Todd:Catalytic, Asymmetric Pictet-Spengler Reaction
The Catalytic, Asymmetric Pictet-Spengler Reaction
Katrina A. Badiola, School of Chemistry, The University of Sydney, NSW 2006, Australia
Murray N. Robertson, School of Chemistry, The University of Sydney, NSW 2006, Australia
Katina J. Selvaraj, School of Chemistry, The University of Sydney, NSW 2006, Australia
Michael A. Tarselli, Novartis Institutes for Biomedical Research, Cambridge, MA, United States of America
Matthew H. Todd, School of Chemistry, The University of Sydney, NSW 2006, Australia
Inga L. Topolnicki, School of Chemistry, The University of Sydney, NSW 2006, Australia
Alice E. Williamson, School of Chemistry, The University of Sydney, NSW 2006, Australia
Additional authors - add alphabetically if you contribute something substantial (e.g., the summary of a paper with a scheme). Please include some public place you can be contacted, e.g. a G+ account.
The licence for this page is CC BY 3.0
How this works:
0. Background described in this blog post. Here are some thoughts on this by others, too.
1. This article will be a stand-alone review. It will be written openly in January and February 2012. Once it is complete and well-written, it will be submitted to an open access journal for publication. This will provide a citation - a static object to which others may refer. Until that time this page is a work in progress and is not to be taken as complete.
2. The article is open source, like Wikipedia. Anyone can add and edit. To contribute you will need to get an account on OWW (quick and easy). People contributing something lasting and substantial will be named authors. Final arbitration on what qualifies as authorship lies with Mat Todd, who is the corresponding author for this paper.
3. References for the papers described here may be found in full at the Mendeley page. Chemdraw files for the schemes are currently held in a Dropbox folder we can share with anyone who needs it. PNG files are hosted on this wiki site.
4. The review incidentally acts as background to the open science project to find a catalytic, asymmetric route to praziquantel. That project is currently active in the lab. A catalytic, enantioselective synthesis of this drug is an alternative to the resolution approach that was discovered, also by open science.
5. If you want to get in touch to ask questions (e.g. if you want to ask "what can I do to help?") please do not use email. The current to-do list is on the talk page (click on the tab above). Questions should go there with your initials. You can also discuss via Google+ pages: Mat, Kat, (please add other public places where you can be contacted if you contribute as an author by inserting links along with your name, above).
6. When editing remember to include a reason for your edit, to make it easier to revert back to an original version if needed.
Important note on simultaneous edits: If you are intending to work for some time on editing the page, we'd recommend writing text elsewhere then pasting it in here, since there is a small but non-zero chance that you might simultaneously edit the same section as someone else, resulting in the chance of the loss of some information.
Schemes: Use Wiley/Angewandte settings for the .cdx files and add below as 300 dpi .png files.
References: Please use the citation style below (click to see the page source for formatting details) and include a link to the DOI of the paper to that it may be checked easily.
Introduction
The Pictet-Spengler (PS) reaction is just over a century old.Pictet, Spengler 1911 The reaction is a cyclization between an amine that carries an aromatic ring, and an aldehyde, usually catalyzed by acid (Scheme 1911 Pictet Spengler 2). The original reaction employed formaldehyde and phenethylamine, to give the tetrahydroisoquinoline scaffold. Several years after the original report tryptamine was found to perform well in the reaction permitting access to a range of tetrahydrocarbolines.[Tatsui 1928] A wide range of variations on these original themes have been investigated,Stöckigt 2011, Whaley, Govindachari[Youn 2006][Larghi 2011] such as cases where the amine component is acylated or alkylated, those where ketones are employed rather than aldehydes so as to generate quaternary centres adjacent to the aromatic ring, and reactions employing alcohols rather than amines - the so-called oxo-PS reaction.

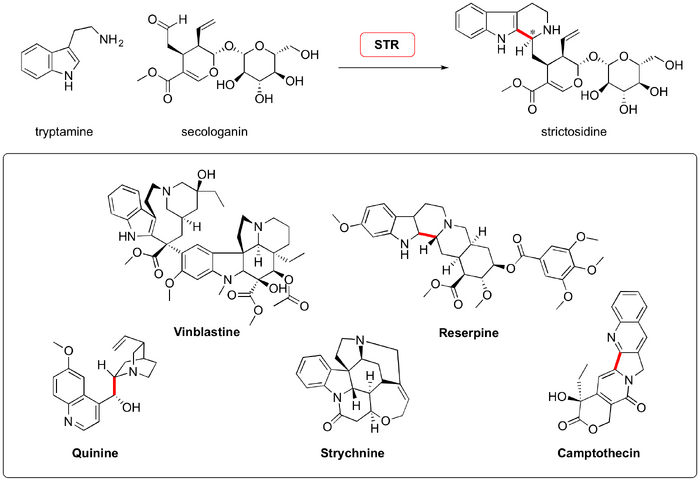
The reaction is important for two reasons. Firstly, Nature uses this chemistry. Enzymes ("pictet spenglerases") carry out the PS cyclization to produce important intermediates which feed into many biological pathways that result in bioactive small molecules, such as strychnine, morphine, vinblastine and reserpine. All monoterpene indole alkaloids are thought to be made via this route, the key intermediate of which is strictosidine, formed from a PS reaction between tryptamine and secologanin.
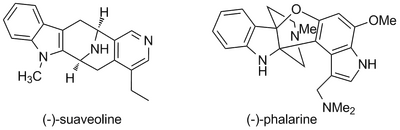
Secondly, and probably consequently, the general structures one can access through the PS reaction - alkaloids with a stereocentre adjacent to an aromatic ring - are often highly bioactive, and are of interest for the development of new medicines. This has led to a great deal of interest in controlling the stereochemical outcome of PS reactions. Most of this work has involved understanding diastereoselective PS reactions, employing either an existing stereocentre that remains in the final product (often derived from an amino acid) or a stereocentre in a chiral auxiliary that is eventually removed, directs the ring closure.Cook 1995(Need: What was the first diastereoselective example? Cook 1992 Cook[Larghi 2005, review, no DOI][Youn 2006] This approach has been used in several notable total syntheses, among them (-)-suaveoline (Cook, 1992) and (-)-phalarine (Danishefsky, 2010). (Need: combine the two structures below with the general scheme for this part)
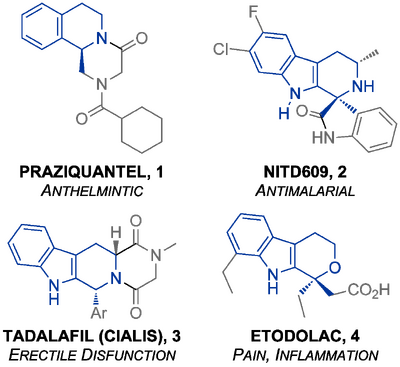
Several important pharmaceuticals may be synthesized with the PS reaction, for example the widely-used anthelmintic praziquantel, tadalafil (Cialis(TM)) used for erectile disfunction, the painkiller Etodolac and the promising new antimalarial compound NITD609 (Scheme Intro - Relevant Drugs). The biological activity of these compounds typically arises from one enantiomer: for example the (S) enantiomer of Etodolac (10.1021/jm00366a025) and the (R)- enantiomer of praziquantel (10.1371/journal.pntd.0000357); ent-NITD609 is inactive (10.1126/science.1193225). The potency of this class of compounds leads to great market value: $1.7 Bn for tadalafil alone in 2010.(Need: full reference inserting at end)(Eli Lilly Annual Report 2010, available at http://investor.lilly.com/annuals.cfm) Efficient enantioselective methods for the PS reaction would be very desirable, and progress to date in this field is the subject of this review. It is now certainly possible to carry out some PS reactions to give products with high enantiomeric excess. However, the scope of these processes is limited (as we shall see) reducing their impact on the preparation of bioactive compounds industrially. The PS reaction has been used in the racemic industrial synthesis of X, for example, but the enantiopure material is obtained through a resolution.(Need: example here - someone please check http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3107522/)
Enantioselective approaches, where the stereocontrol of the cyclization is not governed by a stereocentre already in the cyclization precursor, have received much less attention than the diastereoselective, racemic or achiral version of the PS reaction. It was shown in 1996 that the PS reaction could be effected with superstoichiometric Lewis acid - in this case the cyclization of the (Z)-nitrone derived from Nb-hydroxytryptamine using diisopinocampheylchloroborane (Ipc2BCl) to give the corresponding tetrahydro-β-carboline products (Scheme Nakagawa 1998).(10.1016/0957-4166(96)00134-6 and 10.1021/jo980810h and 10.3987/REV-98-SR1) The racemic cyclization could be effected with Bronsted acid and a number of achiral Lewis acids, but it was found that Ipc2BCl gave high yields and ee with reduced temperatures. Lowering the quantity of Ipc2BCl to 0.5 eq. caused a significant reduction of yield, while attempts to alter the Lewis acidity by substitution of the chloride (with e.g. fluoride or triflate) did not improve yield or ee. The proposed reaction mechanism involved the formation of an iminium ion with coordination of the nitrone oxygen to the Ipc2BCl boron centre, and preliminary modeling confirmed a difference in transition state energies for approach of the indole from the re- and si-faces of the iminium ion. It was found that iminium ions derived from electron-deficient aldehydes gave poor enantioselectivity, but that these cyclizations were effective (with 74% ee in the case of the nitrone derived from 4-nitrobenzaldehyde) when a different reagent was added, a boronate incorporating two BINOL ligands (X) that had been previously described by Yamamoto for aldol-type reactions of imines.(10.1021/ja00102a019) The mechanism of action of such a boronate was suggested to involve replacement of one of the coordinating BINOL oxygen atoms with that on the nitrone, and it is not clear why such a mechanism could not operate catalytically. Nevertheless this was the first report of a reagent-controlled enantioselective PS reaction; earlier work by Nakagawa employing a similar approach had led to cyclization, but to give enantioenriched spiroindoline products.Paper Chiral Bronsted acids were ultimately successfully applied to the catalytic, enantioselective PS reaction through the use of a phosphoric acid, rather than a boronate, as described below.
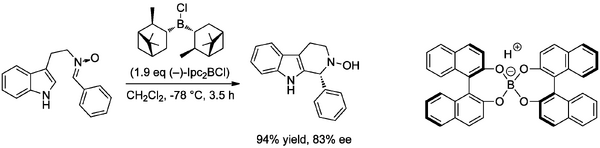
A chiral Lewis acidic silane reagent (X, Scheme Leighton 2009) was shown to be effective in promoting highly enantioselective PS reactions.(Leighton 2009 10.1002/anie.200806110) The substrates (e.g., X) contained α-ketoamide ketimines: NMR studies suggested that following O-silylation, the proton from the NH group transfers to the reagent's nitrogen atom and activates the complex (X) to cyclization. Electron-withdrawing groups on the N-aryl ring improved the reaction rate. The quaternary stereocentre could be generated even with sterically demanding aryl groups appended to the imine, such as 1-naphthyl (product ee 87%). A one-pot procedure was also developed in which the initial amine and α-ketoamide were allowed to react, followed by the addition of the silane (X). The process was shown to be effective on a 5 mmol scale with 1.3 equivalents of silane, which was quantitatively recovered following work-up of the reaction. It was subsequently shown that the same (now commercially-available[Leighton 2010]) reagent could be used with similar effectiveness in the synthesis of the more ususual PS reaction products X and X, derived from the less common indole amines (1H-indol-4-yl)methanamine and 2-(1H-indol-1-yl)ethanamine.[Leighton 2012 10.1021/ol300922b] The latter heterocyclic framework had just been synthesized using the enzyme strictosidine synthase (see section X).

It is important to note that several papers have been published which contain brief sections devoted to topics covered in this review (Botta 2012, Jacobsen 2013, Cozzi 2015, Kroutil 2015, Wang 2016, D'Acquarica 2016). Although these articles do not focus specifically on asymmetric, catalytic Pictet-Spengler reactions, they demonstrate the broad range of applications of related chemistry.
It is perhaps surprising that there are still no examples of Lewis acid-catalyzed asymmetric PS reactions. In a recent study (10.1016/j.tetlet.2011.08.071) a small number of chiral Lewis acidic complexes were shown to effect conversion in a PS reaction between tryptamine and isatins, but there was negligible enantioinduction; high product ee was ultimately achieved with Bronsted acid catalysis, to which we now turn.
Brønsted Acid Organocatalysis
(Note - consider a diagram section at start which includes the structures of all the BINOL-derived catalysts so we don't have to include in the individual schemes). i.e. we draw them at start and then number them then just include the numbers in the schemes.
Chiral Brønsted acids have been shown to be effective in the catalytic, asymmetric PS reaction, which builds on earlier work demonstrating the ability of such compounds to catalyze the reaction between nucleophiles and iminium ions.
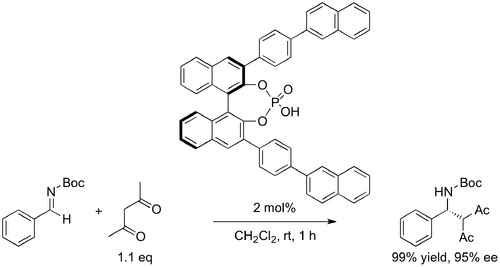
Akiyama (10.1002/anie.200353240) reported chiral phosphoric acids prepared from arylated BINOLs in the enantioselective Mannich-type coupling of silyl enolates with aldimines (Scheme Akiyama 2004). High yields and enantio- (as well as, in appropriate cases, diastereo-) selectivities were observed with a variety of substituted aldimines and enolates (What's "high" for this reaction? Akiyama reports 81-96% ee, and given that the group reports several "100% yields" I'm inclined towards suspicion -MAT). Limitations to the methodology were that an ortho hydroxy group was required on the N-aryl ring of the aldimine (Why? Mechanistic raionale? - MAT), and that aldimines derived from aliphatic aldehydes did not participate effectively. Typical catalyst loading was 10 mol%. The structure of the catalyst itself may be thought of as a chiral proton, i.e., a proton surrounded by a chiral environment, particularly given the aromatic rings of the BINOL and the 3-substituents are non-coplanar. However, the proposed mechanism operates via an ion pair of phosphate and iminium ion. The bond-forming event would naturally disrupt such an ion pair, ensuring catalytic turnover. (ultimately need a scheme of this very general idea [nice example in the Akiyama paper here] but may go in mechanism section)

At the same time Terada (10.1021/ja0491533) reported similar catalysts in the enantioselective Mannich reaction for the synthesis of β-aminoketones (Scheme Terada 2004a), utilizing acetylacetonate nucleophiles in place of silyl enolates. Terada, like Akiyama, notes the important influence of the 3,3'-position of the naphthyl rings on the enantioselectivity of the reaction. A followup paper later that same year incorporates furan nucleophiles (10.1021/ja046185h).
