BioMicroCenter:BioInformaticsStaff
| HOME -- | SEQUENCING -- | LIBRARY PREP -- | HIGH-THROUGHPUT -- | COMPUTING -- | DATA MANAGEMENT -- | OTHER TECHNOLOGY |
BIOINFORMATICS SUPPORT
|
As part of our mission statement, the BioMicro Center is designed to assist users in computational challenges. One key aspect of this is providing our users with the bioinformatics support to interpret their data readily and to assist them in analyzing data for publications and grants. To accomplish this, the BioMicro Center has a team of informatics scientists on staff able to assist labs with experience in a broad number of methodologies. Bioinformatics consultations are available by appointment for CORE lab members. Bioinformatic projects are undertaken by the BioMicro Center on a collaborative basis. If you are looking for informatics support, the easiest way to begin is with an ilabs request with a brief description of your experiment. One of the members of our informatics staff will reach out to you to schedule a one on one meeting. This meeting is free for CORE lab members. Once an experimental plan is approved we will begin work on your project, checking in with you at regular intervals to be sure the project is on track. The project can be paused at any time by the researcher. Significant portions of the informatics staff salaries are paid by direct support from faculty members. As such, these "sponsors" of the informatics groups have priority access on their projects. Additional blocks of time are reserved for each informaticist to work on projects that are billed hourly with priority given to CORE labs. A small number of hours may remain to assist non-MIT labs with data analysis but such projects are always considered low priority. Labs interested in sponsoring the informatics staff at the BioMicro Center should contact Stuart Levine |
 |
AUTHORSHIP
Informatics projects through the BioMicro Center are considered collaborative efforts. Publications resulting from statistic or bioinformatics work frequently merit co-authorship: the order of authorship is not of concern. Placing BioMicro Center staff on publications significantly increases our ability to obtain financial support through grants and is helpful in the renewal process for CEHS and KI core grants.
BIOINFORMATICS STAFF
Vincent Butty, MD, PhD

Informatics Scientist
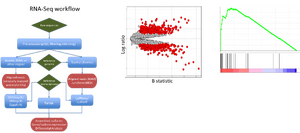
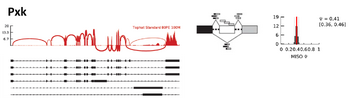
Dr. Vincent Butty joined the core in April 2012. He is an MD/PhD with a strong background in RNA sequencing methodologies and immunology. Vincent obtained his MD at the University of Geneva and received a PhD in Immunology from Harvard where he studied the population genetics of autoimmune diseases such as Type-I diabetes in the Benoist/Mathis lab. Vincent did his postdoctoral training here at MIT in Dr. Chris Burge's group, where he investigated the regulation of RNA processing across a broad variety of disease contexts using RNA sequencing. In the core, Vincent is currently involved in the analysis of long non-coding RNAs, the role of chromatin in development and the response to genotoxic and infectious stresses. Specialties
- RNAseq
- Immunology & Immunogenetics
Duan Ma, PhD

Informatics Scientist
Huiming Ding, PhD

Informatics Scientist
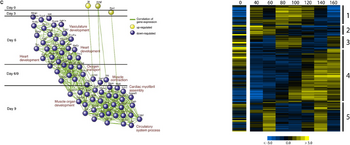
Huiming is the longest serving member of the team. He received his PhD in Physics from Jilin University in China before working for nearly a decade as a senior research associate in bioinformatics at the University of Toronto where he supported the work of Dr. Charlie Boone in studying cellular networks and pathways using synthetic genetic array (SGA) and synthetic dosage lethal (SDL) screens. Huiming has significant experience in deriving interaction networks and in creating statistical scoring algorithms and databases. His current collaborative projects involve the study of functional interaction networks from time course data and generation of genome-scale metabolic pathway models. Specialties
- Statistical Analysis
- Network Analysis
Charlie Whittaker, PhD

Core Director
Stuart Levine's primary responsibility is to direct the BioMicro Center but in a previous life Stuart was a bioinformaticist and is still available to assist on data analysis as time is available. Stuart received his BS in Biology from MIT (where he UROPed with Dr. Peter Sorger) then did his graduate work with Dr. Bob Kingston and Dr. William Forrester at Harvard Medical School where he studied the biochemical activities of the polycomb group of gene regulators. Stuart then did his post-doctoral work with Dr. Richard Young where he switched from biochemistry to bioinformatics, studying gene regulation on a genome wide scale using expression and chromatin immunoprecipitation data. Stuart has numerous publications in the areas of regulation of transcription, genomic architecture and cell fate determination.
Specialties
- Chromatin IP
- expression analysis
- Transcription mechanism