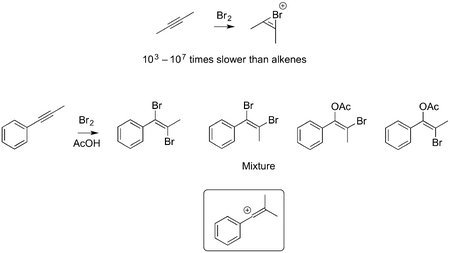
A Note on Alkynes
A bromonium ion derived from an alkyne is more strained than that from an alkene, meaning such a compound is slower to form - the result is that alkynes react more slowly with bromine than alkenes. However, if there is a way to stabilise a charge following addition of bromine to an alkyne (such as the phenyl-substituted compound below) then a vinyl carbocation (rather than a bromonium ion) can form, leading to mixtures of products - there is no control in the addition of the subsequent nucleophile, though because the cation forms next to the Ph, we don't get every conceivable product here. (Examples taken from Anslyn and Dougherty, section 10.5.5)
The Licence for This Page
Is CC-BY-3.0 meaning you can use whatever you want, provided you cite me.