Since these early examples, several reactions have been reported that can be catalysed by these or related structures (reference reviews of chiral bronsted acid catalysts here).(Terada 2010 Bull Chem Soc Jpn 10.1246/bcsj.20090268)
List reported the first Brønsted acid-catalyzed enantioselective Pictet-Spengler reaction in 2006 (10.1021/ja057444l). Chiral, substituted phosphoric acids were shown to be effective in the PS cyclization of tryptamines with a number of aliphatic and aromatic aldehydes (Scheme List 2006). The diester functionality was found to be necessary, presumably due to promotion of a clean reaction through the Thorpe-Ingold effect (and an aldol side reaction was observed when the esters were absent). Lower yields were typically observed when the methoxy group was absent from the tryptamine aromatic ring.

In 2007, Hiemstra reported the enantioselective synthesis of tetra-β-carbolines via the in situ formation of N-sulfenyliminium ions (10.1002/anie.200701808). Stabilization of the intermediate iminium by the N-tritylsulfenyl group was effective at promoting the acid-catalyzed PS reaction by substituted enantiopure binaphthyl-derived phosphoric acids. Several substitutions were assayed in the 2-position of the catalyst, with no clear trend being observed in the ee of product obtained. The N-S bond in the N-tritylsulfenyl product was found to be susceptible to homolytic cleavage, but this could be suppressed by the addition of a radical scavenger. A one-pot process was developed that allowed precipitation of the product as a salt, and this was applied to the synthesis of a variety of substituted tetra-β-carbolines with high yield and high ee. The reaction was also demonstrated on a multi-gram scale.
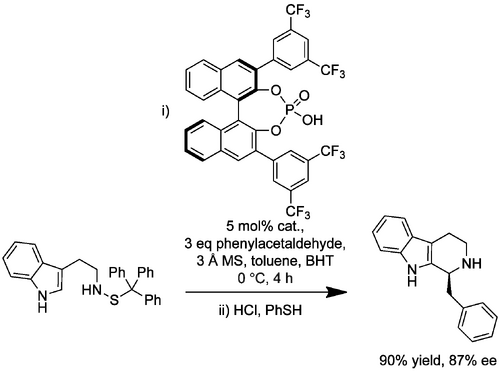
An extension to this methodology was developed that allowed the synthesis of enantioenriched N-benzyl-protected versions of similar products from the relevant protected tryptamines and diverse aldehydes (10.1021/jo8010478). During optimization it was found that removal of water was essential for high enantioinduction presumably because water prevents effective association between catalyst and cyclization precursor. Several control reactions were performed under the optimized conditions that suggested this PS reaction was irreversible. The best-performing catalyst was the triphenylsilyl-substituted binaphthyl system, delivering up to 100% conversion and high ee values (78-85%). The ee obtained was sensitive to the aldehyde employed. Of the aliphatic aldehydes, no product was observed with the enolizable phenylacetaldehyde and low ee (8%) was obtained with 3-phenylpropanal. While electron-deficient aromatic aldehydes generally gave products with high ee as expected, there were exceptions that performed poorly; 3-chlorobenzaldehyde gave near-racemic product, for example.
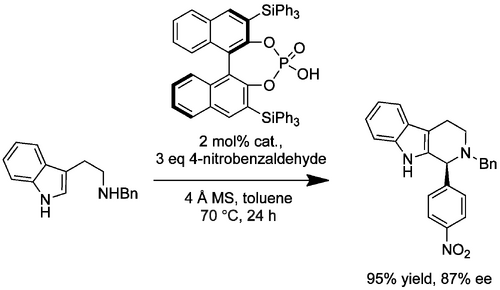
This methodology has been employed in the syntheses of three natural products. The PS reaction used in the synthesis of (-)-arboricine (10.1021/ol900888e) (Scheme Hiemstra 2009) involved an aldehyde containing a dioxolane-protected ketone group. Aminal formation, observed when the ketone was used unprotected, was prevented. It is notable that this protecting group withstands the PS cyclisation, and that the yield and ee of the cyclization were both dramatically improved by its use. The partially saturated (and slightly more sterically crowded) (R)-H8-Binol-PA catalyst was also shown to be effective. This catalyst was subsequently used for the key step in the synthesis of (+)-yohimbine (Scheme Hiemstra 2011).(10.1021/jo201657n). The natural product was to be synthesized via a Diels-Alder precursor that could itself be made using an enantioselective PS reaction. However, the aldehyde required for the PS reaction was β,γ-unsaturated and this was likely to result in the unproductive formation of an enamine from the initially-formed iminium ion. This substrate limitation necessitated use of a latent double bond, in this case a phenylselenide; this group survived the successful PS cyclization and could be eliminated to the double bond via oxidation to the selenoxide. A similar synthetic strategy was employed in the synthesis of the related corynanthe alkaloid family. (10.1002/chem.201103150)


Dixon included a chiral phosphoric acid as part of a reagent cocktail effecting a cascade sequence involving a Pictet-Spengler-like cyclization (Scheme Dixon 2009).(10.1021/ja9024885) Tryptamines and lactones formed ketoamides with an appended π-nucleophile that underwent enantioselective cyclizations in the presence of chiral phosphoric acids, and it was again shown that aromatic substitution of the BINOL ring system was essential for high ee. The method could be used with more substituted lactones to effect high levels of diastereocontrol: the combination of a disubstituted enol lactone with tryptamine gave isolable intermediates, the structures of which implied that the formation of the reactive iminium ion was fast and reversible, and that final ring closure occurred with one matched catalyst/substrate pair. The mechanism of the enantiodetermining cyclization is presumed to be via a tight ion pairing between iminium ion and catalyst conjugate anion. High yields and stereoselectivities could be obtained for diverse products using this methodology, which was shown to be compatible with a one-pot cascade process that also included a gold(I)-catalyzed step to generate the initial lactone.

The method was broadened to allow the use of racemic keto acids and esters in place of the enol lactones, again with polycyclic products being produced in high yield and ee (79-98%). (10.1021/ol101651t). If the reaction time was reduced, enamides could again be isolated, one achiral and the other chiral with an ee of only 7%. Either enamide gave the intended product with an ee of 83% when resubjected to the reaction conditions, supporting the fast, reversible formation of iminium ions which are trapped by an enantioselective cyclization event controlled by the chiral acid. Possibly mention the racemic oxo acid.

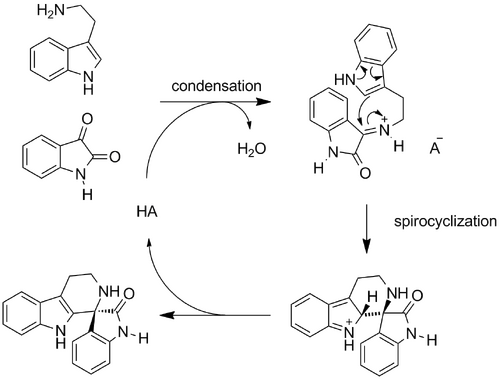
Bernardi and Bencivenni presented the first asymmetric synthesis of medicinally-relevant spiroindolones from tryptamines and isatins (Scheme Spiroindolones 2011) (10.1002/adsc.201100050). A number of (S)-BINOL derived phosphoric acids were screened, but all gave poor enantioselectivity except the (S)-TRIP catalyst originally used by List (A). The reaction performed best in DMF. The addition of molecular sieves gave no improvement, and it was found that the reaction can tolerate moisture. The conditions were applicable to tryptamines with electron-withdrawing or electron-donating substitutents at the 5-position, and to a variety of substituted isatins, although the presence of a bulky benzyl substituent at nitrogen reduced enantioselectivity. At the same time, Franz screened a greater variety of catalysts in reactions between 5-methoxy tryptamine and isatins (10.1016/j.tetlet.2011.08.071). Lewis acidic complexes proved ineffective, and although a thiourea catalyst gave some enantioselectivity, it was determined that (S)- and (R)-BINOL derived phosphoric acids were most effective, giving very high enantioselectivities and yields in some cases. Notably, phosphonic acid gave near quantitative yield but no enantioselectivity. The 3,3'-substituents on the BINOL ring system strongly influenced the enantioselectivity; interestingly, even when the substituents were changed such that the axial chirality was reversed (for example, from 9-anthracenyl to 2,4,6-tri-i-Pr-phenyl), the same spiroindolone enantiomer resulted, that is, the sense of enantioselection was inverted. The (S)-TRIP catalyst used by Bernardi and Bencivenni was investigated again (Scheme Spiroindolones (B)). Various substituted isatins were well tolerated, but tryptamines with substituents at positions other than the 5-position were not. This method was extended to N-propargylated isatins, and the resulting N-propargylated spiroindolones were converted to biologically interesting triazoles via copper-catalysed azide-alkyne cycloaddition reactions (10.1021/co300003c).
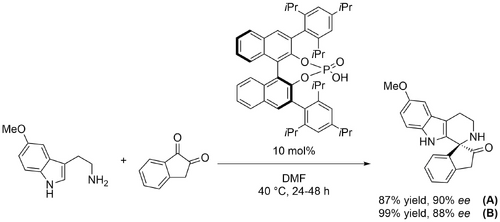
Chiral phosphoric acids have also been used in catalytic asymmetric PS-type reactions to give 7-membered indolo[3,4-cd][1]-benzapines x. Tian and co-workers reported the phosphoric acid catalysed reaction of 4-(2-aminoaryl)insoles x with para-methoxybenezene (PMB) protected aryl imines. X (Scheme X). The researchers screened a number of imine and aldehyde nucleophiles and found that: 1) PMB to be the optimum imine protecting group, 2) the reaction proceeded with higher ee at greater concentration and 3) PMB protected imines reacted with higher enantioselectivity than their corresponding aldehyde (90 compared to 83% ee).
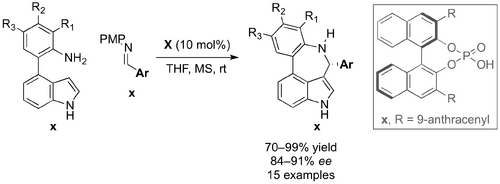
Control reactions with N–Methyl indole x gave the corresponding product in just 3% ee. This result suggested that the indole NH may play an important role in the transition state determining enantioselectivity. Taken together with the postulated importance of the H2NPMP leaving group, Tian proposed a transition complex where the cyclisation precursor x, catalyst x and trans-imination byproduct x are organised through hydrogen bonding, resulting in highly enantioselective cyclisation.

A noteworthy experiment showed the reluctance of 4-(2-aminophenyl)-7-azaindole x to react with PMP protected imine x under analogous reaction conditions. Intriguingly, the corresponding aldehyde x did react and gave the desired product x in good yield and high ee (77 and 90% respectively). This result again highlights the importance of hydrogen-bonding in the transition state and could also suggest that changing the nucleophile from imine to aldehyde results in a different mechanistic pathway.

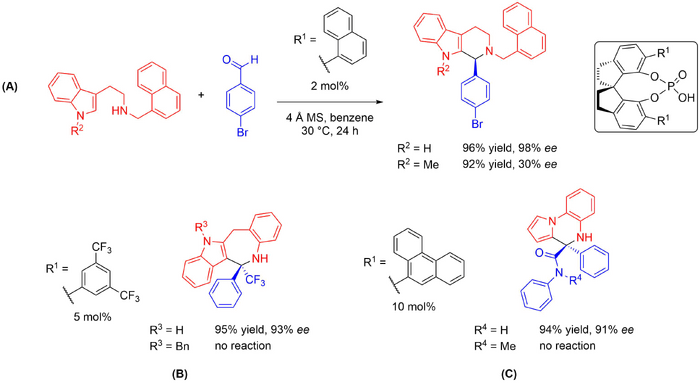
SPINOL-derived phosphoric acids have also found widespread use as catalysts for asymmetric Pictet-Spengler reactions and related reactions. These catalysts are structurally similar to BINOL-derived phosphoric acids, however, they are more rigid. In some reactions, SPINOL-derived catalysts have shown greater activity and enantioselectivity than BINOL-derived catalysts. The first example of their use in enantioselective Pictet-Spengler reactions employed N-protected tryptamines to give N-protected tetrahydro-β-carbolines, which could be further utilised in the synthesis of bioactive compounds (Scheme 2013-2015 SPINOLs (A)) (10.1002/chem.201103207). Using the reaction between N-α-naphthylmethyl tryptamine and p-bromobenzaldehyde as a model, a variety of BINOL- and SPINOL-derived catalysts was screened; the catalyst shown gave the best results. The reaction proceeded well in both polar and non-polar solvents. A range of aliphatic and aromatic aldehydes was tolerated, but it seems that an aromatic N-protecting group was required on the tryptamine. Replacing the indole hydrogen with a methyl group decreased the enantioselectivity significantly. To demonstrate the utility of the reaction, it was employed in an asymmetric total synthesis of (-)-harmicine. Similar SPINOL-phosphoric acids have since been used to catalyse an iso-Pictet-Spengler reaction between C-2-linked o-aminobenzylindoles and trifluoromethyl ketones to give benzazepinoindoles (Scheme 2013-2015 SPINOLs (B)) (10.1039/c4cc02295e), and a Pictet-Spengler-type reaction between 2-(1H-pyrrol-1-yl)anilines and α-ketoamides to give pyrrolobenzo-1,4-diazines (Scheme 2013-2015 SPINOLs (C)) (10.1002/chem/201500823). These examples indicate the broad substrate scope of this approach, and scale-up experiments were successful in both cases. Interestingly, when N-H (capable of hydrogen bonding) was replaced by N-Bn or N-Me (incapable of hydrogen bonding), the reaction did not proceed at all. The catalyst shown in (B) has also been utilised in reactions between N-allyltryptamines and 1,n-allenalaldehydes to give tetrahydro-β-carbolines bearing 1-substituted allenes (10.1021/500448j) and 1,3-substituted allenes (10.1002/chem.201500273).
Hiemstra used the enantiomer of (S)-TRIP, (R)-TRIP, as a catalyst in the synthesis of 1-substituted 1,2,3,4-tetrahydroisoquinolines from N-substituted phenylethylamines (Scheme Hiemstra 2014) (10.1021/jo501099h). The phenylethylamine starting material was essentially a dopamine derivative: the 3-hydroxy substituent activates the para-position for ring closure, while the 4-methoxy substituent protects the catechol moiety against air oxidation. The primary amine itself, and N-methyl and N-benzyl phenylethylamines gave inadequate ortho/para regioselectivity and poor enantioselectivity, but N-(o-nitrophenylsulfenyl) phenylethylamines gave very high regioselectivity (less than 5% ortho product) and improved enantioselectivity. It was suggested that this moderately electron-withdrawing substituent prevented catalysis by weakly acidic phenolic OH, and thus increased catalysis by the BINOL-derived phosphoric acid. The enantioselectivity was further increased by adding (S)-BINOL as cocatalyst, and by flowing argon over the solution at 90 °C to azeotropically remove water (other drying agents had a detrimental effect on yield and enantioselectivity). Benzaldehydes with an electron-withdrawing substituent at the para-position performed well. However, p-methoxy benzaldehyde and benzaldehyde itself gave poor enantioselectivities, as did benzaldehydes with substituents at the meta- or ortho-positions. Aliphatic aldehydes performed better, with further improvement upon addition of acetic acid as cocatalyst. The enantiomeric purity of some products was improved by recrystallisation, but yields were reduced significantly due to crystallisation of a racemate. This methodology was used to synthesise a number of natural products and potential drugs.

This approach was extended to the synthesis of 1-benzyl-1,2,3,4-tetrahydroisoquinolines (10.1021/acs.joc.5b00509). In addition to the methoxy substituent previously employed, a methoxymethyl (MOM) substituent was also employed to protect the catechol. This group is readily removed by acid, and its use allowed the preparation of isoquinolines with varying substituents at the 6- and 7-positions (Scheme Hiemstra 2015). The reaction was applicable to a variety of reactive substituted phenylacetaldehydes, but it had to be performed at room temperature to prevent decomposition. As a result, azeotropic removal of water was not possible, so magnesium sulfate was used as a drying agent instead.
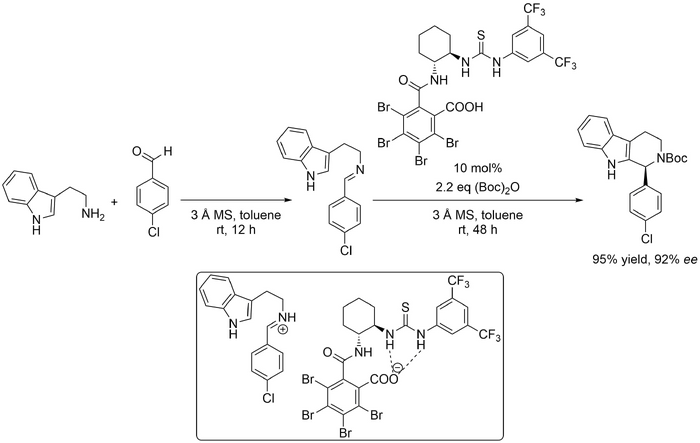
Seidel demonstrated a novel internally conjugate-base-stabilised Brønsted acid catalysis strategy which addresses one of the major challenges in Pictet-Spengler methodology -- the low reactivity of the imine or iminium ion intermediates -- to enable reactions of unmodified tryptamine (Scheme Seidel 2014) (10.1021/ol403773a). The catalyst has both a carboxylic acid group and an anion-recognition site; Seidel proposed that the carboxylic acid group protonates the initially formed imine, giving an iminium ion, while the anion-recognition site stabilises the resulting carboxylate ion via hydrogen bonding. It is thought that the cationic iminium ion and the anionic deprotonated catalyst form an ion pair (inset) in which the iminium ion is more reactive because it is not stabilised by hydrogen bonding. Using the reaction of tryptamine and p-chlorobenzaldehyde as a model reaction, catalysts with various anion-recognition sites were evaluated. A catalyst with a thiourea group gave the greatest enantioselectivity (94% ee), however, yields remained low (38%), even after extending the reaction time from 48 h to 96 h. This was likely due to product inhibition: the product is more basic than the starting materials. Achiral Brønsted acids (unable to promote the racemic reaction) were added to protonate the product and therefore reduce product inhibition. Malonic acid gave the best result, increasing the yield (>95%) without reducing the enantioselectivity significantly, but this additive was ineffective with other aldehydes. Alternatively, the products were Boc-protected in situ, which successfully reduced product inhibition. To prevent the formation of N-Boc tryptamine, tryptamine and p-chlorobenzaldehyde were allowed to form the imine before (Boc)2O was added. This strategy was applicable to benzaldehyde itself, and benzaldehydes with electron-withdrawing subsitutents at the para-position, but not to benzaldehydes with electron-donating substituents or with substituents at other positions. Aliphatic aldehydes also performed poorly.
Thiourea Organocatalysts
In 2004, Jacobsen reported his initial work on asymmetric catalysis of the acyl-Pictet-Spengler reaction using chiral thioureas. Jacobsen realised the inherent challenge of developing an asymmetric Pictet-Spengler catalyst involved low reactivity of the imine substrate. Additionally, previously reported racemic efforts had involved Lewis acid catalysts paired with highly reactive agents at high temperatures. Jacobsen enhanced the reaction by increasing the electrophilicity of the iminium intermediate through formation of the corresponding N-acyliminium ion. A number of studies on the reactivity of iminium ions have revealed that their substitution pattern accounts for their 'cationic character'. N-acyliminium ions represent some of the most reactive electrophilic systems. The electron-withdrawing group reduces the amount of electron density on the nitrogen and therefore its ability to stabilise the cation. Electrophilic addition to acyliminiums is generally fast and irreversible whereas additions to less reactive N-alkyliminiums can be reversible. Ref
Early screening experiments showed cyclization occuring at -30 °C in 59% ee. While screening individual reaction parameters, Jacobsen discovered that product chirality exhibited a strong dependence upon the structure of the acylating agent, reaction solvent, and temperature.

Under optimized conditions (-30 °C, ether, 5 mol% catalyst), Jacobsen's thiourea delivered enriched tetrahydro-B-carbolines in 65-81% yields, and up to 95% ee.
A proposed mechanism for the thiourea-catalysed enantioselective Pictet-Spengler-Type cyclization was published by Jacobsen in 2007. Interestingly, key experimental observations, supported by DFT computational analyses, pointed towards an SN1-type pathway in these cyclizations, with catalysis via a previously unprecedented anion-catalyst hydrogen bonding mechanism.
An extensive screen of acidic additives revealed that either chlorotrimethylsilane or the combination of HCl and 3 Å molecular sieves afforded high levels of conversion and enantioselectivity, but that water had a deleterious effect on catalyst activity. Furthermore, a quite significant inverse correlation between conversion and reaction concentration was observed, with reactions run at lower concentrations affording substantially improved yields.
As a direct demonstration of the applicability of this new methodology, Jacobsen applied the enantioselective hydroxylactam cyclization to the total synthesis of (+)-harmicine with the cyclization proceeding in 97% ee followed by subsequent LiAlH4 reduction affording the natural product in only four steps from tryptamine.
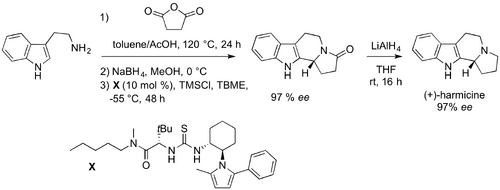
Variable temperature 1H NMR studies of reaction mixtures indicated that formal dehydration and formation of the corresponding chlorolactam is rapid and irreversible. Further observation of enhanced reactivity of alkylated versus reduced derivatives suggests that an SN2-type displacement of chloride is not operative in the cyclization reaction, which points instead to an SN1-type mechanism. Since the enantio-determining step is likely either the addition of the indole to the N-acyliminium ion (b → c or b → d), or alkyl migration of the spiroindoline intermediate (c → d), catalyst interaction with at least one of these species is required. However, there is no viable Lewis basic site for catalyst binding to substrate in c or d.
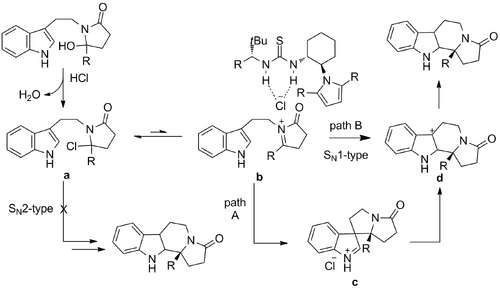
Therefore, Jacobsen proposed that the thiourea catalyst promotes enantioselective cyclization by inducing dissociation of the chloride counterion and forming a chiral N-acyliminium chloride-thiourea complex. Noticeable halide counterion effects and solvent effects on enantioselectivity lend proof to this theory. Furthermore, it was suggested that catalysis and enantioinduction may result from initial abstraction of a chloride anion from a in an SN1-type rate determining step (a → b) and subsequent cyclization mediated by the resulting anion-bound thiourea. This mode of catalytic generation of cationic intermediates was previously reported in the well-established anion-binding properties of ureas and thioureas. Further, the possibility of high levels of enantioinduction induced through counterion interactions is well precedented in chiral phase-transfer catalysis and has recently been demonstrated in the context of asymmetric counterion-directed catalysis.
In 2007, Jacobsen published a review titled “Small-Molecule H-Bond Donors in Asymmetric Catalysis” identifying chiral hydrogen-bond donors used for enantioselective synthesis. The area regarding to the PS reaction referred to previous work reported by Jacobsen. Concluding, Jacobsen stated his surprise at both phosphoric acids and thiourea derivatives being capable of mediating enantioselective transformations of prochiral iminium and N-acyliminium ion intermediates as they exist in opposite ends of the spectrum of the pKa scale of known H-bond donor catalysts.
In 2008, Jacobsen put his previously discovered enantioselective thiourea-acyl-Pictet-Spengler catalyst to use in the total synthesis of (+)-yohimbine. The synthesis was achieved in 11 steps and 14% overall yield. The absolute configuration of the molecule was established through the highly enantioselective thiourea-catalyzed acyl-Pictet-Spengler reaction at the start of the synthesis.
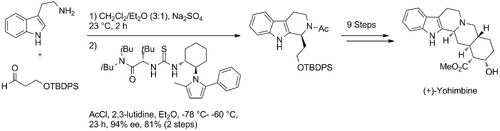
In the same year Jacobsen extended the scope of possible aromatic nucleophiles for the thiourea-catalysed ‘’N’’-acyl-Pictet-Spengler reaction to include pyrroles (Jacobsen_2008). Pyrrolohydroxylactams were shown to undergo Pictet-Spengler cyclisation in good yields using reaction conditions very similar to those employed for corresponding β-indolyl ethyl hydroxylactam condensations. Jacobsen used the same thiourea catalyst employed in the above synthesis of (+)-Harmicine (same as in Scheme: (Jacobsen 2007)). Total Synthesis of (+)-Harmicine), albeit at a loading double that required for the indole nucleophiles. For the majority of pyrrole substrates examined, very high enantioselevtivity was achieved. Further to this, Jacobsen was able to illuminate the regioselectivity of the catalyst by exploiting the fact that pyrroles possess two nucleophilic sites at C2 (or C5) and C3 (or C4). Regiocontrol was induced using the bulky protecting group, triisoproylsilyl (TIPS) attached to the pyrrole nitrogen so as to favour C4-selective cyclisations. High yields were achieved with excellent enantioselectivity reported in most cases.
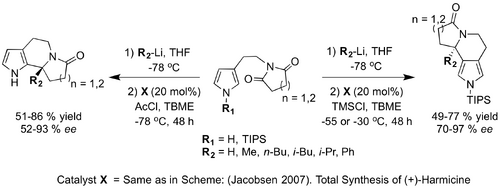
In 2009, Jacobsen published complementary research into the intermolecular addition of indoles to N-acyliminium ions. A chiral thiourea Schiff base was used to catalyse the highly enantioselective reaction of both electron-rich and electron-deficient indole nucleophiles using a catalyst loading of 5 mol % and 10 mol % respectively Jacobsen 2009 (2).
(KAB adding stuff) In the same year, Jacobsen reported asymmetric Pictet-Spengler reactions cocatalyzed by a chiral thiourea and benzoic acid. A number of optically active tetrahydro-β-carbolines were obtained in high ee. Benzoic acid was required for the reaction to proceed with aryl aldehydes. Aliphatic aldehydes did not require benzoic acid - increased ee. Less nucleophilic tryptamines, such as the 5-methoxy were unreactive under neutral conditions. Unclear why.
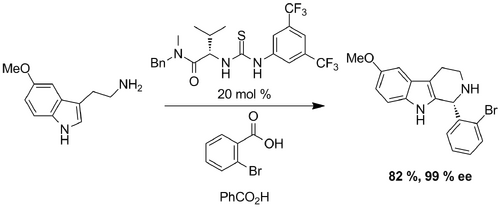
The proposed catalytic cycle describes imine protonation induced by a thiourea catalyst via H-bonding to the conjugate base of a weak Bronsted acid additive. The highly reactive protioiminium ion then cyclizes and aromatizes to generate the desired product and Bronsted acid cocatalyst. Examples also show that this thiourea catalyst promotes highly enantioselective Pictet-Spengler reactions on electronically and structurally diverse substrates.
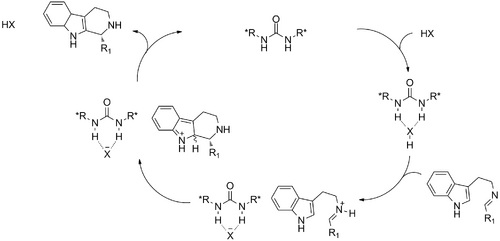
In 2011, Jacobsen, Lee, and Klausen published further work on a thiourea/benzoic acid cocatalyzed "iso-Pictet-Spengler" reactions, so named due to the alternate connectivity of the 2-substituted isotryptamine starting material. This altered substrate permits synthesis of optically pure tetrahydro-γ-carbolines. Jacobsen reports a straightforward procedure for upgrading the produced tetrahydro-γ-carbolines' enantiopurity through Boc protection of the free amine, followed by crystallization or trituration. This simple preparative step elevates the ee to >99% in nearly all published examples. The group's optimized conditions are applied to a single aromatic ketone, generating a quaternary center with high ee (98% post-trituration), albeit with reduced yield (53%, 2 steps). Jacobsen cautions that one limitation of the method is the need for a slight excess of the thiourea catalyst relative to BzOH, to avoid a deleterious racemic background reaction.
In 2010, for use in a non-Pictet-Spengler reaction, Jacobsen highlighted the correlation between an increase in the stabilising cation-π interaction abilities of a thiourea catalyst and an increase in reaction enantioselectivity. Incrementally larger arene substituents were attached to a thiourea catalyst for use in the bicyclisation of hydroxylactam Jacobsen 2010. Yield and ee increased correspondingly. However, the use of nucleophiles with aromatic systems larger than 4-pyrenyl had a negative effect on either enantioselectivity or reactivity. Whether a similar trend would be observed when applying such catalysts to the Pictet-Spengler reaction is yet to be determined.
Alternative Nucleophiles in the Catalytic Asymmetric Pictet Spengler Reaction
What is Known of the Mechanisms of Existing Systems
Terada (10.1021/ja0491533) mentions (wrt Bronsted acids) "1) Tetradentate structure around the phosphorus(V) atom would prevent free rotation at R of the phosphorus center by formation of a ring structure. This characteristic feature cannot be found in other possible Brønsted acids, such as carboxylic and sulfonic acids, etc. 2) Their appropriate acidity16 should catch up the imine through hydrogen bonding without loose ion-pair formation. 3) Their phosphoryl oxygen should function as a Lewis basic site, and thus a phosphoric acid could function as a bifunctional catalyst."
Interesting --> Cook et al, "Study of the Cis to Trans Isomerization of 1-Phenyl-2,3-disubstituted Tetrahydro-β-carbolines at C(1). Evidence for the Carbocation-Mediated Mechanism" Paper - Proposes mechanism for the racemisation via retro Pictet-Spengler of enantioenriched tetrahydro-β-carbolines synthesised from tryptamines and aldehydes.
For binaphthyl-derived phosphoric acids are there any trends in the nature of the substituents vs. ee obtained? In Hiemstra 2007 no clear trend is visible in Table 1. Franz noticed strong effect of 3,3'-substituents, with similar sterically-demanding groups reversing enantioinduction.
Limitation: avoiding β,γ-unsaturated aldehydes, which tend to tautomerise from the intermediate iminium ion to the unreactive, conjugated enamine, e.g. in Hiemstra 2011.
Dixon 2010 (10.1021/ol101651t). Both isolated enamide intermediates (epimers?) gave the same ee on treatment with the chiral BINAP (to drive rxn to completion). The proposed mechanism was that both reactions underwent rapid epimerisation through a common prochiral enamide intermediate (steady state?). Also, suggested enantioselectivity arose from facial differentiation imposed by the tight ion pair between the binol phosphoric acid conjugate base and the iminium ion.
Terada Review (10.1246/bcsj.20090268): Phosphoric acids as stronger Bronsted acids than thioureas or than TADDOL (used in the Rawal Nature paper). Considered other possible acids including sulfonic (too strong), carboxylic and sulfuric (free rotation problem), and phosphoric - just right, and chiral info is closer to proton. (When deprotonated, the O minus and P=O sites interconvert, right, but this is unimportant?) Phosphoric acids not expected to form loose ion pairs. Expected to be H-bonding etc that keeps components together. Ring system employed in the BINOL derivatives makes more rigid. Good? Mechanistic proposal in Figure 4. H-bonding network, not ion pair. Developed phosphorodiamidic acid in Synlett 2006, 133. Figure 11 has mechanistic cycle that may be of interest to PZQ. Do all the enecarbamate reactions known to function have N-H's?
Solvents: toluene found to be a good solvent for a number of these reactions, e.g. 10.1021/ja9024885. No clear trend observed in Franz 2011; DCM happens to be the best, but...
Thiourea mechanism: Franz 2011 has Jacobsen ligands as giving good conversion but moderate ee, but the Takemoto ligand giving no conversion.
Clearly the main issue with regards the mechanism is the need for an electron-rich ring for the PS reaction to occur. Franz 2011 looked at this a little, though there are two steps in the mechanism - imine formation and cyclization, so one needs to be careful interpreting results.
Strictosidine Synthase Mechanism: O'Connor 2008 compares acid-base effects of non-enzymatic aqueous solution vs. enzyme-catalysed reactions (using kinetic isotope effects). The rate-determining step appears to be the same for both systems. O'Connor also discusses the pH dependence of enzymatic catalysis, but not for binding of substrate, and proposes an enzyme mechanism involving key residue Glu309; deprotonation of tryptamine increases its nucleophilicity for aldehyde attack (emphasis on proximity of the Glu residue to the substrate). Lack of significant conformational change on binding of the substrate.
O'Connor 2010 TL - A computational model was generated for the OpSTS active site, which determined that a reversible mixture of diastereomeric intermediate carbolines were formed, but that only the 2(R)3(S) diastereomer was capable of subsequent deprotonation by key Glu309, a carboxylate residue in the active site.
With regard to the mechanisms of SPINOL-derived phosphoric acid catalysed reactions:
- Lin and Wang (10.1002/chem.201103207) propose that "the [indole] N-H bond of the tryptamine enhances the enantioselectivity...the bifunctional nature of the chiral phosphoric acid concurrently activates both the nucleophilic group and the electrophilic iminimum intermediate, generated in situ through hydrogen-bonding interactions in the Pictet-Spengler reaction".
- Lin (10.1039/c4cc02295e) again suggests that "the bifunctional nature of the chiral phosphoric acid concurrently activates both the nucleophilic group and the electrophilic group of the ketoimine intermediate through hydrogen bonding".
- Lin (10.1002/chem.201500823) presents computational studies suggesting "an unexpected attractive non-classical arene C-H···N hydrogen bond". Again, "the chiral phosphoric acid concurrently activates both the nucleophilic group and the electrophilic group of the substrate through a two-point catalyst-substrate hydrogen bonding interaction". Also, "triple hydrogen bonds hold the transition structure rigidly and allow the catalyst's 9-phenanthryl groups to influence the enantioselectivity...the directing effect of the triple hydrogen-bonding interaction is crucial in the catalyst activating the reaction and inducing chirality".
Mechanism explanations? Mechanistic Insights into a BINOL-Derived Phosphoric Acid-Catalysed Asymmetric Pictet-Spengler Reaction, L. M. Overvoorde, M. N. Grayson, Y. Luo, J. M. Goodman, J. Org. Chem 2015, 80, 2634-2640. [htt://dx.doi.org/10.1021/jo5028134 Paper]
ILT - Mar 29 2016 - Interactions between Thiourea and Imines. Prelude to Catalysis, V. de P.N. Nziko and Steve Scheiner, J. Org. Chem. 2015, 80, 10334−1034. Paper - ab initio calculations for thiourea-imine interaction
ILT - June 26 2016 - The following papers may be useful when writing up the mechanism section:
- Stöckigt 2006 10.1105/tpc.105.038018 - Crystal structure of STR1
- Tanner 2007 10.1021/bi700752n - Mechanistic studies of NCS
- O'Connor 2008 10.1021/ja077190z - STR
- Ilari 2009 10.1074/jbc.M803738200 - Mechanistic studies of NCS and comparing results to what is known of mechanism for STR
- Macone 2010 10.3390/molecules15042070 - Mechanistic studies of NCS
- Waldmann 2011 10.1002/anie.201008071 - Both STR and NCS
- Quevedo 2015 10.1007/s11164-015-1987-4 - Phenylethylamine electronics → computational calculations
- Scheiner 2015 10.1021/acs.joc.5b01985 - Interactions between Thiourea and Imines → ab initio calculations
- Abe 2015 10.1016/j.chembiol.2015.06.006 - McbB crystal structure
- Ward 2015 10.1111/febs.13208 - 'Dopamine-first' NCS mechanism
- Maresh 2016 10.1016/j.comptc.2016.02.014 - Computational study of phosphate-mediated PS reaction
Enzymatic Catalysis
(Aim of this section should be to describe preparative uses of these enzymes, and whether they are able to do reactions we can't with small molecules. Mechanistic insights (crucial here) should go in the mechanism section, to provide a comparison with what's known of small molecule systems.)
Stockigt and Waldmann's 2011 ACIEE review on the Pictet-Spengler reaction eloquently opens with discussion of the two known families of "Pictet-Spenglerases" - enzymes that take as their substrates an electron-rich aromatic, appended to an ethylamine, and an aldehyde - and transform them into asymmetric tetrahydroisoquinoline or tetrahydro-β-carboline motifs. Strictosidine synthase (STR; EC 4.3.3.2), first isolated in 1975 by Scott and Lee, cyclizes natural product precursors belonging to the strychnos and ajmaline alkaloid pathways. Norcoclaurine synthase (NCS; EC 4.2.1.78), isolated in 1981 by Nagakura from plant cell cultures, which condenses tyrosine-derived aldehydes and dopamine to form a variety of benzoisoquinoline precursors.
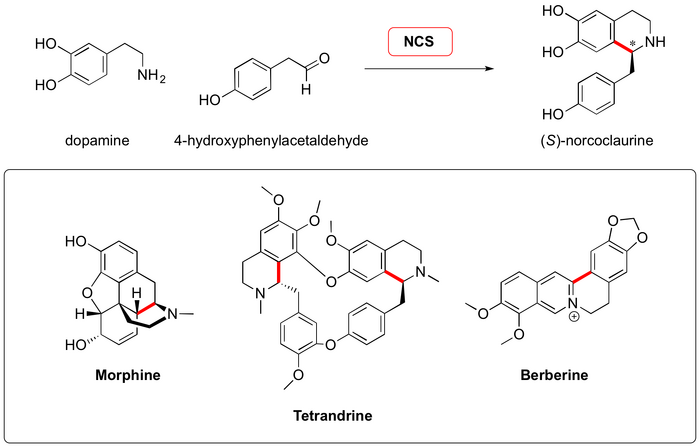
The strictosidine synthase proteins sourced from the Apocynaceae plants Rauvolfia serpentine (STR1) (Zenk, 1988 Paper) and Catharanthus roseus (CrSTSR) (McKnight, 1990 Paper) have 82 % sequence identity and are the best understood STR homologs. In contrast, the STR from Ophiorrhiza pumila (OpSTR) is from the Rubiaceae family (Saito, 2003 Paper) and shares only 54 % sequence identity with CrSTR and 60 % with STR1. These homologs can be functionally expressed in various hosts such as E. coli., Spodoptera frugiperda insect cells and several strains of yeast (Waldmann, 2011 Paper).
Of the norcoclaurine synthase proteins discovered, those from Thalictrum flavum (TfNCS) (Facchini, 2004 Paper) and Coptis japonica (CjNCS2) (Sato, 2007 Paper) have been most extensively studied. NCS belongs to the Bet v1 family and as such, does not share any sequence identity with STR1 (Stöckigt, 2006 Paper). Heterologous expression of NCS has been achieved in various strains of yeast and E. coli (Waldmann, 2011 Paper).
An alternative approach to the use of solely plant-derived cultures for the chemoenzymatic synthesis of indole alkaloids has been discovered in the use of a transgenic yeast culture of Saccharomyces cerevisiae (Verpoorte, 2001 Paper). The cDNAs of STR and strictosidine glucosidase (SGD) from Catharanthus roseus were introduced into a buffered solution of the yeast culture and left to grow for two days. Secologanin and tryptamine were then added and after a two-day incubation period, strictosidine formed in concentrations up to 2 gL-1 in the medium. Moreover, Verpoorte et al. demonstrated the effective use of Symphoricarpus albus berry extract as a more affordable, alternative source of secologanin compared to the commercially available substrate. No negative effect upon alkaloid yield was observed when the extract was used in place of pure secologanin.
In 2016 Kroutil et al. reported the two-step, catalyst-directed diastereoselective synthesis of novel C3-methylated strictosidine derivatives using ω–transaminase and STR enzymes (Kroutil, 2016 Paper). The first stereogenic centre was created during the amination of five different prochiral indolylketones employing L- and D-alanine catalysed by (S)- and (R)-selective transaminase, respectively. The (S)- and (R)-α-methyltryptamine enantiomerically pure products were subsequently condensed with secologanin in a Pictet-Spengler reaction using STRs sourced from five different plants. Of the five enzymatic catalysts employed, the STR procured from Ophiorriza pumila (OpSTR) resulted in the highest yields of the C3-methyl-substituted strictosidine derivatives (up to 97 %) with diastereomeric excesses of >98 % reported. The STR derived from Rauvolfia serpentina (RsSTR) and a mutant V208A (RvSTR) variant were also capable of catalyzing this reaction however they proved to be three times less active than OpSTR. Synthesis was conducted both simultaneously, using a one-pot cascade approach, and in a stepwise manner. For all active STRs, performing the reaction in a stepwise fashion resulted in higher yields. Kroutil hypothesised that this may be the result of transaminase catalysing the amination of secologanin when using the one-pot approach.
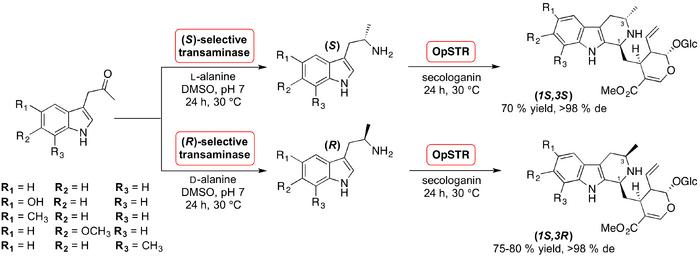
A complementary study was undertaken to establish whether the stereoselectivity of the Pictet-Spengler reaction is dependent upon the presence of the STR. An experiment was designed which compared the stereochemical outcome of the Pictet-Spengler reaction with the presence of OpSTR being the only variable. When OpSTR was used the Pictet-Spengler reaction product with an (S)-configuration at C1 formed preferentially over the other diastereomer. When the reaction proceeded in the absence of the enzyme, the (R)-diastereomer formed preferentially; thus demonstrating that the stereochemistry of the reaction is indeed catalyst-directed.
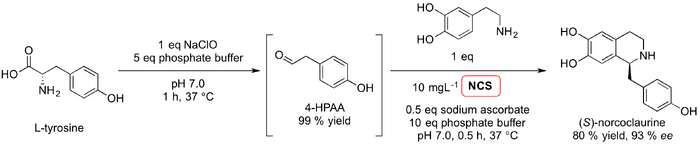
We now turn to the preparative applications of another well-documented Pictet-Spenglerase, norcoclaurine synthase (NCS). In 2010, Macone et al. published what they claimed to be the first example of a green Pictet-Spengler reaction (Paper). With the aim of producing a scalable, clean and efficient synthetic route to (S)-norcoclaurine, a one-pot chemoenzymatic approach was developed using recombinant NCS from Thalictrum flavum in aqueous conditions. Using a bacterial E. coli expression system optimised in previous work (Ilari 2008, 2009), His-tagged NCS was employed to catalyse the Pictet-Spengler condensation of dopamine with 4- hydroxyphenylacethaldehyde (4-HPAA). This afforded (S)-norcoclaurine in high yield (>80 %) and 93 % ee. The main factor limiting attempts to scale up such a synthesis is the instability of the substrates in aqueous conditions at pH 7. Dopamine readily oxidises in aqueous solutions exposed to air and 4-HPAA polymerises in both acidic and basic conditions. In order to address the problem associated with dopamine, the authors trialled a variety of modifications to the reaction conditions including de-aeration, different solvent mixtures and the addition of reductants. The addition of sodium ascorbate to the reaction mixture was found to be the most effective approach and in the presence of this reductant, dopamine concentrations increased ten-fold. Synthesis of 4-HPAA in situ in the presence of hypochlorite reduces the likelihood of polymerisation. However, the authors agreed that this unwanted side reaction still limits the potential of this chemoenzymatic synthesis.
In 2015, Hailes et al. reported the synthesis of (S)-benzylisoquinoline alkaloids (BIAs) via a one-pot, one-substrate enantioselective Pictet-Spengler reaction. A transaminase enzyme (TAm CV2025 lysate) was employed to catalyse the condensation of dopamine (2 equiv.) and pyruvate (1 equiv.) to generate 3,4 dihydroxyphenylacetaldehyde. The Pictet-Spenglerase, Δ29TfNCS also present in the reaction mixture then catalysed the condensation of dopamine with the acetaldehyde formed in situ to produce the chiral BIA, (S)-1-(3,4 dihydroxybenzyl)-1,2,3,4-tetrahydroisoquinoline-6,7-diol ((S)-norlaudanosoline; (S)-Xa) in 87 % yield and 99 % ee. As dopamine and 3,4-dihydroxyphenylacetaldehyde are in equilibrium, and TfNCS consumes an even ratio of the two substrates, this system avoids the scenario in which TAm converts all dopamine to acetaldehyde, preventing Pictet-Spengler condensation. This (S)-norlaudanosoline is synthetically valuable as it is the precursor to a variety of medically very important compounds such as morphine, codeine and berberine (Boettcher, 2005 Paper; You, 2005 Paper). In order to demonstrate the adaptability of their synthetic method, Hailes et al. repeated the reaction using 2-(3-hydroxyphenyl)ethylamine in place of dopamine. This substrate was selected based on results from their fluorescamine assay published in 2012 (Hailes, 2012, Paper which showed it to be fairly well tolerated by CjNCS2 (47 % conversion in condensation with dopamine). Using the same reaction conditions employed for dopamine, the ethylamine condensed with the 2-(3-hydroxyphenyl)acetaldehyde formed in situ to produce 1-(3-hydroxybenzyl)-1,2,3,4-tetrahydroisoquinolin-6-ol ((S)-Xb) in 56 % and and ee of 90 %. The authors suggested that the lower conversion rate compared to that for (S)-norlaudanosoline was a result of TfNCS having a greater tolerance towards dopamine as opposed to 2 (3-hydroxyphenyl)ethylamine. Hailes et al. further extended their system in the design of a three-step, one-pot chemoenzymatic cascade synthesis of two (S)-tetrahydroprotoberberine alkaloids (THPBs) which have shown promise as cancer chemotherapeutics (Iwasa, 2006 Paper). Firstly, (S)-Xa was synthesized following the above method. After 3 h, formaldehyde was added to the reaction mixture, initiating a second Pictet-Spengler condensation to produce (S)-Y and (S)-Z in a 7:1 ratio with yields of 56 % and 8 %, respectively. An ee of >95 % was reported for (S)-Y. In addition, synthesis of (S)-Xa and (S)-Y was achieved on a 0.5 mmol scale in yields of 62 % and 42 %, respectively. The ee for both products was >95 %. These results illustrate the potential for larger-scale chemoenzymatic syntheses of BIAs.

Substrate Specificity
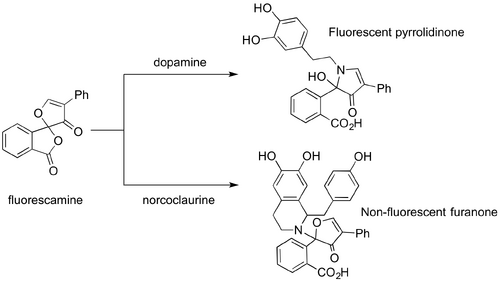
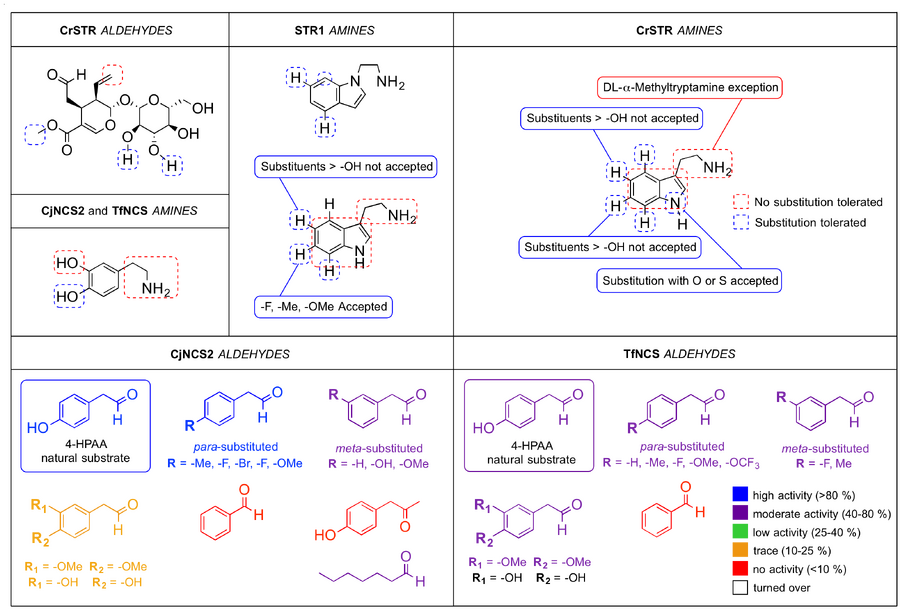
The use of enzymes to catalyse the Pictet-Spengler reaction has increased the versatility of the Pictet-Spengler reaction and allowed for access to a greater variety of asymmetric tetrahydroisoquinoline and tetrahydro-β-carboline motifs. However, the in the presence of Pictet-Spenglerases only a relatively small number of substrates show high activity. In order to address this issue and expand the substrate library, Hailes et al. developed a novel fluorescence-based high throughput assay designed to determine the tolerance of Coptis japonica norcoclaurine synthase (CjNCS2) for a range of non-natural substrates (10.1002/adsc.201200641). The assay was used to investigate the activity of nearly forty aldehyde and amine analogs in CjNCS2. The assay was comprised of fluorescamine in spirolacetone which produces highly fluorescent pyrrolidinones in the presence of primary amines, such as dopamine, and non-fluorescent furanones in the presence of secondary amines, such as norcoclaurine. In this way it was possible to quantitatively track the depletion of the primary amine substrate during the Pictet-Spengler condensation, and thus determine the substrate’s activity in the enzymatic catalyst. Hailes et al. conducted assays of numerous aryl and aliphatic acetaldehyde and aldehyde analogs with varying substitution patterns, and electron-withdrawing and donating groups. These studies revealed that para-substituted arylacetaldehydes were highly active in CjNCS2 with conversion rates >80 % for both electron-withdrawing and donating substituted analogs. In contrast, meta-substituted analogs were tolerated to a smaller extent with conversion rates near 50 %. The enzyme shows little or no tolerance for the benzaldehydes studied and 2-napthaldehyde. One ketone, 4-hydroxyphenylacetone, was assayed but showed no activity. In addition, it was demonstrated that CjNCS2 does not tolerate sterically hindered substrate analogs, such as cinnamaldehyde. Similarly, the group investigated the activity of CjNCS2 against an array of aromatic amine substrates. In their reaction with 4-HPAA, amine analogs possessing a meta-OH group were shown to be particularly well tolerated by the enzyme with conversion rates ranging from 33-81 %. However, the introduction of a hydroxyl moiety to the ethylamine functional group significantly reduced the enzyme’s tolerance of the substrate. In the absence of this meta-OH, substrates proved non-active. Similarly, electron-donating amines and halides in the meta-position were not tolerated by the enzyme.
To demonstrate the assay’s applications in chemosenzymatic synthesis, five substrate analogs identified from the assay as good candidates were used in enantioselective Pictet-Spengler reactions catalysed by CjNCS2 (see scheme X). Condensation of the natural substrates of NCS (dopamine and 4-HPAA) produced (S)-norcoclaurine in 99 % yield. Dopamine was also paired withwith (4-bromophenyl)acetaldehyde and heptanal to generate the corresponding tetrahydroisoquinolines in 77 and 56 % yields, respectively. In addition, 3-hydroxy-phenethylamine condensed with 4-HPAA in good yield (73 %). All tetrahydroisoquinolines formed with >95 % ee.
Moreover, with the aim of further broadening the substrate library, in 2014 Nishihachijo et al. investigated the potential of a variety of α-substituted and n-alkyl aldehyde analogs. Similar to the above study, NCS derived from C. japonica and expressed in an E. coli host was used. Substrate specificity was determined by observing the decrease in the amount of dopamine substrate as it underwent bioenzymatic Pictet-Spengler condensation with the aldehyde. Of the substrates studied, aldehydes that were α-substituted proved to be the least tolerated by CjNCS2 with conversion rates ranging from 2-8 %. This result is in agreement with that found by Hailes et al.. Aryl aldehydes were better tolerated than aliphatic analogs with conversion rates of 82-100 % observed for the aromatic substrates and 27-68% for the n-alkyl analogs. The activity of homobifunctional glutaraldehyde was also investigated however conversion was low at 10 %. Following these investigations Nishihachijo et al. synthesized two unnatural tetrahydroisoquinolines via the CjNCS2-catalysed enantioselective Pictet-Spengler condensation of dopamine with two aldehyde analogs selected based on the results of substrate screening. Hydrocinnamaldehyde and 1-butylaldehyde reacted with dopamine to form the corresponding 1-substituted-1,2,3,4-tetrahydroisoquinolines with high to very high yields, and in 95.3 and 98.0 % ee, respectively.
Likewise, in 2012 O’Connor et al. explored the aldehyde substrate tolerance of NCS using a very similar method. The enzyme used however, was derived from Thalictrum flavum rather than C. japonica (TfNCS). The activities of nineteen aldehydes were screened including various substituted phenylacetaldehydes, bicyclic aromatic compounds, and aliphatic open and ring analogs. The aldehyde substrate specificity of the enzyme observed agreed with the results discussed above. α-Substituted and short-chain aliphatic aldehydes were not turned over by TfNCS after 3 h and aromatic acetaldehydes were better tolerated than aliphatic analogs. One notable exception to this was the enzyme’s relatively high tolerance for 2-cyclohexylacetaldehyde whose conversion after 3 h was 71 %. The authors also explored the enzyme’s tolerance towards four unnatural phenylethylamine analogs and the natural substrate of STR1, tryptamine. The enzyme showed no activity against all the unnatural amines assayed. Interestingly, TfNCS did not tolerate tryptamine either.
In a 2006 J. Am. Chem. Soc. paper, O’Conner et al. (10.1021/ja066787w) probed the downstream effect of feeding C. roseus cultures unnatural tryptamine analogs. Several simple tryptamine derivatives were successfully incorporated into the terpene indole alkaloid metabolic pathway. However, the substrate specificity of downstream enzymes and the altered electronic and steric properties of the unnatural substrates impacted upon which biosynthetic branch the substrate followed. In 2009, O’Connor extended this research (10.1073/pnas.0903393106) through the development of a method of silencing the RNA of tryptophan decarboxylase which is directly upstream from the Pictet-Spenglerase (CrSTR) in C. roseus hairy root culture. The suppression of tryptamine and the addition of an exogenous tryptamine derivative resulted in the production of unnatural alkaloids exclusively.
In 2012, Stöckigt et al. extended the substrate scope of STR1 to include 1H-indole-1-ethanamine (Ya), heteroatom-containing derivatives (Yb,c) and a methylated derivative (Yd) (10.1021/ja211524d). This publication marks the first reported synthesis of a monoterpenoid alkaloid with a piperazino[1,2-a]indole core. The ~26 % activity of STR1 for analog Ya relative to tryptamine is the highest reported activity for any tryptamine analog tested so far. The X-ray crystal structure of Ya in complex with STR1 was also reported. This highlighted possible structural reasons why the activity of Ya is lower than it is for tryptamine. For instance, when the enzyme is in complex with tryptamine, π-π interactions with Try151 and Phe226 residues hold the substrate in place. In contrast, in the binding pocket Ya lies almost perpendicular to these residues’ π-systems and the strength of aromatic interactions is decreased. As a result, the tryptamine derivative has more room to move, causing a decrease in the rate of turnover.
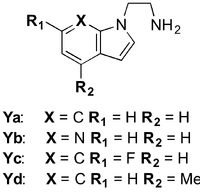
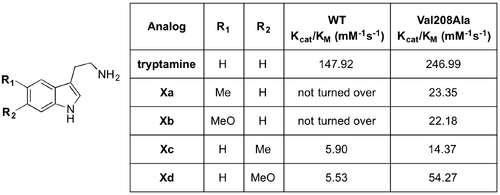
The low sequence identity of OpSTR compared to CrSTR prompted O’Connor et al. to conduct an investigation into the differences in substrate specificity of the two enzymes (10.1016/j.tetlet.2010.06.075). The sequence of OpSTR shows a four-amino acid deletion compared to the sequences of CrSTR and STR1, and near to the position where secologanin binds in the enzymatic pocket. Based on this difference, it was theorised that OpSTR may have lower aldehyde substrate specificity. Although catalytic rates were low, OpSTR turned over a variety of aliphatic and aromatic aldehydes that were not recognised by CrSTR. Both OpSTR and CrSTR accepted 2-(tetrahydro-2H-pyran-4-yl)acetaldehyde (Xa). On the other hand, the β-substituted acetaldehydes (Xb,c,d,e) were tolerated by only OpSTR. α-Substituted analogs (Xg,h,i) had a very low turnover rate with OpSTR, or were not accepted. All tetrahydro-β-carboline products were generated in >98 % ee.

Summary and comparison of substrate specificity for Pictet-Spenglerases
Here an attempt has been made to consolidate all the data to date concerning the substrate specificity of STR and NCS. Comparison has been complicated by the fact that different research groups have used different methods to determine the activity of substrate analogs. Nonetheless, some interesting general comparisons and trends have come to light. For a complete catalogue of assayed substrate analogs, see Appendix 1.
STR has very high aldehyde substrate specificity though shows much greater versatility towards derivatives of its natural amine substrate, tryptamine. Secologanin analogs that are alkyl-substituted at the vinyl position are not tolerated by CrSTR. However, the analog wherein this vinyl group is hydrogenated, is accepted. Secologanic acid and secologanin derivatives with alkyl substitution at the methyl ester are turned over by this homolog. CrSTR also tolerates analogs with methylated glucosyl hydroxyl groups (Zenk, 1979, O’Connor, 2006 BMCL). In 2009, O’Connor et al. published research demonstrating the turnover of both cis- and trans-des-vinyl aglucone O-analogs. However, it is not clear which STR homolog these secologanin derivatives were tested against (O’Connor, 2009 TL).
With regards to STR amine substrate specificity, it is evident that the indole moiety of the natural substrate, tryptamine, is essential for turnover for both STR1 and CrSTR. For example, the activity of various phenylethylamine analogs, histamine and imidazole-4-ethylamine in STR1 and CrSTR has been tested however none were accepted by either homolog. To date, there are only two exceptions to this rule. Firstly, STR1 shows high tolerance towards 1H-indole-1-ethanamine. Alkyl and heteroatom-substituted variants of this unnatural substrate are also well-tolerated (Stöckigt, 2012). Secondly, tryptamine derivatives where the indole N has been replaced by O or S are turned over by CrSTR, albeit at a much lower rate (O’Connor, 2006 BMCL).
Tryptamine analogs with α- and N-substituted amines are not accepted by STR1 or CrSTR with the exception of DL-α-methyltryptamine which shows low activity in CrSTR. In addition, methyl- and propyl-amine tryptamine derivatives are not turned over by the C. roseus homolog. 5-Substituted tryptamine analogs are turned over by STR1 and CrSTR though only with functional groups no larger than hydroxyl. CrSTR show greater activity towards 6-substituted tryptamine derivatives compared to 5-substituted analogs. However, if the substituent is larger than hydroxyl, the substrate is not turned over. In contrast, STR1 turns over –F, –Me and –OMe 6-substituted tryptamine analogs. Substitution at C4 is well-tolerated by CrSTR. Likewise, tryptamine derivatives that are C7-substituted are turned over by STR1 and CrSTR, though only if C5 is unsubstituted (Zenk, 1979, Stöckigt, 2006, O’Connor, 2006 BMCL, O’Connor, 2007 CB, Stöckigt, 2007, Stöckigt, 2012).
In contrast to STR, both CjNCS2 and TfNCS show high amine substrate specificity and are more versatile towards unnatural aldehyde substrates. For both NCS homologs, a meta–OH is essential for turnover though a para–OH is not required. In addition to this, meta-OH containing analogs with a methyl- rather than an ethylamine are not accepted (Tanner, 2007, O’Connor, 2012 TL, Hailes, 2012 ASC).
Concerning aldehyde specificity, CjNCS2 shows a greater tolerance towards the natural NCS substrate, 4-HPAA, compared to the T. flavum homolog. Para-substituted 4-HPAA derivatives show higher activity than meta-substituted analogs when assayed against CjNCS2. TfNCS, on the other hand, tolerates ortho-, meta- and para¬-substituted analogs to a similar extent. Analogs that are both meta- and para-substituted show only very low conversion with CjNCS2, though show moderate activity against TfNCS. α-Substituted aldehydes show little activity or are not turned over by both NCS homologs. Aliphatic aldehydes are accepted by CjNCS2 but show lower activity than arylacetaldehydes. The only ketone that has been assayed against CjNCS2, 4-hydroxyphenylacetone, was not turned over (Tanner, 2007, O’Connor, 2012 TL, Hailes, 2012 ASC, Nishihachijo, 2014).
Rational Reengineering of Pictet-Spenglerases
In 2006, Stöckigt et al. (10.1105/tpc.105.038018)published the crystal structure of STR1 and the structure of the enzyme with its natural substrates bound in the catalytic pocket. The protein revealed itself to possess a six-bladed four-stranded β-propeller fold with a predominantly hydrophobic binding site. Mutagenesis studies of STR1 demonstrated that the catalytic residue, Glu309 is essential for catalytic turnover. In CrSTR, Glu309 corresponds to the residue Glu315 (O’Connor 2007, 10.1016/j.chembiol.2007.07.008).
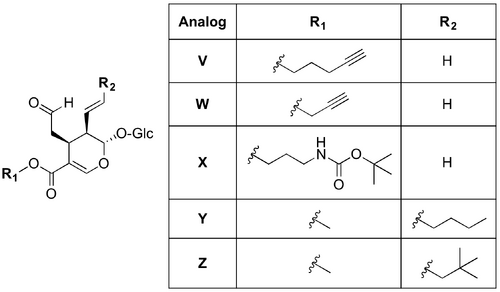
This discovery illuminated for the first time the chemistry of the catalytic pocket of a Pictet-Spenglerase. Consequently, it opened the way for research which aimed to alter the enzyme’s substrate specificity through rational reengineering of its active site. In November 2006, O’Connor (10.1016/j.chembiol.2006.10.009) published the results of their work aimed at expanding the substrate library of STR through the development of CrSTR mutants capable of turning over secologanin analogs. Sequence similarities across STR homologs and information illuminated by the crystal structure of STR1 directed the researchers’ redesign of the catalytic pocket. The residues Asp177 and Trp153 were selected for mutation as they are both located near the methyl ester of secologanin when it is bound in the protein. Competitive substrate HPLC assays using tryptamine and an equimolar mixture of secologanin and secologanin derivative revealed that the D177A mutant shows preference for secologanin analogs substituted at the methyl ester position. Long aliphatic substituents were favoured over shorter hydrophobic and bulky hydrophilic substituents. For example, the D177A mutant conversion rate for secologanin compared to the pentynyl secologanin analog (V) was 0.66:1 whereas for the propargyl analog (W) and the tert-butyl butylcarbamate (X) the ratios were 46:1 and 1.7:1, respectively. Mutating W153 to phenylalanine, histidine or glycine lead to an increased preference for secologanin relative to the secologanin analogs assayed. The n-butyl (Y) and neopentane (Z) vinyl-substituted secologanin analogs were not turned over by any of the CrSTR mutants tested. Spectroscopic and HPLC analysis of the (S)-strictosidine analog produced via the D177A-catalysed Pictet-Spengler condensation of tryptamine and secologanin analog V, and of the diastereomer (R)-vincoside confirmed that mutation of the enzyme did not affect the stereoselectivity of the enzyme.
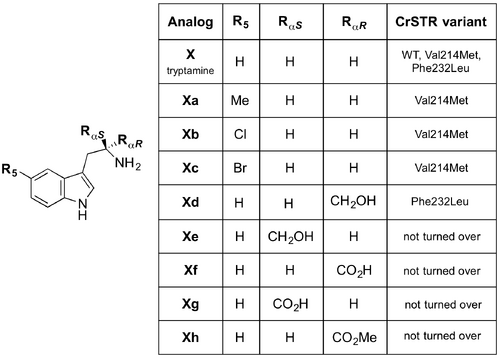
The following year, O’Connor et al. (10.1016/j.chembiol.2007.07.008) developed a Saccharomyces cerevisiae BJ5465 yeast expression system and a colorimetric assay to identify CrSTR mutants that tolerate tryptamine-derived substrates not accepted by the wild-type protein. In this way, they were able to synthesise unnatural isolable strictosidine analogs that were then taken up by plant cell cultures downstream to form unnatural monoterpene indole alkaloids. In most cases, tryptamine derivatives substituted at C5 and Cα are not turned over by CrSTR. In the enzyme binding pocket the Val214 residue is near to C5 of tryptamine. Substitution of this valine residue with methionine altered the substrate specificity such that 5-methyl- (Xa), 5-chloro- (Xb) and 5-bromo-tryptamine (Xc) —which are not tolerated by the WT enzyme— were accepted by the mutant. Following promising assay studies, Phe232, which is close to Cα of tryptamine in the enzymatic pocket, was substituted with leucine. The Phe232Leu mutant turned over (2R)-tryptophanol (Xd). However it did not accept (2R)-tryptophan (Xf) and (2R)-tryptophan methyl ester (Xh). It was hypothesised that as Phe232 is so close to the amine moiety, the bulkier substituents of Xf and Xh may prevent turnover. Regarding the tryptamine analogs, (2S)-tryptophanol (Xe) and (2S)-tryptophan (Xg), it is thought that the proximity of Glu315 prevents these analogs from reacting. Mutagenesis at this residue is not possible as it plays an essential role in the catalytic mechanism.
In 2010, an extension of this work was published by the same group (10.1038/nature09524) in which C5- and C7-halogented tryptamine analogs were integrated into the biosynthetic pathway of C. roseus hairy root culture. The group transferred the genes encoding tryptamine halogenase RebH from soil bacteria into C. roseus hairy root culture and coupled this with a modified STR enzyme, STRvm, capable of recognizing and turning over halogenated Trp precursors. The group used MS and 2D NMR methods to confirm detection of downstream halogenated alkaloids.
Similar metabolic reengineering has also been conducted with STR1. In 2007, Stöckigt et al. (10.1016/j.chembiol.2007.08.009) published the results of their findings concerning the rational redesign of the hydrophobic residue Val208. In the enzymatic pocket, Val208 is positioned near to C5 of tryptamine. Paralleling the effect of the Val214 residue in the binding pocket of CrSTR, Val208 prevents substrates with bulky substituents at C5 from being accepted by STR1. Thus, Stöckigt et al. substituted Val208 for the smaller residue, alanine. As expected, the mutant Val208Ala was able to turn over 5-methyl- (Xa) and 5-methoxytryptamine (Xb) which are not accepted by the wild-type enzyme. In addition, Val208Ala turned over 6-methyl- (Xc) and 6-methoxytryptamine (Xd) with greater efficiency compared to wild-type STR1. Increasing the activity and variety of accepted tryptamine analogs substituted at C5 and C6 is of great importance in drug design as many highly bioactive downstream alkaloids, such as vinblastine and quinine, are substituted at these positions.
Expanding our Knowledge of the Pictet-Spenglerase Family
In recent years, research aimed at increasing our understanding of the Pictet-Spenglerase family has resulted in the discovery of several enzymes that show great potential for use in the synthesis of isoquinoline and indole alkaloids. Although they are not as well understood as STR1 and NSC, it is hoped that research in this field over the coming years will lead to an increase in the versatility of biocatalytic enantioselective Pictet-Spengler reactions and a broader substrate library.
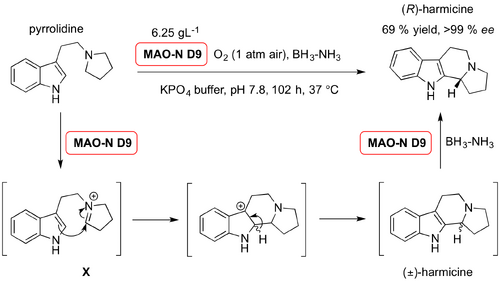
For example, in 2013, Turner et al. demonstrated the potential of a monoamine oxidase (EC 1.4.3.4) variant from Aspergillus niger (MAO-N D9) as a catalyst in asymmetric oxidative Pictet-Spengler condensation. Although the condensation resulted in a racemic mixture, the enzyme mediated deracemisation in situ, to leave only one enantiomer in very high ee. The authors demonstrated that, in the presence oxygen and MAO-N D9 in aqueous conditions, pyrrolidine oxidized to form a reactive imminium intermediate ion (X). Subsequent cyclisation yielded harmicine as a racemate. The reductant, ammonia borane was added to the reaction mixture after 96 h, and successive cycles of oxidation and reduction provided a single enantiomer of (R)-harmicine in moderate yield (69 %) and >99 % ee.
Similarly, in the same year Ju et al. reported the discovery of a novel Pictet-Spenglerase, McbB (Ju, 2013, Source), sourced from the actinomycete, Marinactinospora thermotolerans SCSIO 00652 from in the South China Sea (Ju, 2011, Source). McbB shows only 10 % identity to STR from Desulfatibacillum alkenivorans (Zhang, 2014, Source). Despite this, it was found that McbB catalyses a cascade reaction consisting of a Pictet-Spengler condensation, a decarboxylation and finally an oxidation to yield the marinacarboline scaffold 1-acetyl-3-carboxy-β-carboline (X) as the major product, and the minor metabolite, 1-acetyl-β-carboline and 1-acetyl-3-hydroxy-β-carboline (Scheme Ju, 2013). Using feeding experiments with 13C-labeled acetate and 5-fluoro-tryptophan, Ju and his coworkers established that McbB condenses two acetate units and one tryptophan to form X. These studies also suggested that oxaloacetaldehyde from oxaloacetic acid is the natural aldehyde precursor of this reaction.

Concerning the catalytic mechanism of McbB, Ju et al. demonstrated that presence of the conserved amino acid residue Glu97 is essential for catalytic turnover. The researchers highlighted that a conserved catalytic residue, Glu309, also plays an essential role in the catalytic mechanism of STR1 (Stöckigt, 2006, Source) which suggested to them that STR1 and McbB may operate via a similar catalytic mechanism.
The validity of this comparison was confirmed when in 2015, Abe et al. (10.1016/j.chembiol.2015.06.006) reported that they had solved the X-ray crystal structure of L-tryptophan bound in M. thermotolerans McbB. The protein was shown to possess a very different structure compared to that of STR1. McbB exhibits a novel fold comprised of a closed β barrel and long α helices wherein the enzymatic pocket is created by the dimerization of two protein monomers. Importantly, it was found that McbB complexes to L-tryptophan in a manner paralleling the binding of tryptamine in STR1. Various L-tryptophan substrate analogs were assayed against the protein and, again, as with STR1, the architecture of the catalytic site prevents the turnover of analogs with bulky groups at C5 of the indole. It should be noted that the other enantiomer of the enzyme’s natural amine substrate is not tolerated by McbB, nor is tryptamine, the natural substrate of STR. In comparison, the enzyme accepts a greater variety of aldehyde substrates such as formaldehyde, methylglyoxal and isobutyraldehyde. However, aldehyde substrates larger than oxaloacetaldehyde, the enzymes natural aldehyde substrate, are not turned over by McbB.
Although the enantioselectivity of this enzyme has not been measured yet, because STR and NCS exhibit such high substrate specificity, the discovery of a novel Pictet-Spenglerase capable of accessing a different range of β-carboline alkaloids is worthy of note.
MHT note 27/4/17: to fold in: 2017 paper from Hailes et al.
Miscellaneous Related Systems, or Known Catalysts not yet used for the PS
This is a very important section where we describe some obvious things that can be tried next in the field. Reviews are not proposals, but making maps gives you a clear sense of what has not yet been explored.
Non-BINOL derived Bronsted acids (other relevant, perhaps tartrate-derived, biologically relevant?)
Internal Lewis-acid assisted urea catalysis (see: Mattson, OL 2011 and 2012)
Enzyme mutagenesis to accept varied heterocyclic precursors (thiophenes, furans, pyrroles), extension of Stockigt 2012 work
"Interrupted" Pictet-Spengler reactions, perhaps to start cascade synthesis
Merge Denmark and Leighton work, see if "base-assisted silicon catalysis" might turn over the PS
Lewis acids for Friedel-Crafts-type reactions: http://www.sciencedirect.com/science/article/pii/S0040402009009302
PS of non-activated phenethylamine with aldehydes to give THIQs: One-step preparation of 1-substituted tetrahydroisoquinolines via the Pictet–Spengler reaction using zeolite catalysts, Adrienn Hegedus and Zoltan Hell, TetLett 2004 45, 8553–8555. DOI: 10.1016/j.tetlet.2004.09.097 Paper
PS of non-activated phenethylamine to give THIQ (clay): Pictet-Spengler condensation reactions catalyzed by a recyclable H+-montmorillonite as a heterogeneous Brønsted acid, DOI: 10.1007/s11426-010-0073-4
Achiral LA-catalysed PS to give THIQs: Catalytic Pictet-Spengler reactions using Yb(OTf)3, Manabe, K., D. Nobutou, et al. Bioorganic & Medicinal Chemistry 2005 13(17): 5154-5158. DOI: 10.1016/j.bmc.2005.05.018 Paper
- Development of the Pictet-Spengler Reaction Catalyzed by AuCl3/AgOTf, S. W. Youn, The Journal of Organic Chemistry 2006 71(6), 2521-2523. DOI: 10.1021/jo0524775 Paper
- Calcium-Promoted Pictet-Spengler Reactions of Ketones and Aldehydes, M. J. V. Eynden, K. Kunchithapatham, J. P. Stambuli, Journal of Organic Chemistry 2010, 75, 8542. DOI: 10.1021/jo1019283 Paper
Conclusions, and what's needed in this field
We will write this section last. Conclusions to address advantages and disadvantages of known systems, wrt temp, recoverability, ee, substrate scope.
Currently no examples of catalytic, asymmetric oxa-PS reaction? Review of oxo-PS is Larghi 2011, and has no cat. enantioselective examples
There might be, but if so, they're covered under different names...like Prins cyclizations, Friedel-Crafts, or anomeric (sugar) arylation. It might require a lot of digging! - MAT
An (S)-BINOL-phosphoric acid was employed to catalyse an asymmetric oxa-Pictet-Spengler reaction as part of an iridium(III)-catalysed isomerisation/Brønsted acid catalysed Prins-type cyclisation sequence. It was suggested that the acid protonates the enol ether substrate to generate an oxocarbenium ion intermediate, which then forms an ion pair with the chiral conjugate base and undergoes an oxa-Pictet-Spengler reaction. Pyran-fused indoles were obtained in up to 75% yield with 60% ee. (A Tandem Isomerization/Prins strategy: Iridium(III)/Bronsted Acid Cooperative Catalysis, V. M. Lombardo, C. D. Thomas and K. A. Scheidt, Angew. Chem. Int. Ed. 2013, 52, 12910-12914. Paper)
Design of "mini-proteins" (think Marcey Waters, Manfred Reetz, or Scott Miller) to accomplish PS reaction in non-biological environment
Substrate limitations: all known examples are based on tryptamine. No examples with original PS system of an activated benzene ring. No examples with other Ar rings such as furans, thiophenes? One with pyrroles (Jacobsen). One iso-PS reaction (Jacobsen). Recent example of cat asymm on ISQ ring system - first reported: Monitoring On-Chip Pictet-Spengler Reactions by Integrated Analytical Separation and Label-Free Time-Resolved Fluorescence, Ohla Stefan; Beyreiss Reinhild; Fritzsche Stefanie; et al. Source: CHEMISTRY-A EUROPEAN JOURNAL 2012, Volume: 18, 1240-1246 DOI: paper
No examples yet of ketones tolerated by Pictet-Spenglerases.
Appendices
Appendix 1
References
Papers discussed in the review should ONLY be listed here when the summary of the science in the review is complete. The papers may be found in full at the Mendeley page)
Introduction:
- Ueber die Bildung von Isochinolin-derivaten durch Einwirkung von Methylal auf Phenyl-aethylamin, Phenyl-alanin und Tyronsin, A. Pictet and T. Spengler, Ber. Dtsch. Chem. Ges. 1911, 44, 2030-2036. Paper
- Tatsui, G. J. Pharm. Soc. Jpn. 1928, 48, 92 (may be 453-459). Need to find this paper!
- The Pictet-Spengler Condensation: A New Direction for an Old Reaction, E. D. Cox and J. M. Cook, Chem. Rev. 1995, 95, 1797- 1842 Paper
- Asymmetric Synthesis of Isoquinoline Alkaloids, M. Chrzanowska and M. D. Rozwadowska, Chem. Rev. 2004, 104, 3341-3370 Paper
- The Pictet-Spengler Reaction in Nature and in Organic Chemistry, J. Stöckigt, A. P. Antonchick, F. Wu and H. Waldmann, Angew. Chem. Int. Ed. 2011, 50, 8538-8564 Paper
- The Pictet-Spengler Synthesis of Tetrahydroisoquinolines and Related Compounds, W. M. Whaley and T. R. Govindachari, Organic Reactions 1951, 6, 151-190 Paper
- Enantiospecific Total Synthesis of the Ajmaline Related Alkaloids (-)-Suaveoline, (-)-Raumacline, and (-)-Nb-Methylraumacline, X. Fu and J.M. Cook, J. Am. Chem. Soc. 1992, 114, 6910-6912 Paper
- Resolution of etodolac and anti-inflammatory and prostaglandin synthetase inhibiting properties of the enantiomers, C. A. Demerson, L. G. Humber, N. A. Abraham, G. Schilling, R. R. Martel and C. Pace-Asciak, J. Med. Chem 1983, 26, 1778-1780. Paper
- The Pictet-Spengler Reaction: Efficient Carbon-carbon Bond Forming Reaction in Heterocyclic Synthesis, S. W. Youn, Org. Prep. Proc. Int. 2006, 38, 505-591 [Paper]
- Taste, A New Incentive to Switch to (R)-Praziquantel in Schistosomiasis Treatment, T. Meyer, H. Sekljic, S. Fuchs, H. Bothe, D. Schollmeyer, C. Miculka, PLoS Negl Trop Dis 2009, 3, e357 Paper
- Spiroindolones, a Potent Compound Class for the Treatment of Malaria, M. Rottmann, C. McNamara, B. K. S. Yeung, M. C. S. Lee, B. Zou, B. Russell, P. Seitz, D. M. Plouffe, N. V. Dharia, J. Tan, S. B. Cohen, K. R. Spencer, G. E. González-Páez, S. B. Lakshminarayana, A. Goh, R. Suwanarusk, T. Jegla, E. K. Schmitt, H.-P. Beck, R. Brun, F. Nosten, L. Renia, V. Dartois, T. H. Keller, D. A. Fidock, E. A. Winzeler, T. T. Diagana, Science 2010, 329, 1175-1180 Paper
- The Total Synthesis of Enantiopure Phalarine via a Stereospecific Pictet-Spengler Reaction: Traceless Transfer of Chirality from L-tryptophane, J.J. Trzupek, D. Lee, B.M. Crowley, V.M. Marathias, S.J. Danishefsky, J. Am. Chem. Soc. 2010, 132, 8506-8512 Paper
- Synthesis of Oxacycles Employing the Oxa-Pictet-Spengler Reaction: Recent Developments and New Prospects, E. L. Larghi and T. S. Kaufman, Eur. J. Org. Chem. 2011, 5195-5231 Paper
Diastereoselective Intro Section:
- Enantiospecific Formation of Trans 1,3-Disubstituted Tetrahydro-β-carbolines by the Pictet-Spengler Reaction and Conversion of Cis Diastereomers into Their Trans Counterparts by Scission of the C-1/N-2 Bond, E. D. Cox, L. K. Hamaker, J. Li, P. Yu, K. M. Czerwinski, L. Deng, D. W. Bennett and J. M. Cook, J. Org. Chem. 1997, 62, 44-61. Paper
- The Intermolecular Pictet-Spengler Condensation with Chiral Carbonyl Derivatives in the Stereoselective Syntheses of Optically-active Isoquinoline and Indole Alkaloids, E. L. Larghi, M. Amongero, A. B. J. Bracca and T. S. Kaufman, ARKIVOC 2005 (xii) 98-153. [Link here: http://www.arkat-usa.org/arkivoc-journal/browse-arkivoc/2005/12/]
Lewis Acid Intro Section:
- A New Evidence for the Presence of a Spiroindolenium Species in the Pictet-Spengler Reaction, T. Kawate, M. Nakagawa, K. Ogata, and T. Hino, Heterocycles, 1992, 33, 801-811. [Need DOI]
- A New Chiral BLA Promoter for Asymmetric Aza Diels-Alder and Aldol-Type Reactions of Imines, K. Ishihara, M. Miyata, K. Hattori, T. Tada and H. Yamamoto, J. Am. Chem. Soc. 1994, 116, 10520–10524. Paper
- Enantioselective Asymmetric Pictet-Spengler Reaction Catalyzed by Diisopinocampheylchloroborane, T. Kawate, H. Yamada, T. Soe and M. Nakagawa, Tetrahedron: Asymmetry 1996, 7, 1249-1252 Paper
- Chiral Lewis Acid-mediated Enantioselective Pictet-Spengler Reaction of N-b-Hydroxytryptamine with Aldehydes, H. Yamada, T. Kawate, M. Matsumizu, A. Nishida, K. Yamaguchi and M. Nakagawa, J. Org. Chem. 1998, 63, 6348-6354. Paper
- Pictet-Spengler Reactions of N-b-Hydroxytryptamines and Their Application to the Synthesis of Eudistomins, T. Hino and M. Nakagawa, Heterocycles 1998, 49, 499-531 Paper
- Highly Enantioselective Pictet–Spengler Reactions with α-Ketoamide- Derived Ketimines: Access to an Unusual Class of Quaternary α-Amino Amides, F. R. Bou-Hamdan and J. L. Leighton, Angew. Chem. Int. Ed. 2009, 48, 2403-2406 Paper
- Powerful and Versatile Silicon Lewis Acids for Asymmetric Chemical Synthesis, J. L. Leighton, Aldrichimica Acta 2010, 43, 3-12 [Paper available at http://www.sigmaaldrich.com/chemistry/chemical-synthesis/learning-center/aldrichimica-acta.html]
- Direct and Highly Enantioselective Iso-Pictet Spengler Reactions with α-Ketoamides: Access to Underexplored Indole Core Structures, Heike Schönherr and James L. Leighton, Org. Lett. 2012, 14, 2610-2613 Paper
Brønsted Section:
- Enantioselective Mannich-Type Reaction Catalyzed by a Chiral Brønsted Acid, T. Akiyama, J. Itoh, K. Yokota and K. Fuchibe, Angew. Chem. Int. Ed. 2004, 43, 1566-1568. Paper
- Chiral Brønsted Acid-Catalyzed Direct Mannich Reactions via Electrophilic Activation, D. Uraguchi and M. Terada, J. Am. Chem. Soc. 2004, 126, 5356-5357. Paper
- Catalytic Asymmetric Pictet-Spengler Reaction, J. Seayad, A. M. Seayad and B. List, J. Am. Chem. Soc. 2006, 128, 1086-1087. Paper
- Catalytic Asymmetric Pictet-Spengler Reactions via Sulfenyliminium Ions, M. J. Wanner, R. N. S. van der Haas, K. R. de Cuba, J. H. van Maarseveen and H. Hiemstra, Angew. Chem. Int. Ed. 2007, 46, 7485-7487. Paper
- Enantioselective BINOL-phosphoric Acid Catalyzed Pictet-Spengler Reactions of N-benzyltryptamine, N. V. Sewgobind, M. J. Wanner, S. Ingemann, R. de Gelder, J. H. van Maarseveen and H. Hiemstra, J. Org. Chem. 2008, 73, 6405-6408. Paper
- Organocatalytic Enantioselective Total Synthesis of (-)-Arboricine, M. J. Wanner, R. N. A. Boots, B. Eradus, R. de Gelder, J. H. van Maarseveen and H. Hiemstra, Org. Lett. 2009, 11, 2579-2581. Paper
- Enantioselective Brønsted Acid-Catalyzed N-Acyliminium Cyclization Cascades, M. E. Muratore, C. A. Holloway, A. W. Pilling, R. I. Storer, G. Trevitt and D. J. Dixon, J. Am. Chem. Soc. 2009, 131, 10796-10797. [1]
- Chiral Phosphoric Acids as Versatile Catalysts for Enantioselective Carbon-Carbon Bond Forming Reactions, M. Terada, Bull. Chem. Soc. Jpn. 2010, 83, 101-119. Paper
- Direct Enantioselective Brønsted Acid Catalyzed N-Acyliminium Cyclization Cascades of Tryptamines and Ketoacids, C. A. Holloway, M. E. Muratore, R. I. Storer and D. J. Dixon, Org. Lett., 2010, 12, 4720-4723 Paper
- Total Synthesis of (+)-Yohimbine via an Enantioselective Organocatalytic Pictet-Spengler Reaction, B. Herle, M. J. Wanner, J. H. van Maarseveen and H. Hiemstra, J. Org. Chem. 2011, 76, 8907-8912. Paper
- Enantioselective Syntheses of Corynanthe Alkaloids by Chiral Brønsted Acid and Palladium Catalysis, M. J. Wanner, E. Claveau, J. H. van Maarseveen and H. Hiemstra, Chem. Eur. J. 2011, 17, 13680-13683. Paper
- An Easy Entry to Optically Active Spiroindolinones: Chiral Brønsted Acid-Catalysed Pictet-Spengler Reactions of Isatins, S. Duce, F. Pesciaioli, L. Gramigna, L. Bernardi, A. Mazzanti, A. Ricci, G. Bartoli and G. Bencivenni, Adv. Synth. Catal. 2011, 353, 860-864. Paper
- Enantioselective Pictet-Spengler Reactions of Isatins for the Synthesis of Spiroindolones, J. J. Badillo, A. Silva-Garcia, B. H. Shupe, J. C. Fettinger and A. K. Franz, Tetrahedron Lett. 2011, 52, 5550-5553. Paper
- Catalytic Stereoselective Synthesis of Diverse Oxindoles and Spirooxindoles from Isatins, J. P. MacDonald, J. J. Badillo, G. E. Arevalo, A. Silva-García and A. K. Franz, ACS Comb. Sci. 2012, 14, 285-293. Paper
- Catalytic Asymmetric Pictet–Spengler- Type Reaction for the Synthesis of Optically Active Indolo[3,4-cd][1]benzazepines, D-J. Cheng, H-B Wu, S-K. Tian, Org. Lett. 2011, 13, 5636-5639. Paper
- Highly Enantioselective Pictet-Spengler Reaction Catalyzed by SPINOL-Phosphoric Acids, D. Huang, F. Xu, X. Lin and Y. Wang, Chem. Eur. J. 2012, 18, 3148-3152. Paper
- Enantioselective synthesis of benzazepinoindoles bearing trifluoromethylated quaternary stereocenters catalyzed by chiral spirocyclic phosphoric acids, X. Li, D. Chen, H. Gu and X. Lin, Chem. Commun. 2014, 50, 7538-7541. Paper
- Triply Hydrogen-Bond-Directed Enantioselective Assembly of Pyrrolobenzo-1,4-diazine Skeletons with Quaternary Stereocenters, X. Shen, Y. Wang, T. Wu, Z. Mao and X. Lin, Chem. Eur. J. 2015, 21, 9039-9043. Paper
- Pd(0)-Catalyzed Tandem Deprotection/Cyclization of Tetrahydro-β-carbolines on Allenes: Application to the Synthesis of Indolo[2,3-ɑ]quinolizidines, Valerian Gobé and Xavier Guinchard, Org. Lett. 2014, 16, 1924-1927. Paper
- Stereoselective Synthesis of Chiral Polycyclic Indolic Architectures through Pd0-Catalysed Tandem Deprotection/Cyclization of Tetrahydro-β-carbolines on Allenes, Valérian Gobé and Xavier Guinchard, Chem. Eur. J. 2015, 21, 8511-8520. Paper
- Organocatalytic Enantioselective Pictet-Spengler Reactions for the Syntheses of 1-Substituted 1,2,3,4-Tetrahydroisoquinolines, E. Mons, M. J. Wanner, S. Ingemann, J. H. van Maarseveen and H. Hiemstra, J. Org. Chem. 2014, 79, 7380-7390. Paper
- Organocatalytic Enantioselective Pictet-Spengler Approach to Biologically Relevant 1-Benzyl-1,2,3,4-Tetrahydroisoquinoline Alkaloids, A. Ruiz-Olalla, M. A. Würdemann, M. J. Wanner, S. Ingemann, J. H. van Maarseveen and H. Hiemstra, J. Org. Chem. 2015, 80, 5125-5132. Paper
- Conjugate-Base-Stabilized Bronsted Acids: Catalytic Enantioselective Pictet-Spengler Reactions with Unmodified Tryptamine, N. Mittal, D. X. Sun and D. Seidel, Org. Lett. 2014, 16, 1012-1015. Paper
Organocatalysis Section:
- Highly Enantioselective Catalytic Acyl-Pictet-Spengler Reactions, M. S. Taylor and E. N. Jacobsen, J. Am. Chem. Soc. 2004, 126, 10558-10559. Paper
- Small-Molecule H-Bond Donors in Asymmetric Catalysis, A. G. Doyle and E. N. Jacobsen, Chem. Rev. 2007, 107, 5713-5743. Link? <-- (MHT, Jan 22) - has this paper been read and relevant mechanistic ideas incorporated?
- Enantioselective Pictet-Spengler-Type Cyclizations of Hydroxylactams: H-Bond Donor Catalysis by Anion Binding, I. T. Raheem, P. S. Thiara, E. A. Peterson and E. N. Jacobsen, J. Am. Chem. Soc. 2007, 129, 13404-13405. Paper
- Catalytic Asymmetric Total Synthesis of (+)-Yohimbine, D. J. Mergott, S. J. Zuend and E. N. Jacobsen, Org. Lett. 2008, 10, 745-748. Paper
- Regio- and Enantioselective Catalytic Cyclization of Pyrroles onto N-Acyliminium Ions, I. T. Raheem, P. S. Thiara, and E. N. Jacobsen, Org. Lett. 2008, 10, 1577-1580. Paper
- Enantioselective, Thiourea-Catalyzed Intermolecular Addition of Indoles to Cyclic N-Acyl Iminium Ions, E.A. Peterson and E.N. Jacobsen, Angew. Chem. Int. Ed. 2009, 48, 6328–633. Paper
- Weak Brønsted Acid-thiourea Co-catalysis: Enantioselective, Catalytic Protio-Pictet-Spengler Reactions, R. S. Klausen and E. N. Jacobsen, Org. Lett. 2009, 11, 887-890. Paper
- Enantioselective Thiourea-Catalyzed Cationic Polycyclizations, R.R. Knowles, S. Lin, and E.N. Jacobsen, J. Am. Chem. Soc. 2010, 132, 5030–5032. Paper
- Thiourea-Catalyzed Enantioselective Iso-Pictet-Spengler Reactions, Y. Lee, R. S. Klausen and E. N. Jacobsen, Org. Lett. 2011, 13, 5564-5567. Paper
Enzyme Section:
- Biosynthesis of the indole alkaloid. Cell-free system from Catharanthus roseus, A.I. Scott, S.-L. Lee, J. Am. Chem. Soc. 1975, 97, 6906-6908. Paper
- Purification and Properties of Strictosidine Synthase, the Key Enzyme in Indole Alkaloid Formation, J.F. Treimer, MH. Zenk, Eur. J. Biochem. 1979, 101, 225-233. Paper
- (S)-Norlaundanosoline Synthase: The first enzyme in the benzylisoquinoline biosynthetic pathway, M. Rueffer, H. El-Shagi, N. Nagakura and M.H. Zenk, FEBS Lett. 1981, 129, 5-9. Paper
- The cDNA clone for strictosidine synthase from Rauvolfia serpentina DNA sequence determination and expression in Escherichia coli, T.M. Kutchan, N. Hampp, F. Lottspeich, K. Beyreuther and M.H. Zenk, FEBS Lett. 1988, 237, 40-44. Paper
- Nucleotide sequence of a cDNA encoding the vacuolar protein strictosidine synthase from Catharanthus roseus, T.D. McKnight, C.A. Roessner, R. Devagupta, A.I. Scott and C.L.Nessler, Nucleic Acids Res. 1990, 18, 4939. Paper
- Biotransformation of tryptamine and secologanin into plant terpenoid indole alkaloids by transgenic yeast, A. Geerlings, F. Redondo, A. Contin, J. Memelink, R. van der Heijden and R. Verpoorte, Appl. Microbiol. Biotechnol. 2001, 56, 420-424. Paper
- Camptothecin Biosynthetic Genes in Hairy Roots of Ophiorrhiza pumila: Cloning, Characterization and Differential Expression in Tissues and by Stress Compounds, Y. Yamazaki, H. Sudo, M. Yamazaki, N. Aimi and K. Saito, Plant Cell Physiol. 2003, 44, 395-403. Paper
- Molecular cloning and characterization of norcoclaurine synthase, an enzyme catalyzing the first committed step in benzylisoquinoline alkaloid biosynthesis, N. Samanani, D.K. Liscombe, P.J. Facchini, Plant J. 2004, 40, 302–313. Paper
- How human neuroblastoma cells make morphine, C. Boettcher, M. Fellermeier, C. Boettcher, B. Dräger, and M.H. Zenk, Proc. Natl. Acad. Sci. U. S. A. 2005, 102, 8495-8500. Paper
- Antimicrobial Activity of Berberine Alone and in Combination with Ampicillin or Oxacillin Against Methicillin-Resistant Staphylococcus aureus, H.-H. Yu, K.-J. Kim, J.-D. Cha, H.-K. Kim, Y.-E. Lee, N.-Y. Choi, and Y.-O. You, J. Med. Food 2005, 8, 454–461. Paper
- Potential cancer chemopreventive activity of simple isoquinolines, 1-benzylisoquinolines, and protoberberines, W. Cui, K. Iwasa, H. Tokuda, A. Kashihara, Y. Mitani, T. Hasegawa, Y. Nishiyama, M. Moriyasu, H. Nishino, M. Hanaoka, C. Mukai, K. Takeda, Phytochemistry 2006, 67, 70-79. Paper
- The Structure of Rauvolfia serpentina Strictosidine Synthase Is a Novel Six-Bladed β-Propeller Fold in Plant Proteins, X, Ma, S. Panjikar, J. Koepke, E. Loris and J. Stöckigt, Plant Cell 2006, 18, 907-920. Paper
- Redesign of a Central Enzyme in Alkaloid Biosynthesis, S. Chen, M.C. Galan, C. Coltharp and S.E. O’Connor, Chem. Biol. (Oxford, U. K.) 2006, 13, 1137-1141. Paper
- Substrate specificity of strictosidine synthase, E. McCoy, M.C. Galan and S.E. O’Connor, Bioorg. Med. Chem. Lett. 2006, 16, 2475–2478. Paper
- Directed Biosynthesis of Alkaloid Analogs in the Medicinal Plant Catharanthus roseus, E. McCoy and S.E. O’Connor, J. Am. Chem. Soc. 2006, 128, 14276-14277. Paper
- Functional Analysis of Norcoclaurine Synthase in Coptis japonica, H. Minami, E. Dubouzet, K. Iwasa and F. Sato, J. Biol. Chem. 2007, 282, 6274-6282. Paper
- Rapid Identification of Enzyme Variants for Reengineered Alkaloid Biosynthesis in Periwinkle, P. Bernhardt, E. McCoy and S.E. O’Connor, Chem. Biol. (Oxford, U. K.) 2007, 14, 888–897. Paper
- Structure-Based Engineering of Strictosidine Synthase: Auxiliary for Alkaloid Libraries, E.A. Loris, S. Panjikar, M. Ruppert, L. Barleben, M Unger, H Schübel and J Stöckigt, Chem. Biol. (Oxford, U. K.) 2007, 14, 979–985. Paper
- Mechanistic Studies on Norcoclaurine Synthase of Benzylisoquinoline Alkaloid Biosynthesis: An Enzymatic Pictet-Spengler Reaction, L.Y.P. Luk, S. Bunn, D.K. Liscombe, P.J. Facchini and M.E. Tanner, Biochemistry 2007, 46, 10153-10161. Paper
- Strictosidine Synthase: Mechanism of a Pictet-Spengler Catalyzing Enzyme, J.J. Maresh, L-A. Giddings, A. Friedrich, E.A. Loris, S. Panjikar, B.L. Trout, J. Stockigt, B. Peters, S.E. O'Connor. J. Am. Chem. Soc. 2008, 130, 710-723. Paper
- Cloning, expression, crystallization and preliminary X-ray data analysis of norcoclaurine synthase from Thalictrum flavum, A. Pasquo, A. Bonamore, S. Franceschini, A. Macone, A. Boffi and A. Ilari, Acta Crystallogr., Sect. F: Struct. Biol. Cryst. Commun. 2008, 64, 281–283. Paper
- Synthesis and biochemical evaluation of des-vinyl secologanin aglycones with alternate stereochemistry, P. Bernhardt, S.E. O’Connor, Tetrahedron Lett. 2009, 50, 7118–7120. Paper
- Structural Basis of Enzymatic (S)-Norcoclaurine Biosynthesis, A. Ilari, S. Franceschini, A. Bonamore, F. Arenghi, B. Botta, A. Macone, A. Pasquo
