BISC209: Stains
Special Staining Techniques
Capsule Stain
A capsule is a gelatinous, slimy, carbohydrate material surrounding the bacterial cell. The capsule has many functions including, preventing dessication, aiding in biofilm formation and adherence to a surface or to another bacterium, and protecting the cell against phagocytosis which makes parasitic organisms more resistant to a host's natural defenses and free living bacterium less likely to be engulfed by a predator.
Sometimes it is helpful to determine cell morphology and cellular arrangement of a bacterium without the damage associated with heat fixing a smear. A staining technique, called negative staining, is frequently used because it outlines a bacterium against a dark background field. The bacterium will appear clear (unstained). The background dye doesn't stain the organism because its acidic chromagen is repelled by the negative charge on the bacterial cell surface.
In a capsule stain, the negative background stain is combined with a positive stain that dyes the cytoplasm of the bacterial cell. This composite stain is useful when you are trying to observe both the cell morphology and the presence of a capsule around the cell. Sometimes the desired organism is washed away by the application and rinsing of the positive dye, but when the procedure works, both the stained bacterium and the clear capsular zone are quite beautiful.
Positive stains alone can sometimes demonstrate capsules, but capsules are clearer when negative stains are employed. In
this procedure a negative stain, such as India ink, creates a dark particulate background in which the capsules show up as clear areas surrounding the purple positively stained cell. Because capsule production is sometimes dependent on many factors, staining an older culture, particularly one that is slimy, is a good idea if you believe your organism has a capsule.
Capsule Stain Procedure
(A modified Gin stain adapted from: Beishir, Lois. 1996. Microbiology in Practice. 6th edition. Harper Collins College Publishers. New York, NY. p.227-230 and Leboffe, Michael J. and Burton E. Pierce. 2002. Microbiology Laboratory Theory and Application. Morton Publishing Company. Englewood Colorado. p.84-85.)
- Clean a new slide with BonAmi cleanser to remove any film or oils that might be on the slide. You will need 1 cleaned slide for each bacterial isolate you wish to examine.
- Place a small droplet (loop) of 10% Nigrosin, 2% Congo Red, or India Ink and an equal size small droplet (loop) of water on the far right end of the cleaned slide. Mix the stain and water gently, without spreading, using a sterilized loop
- Aseptically transfer a barely visible amount of bacteria from a colony or slant culture to the mixed drop and mix gently, again, without spreading.
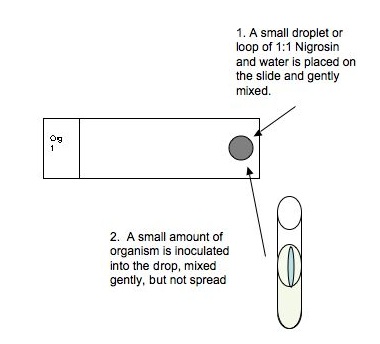
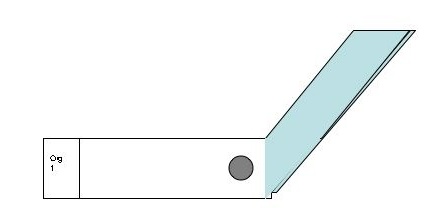
- Take a second slide (does not have to be cleaned with BonAmi), Hold the short side of the slide next to the drop at a slight angle. Push the opposite short edge of the slide through the drop to spread it along the whole length of slide in one smooth motion.
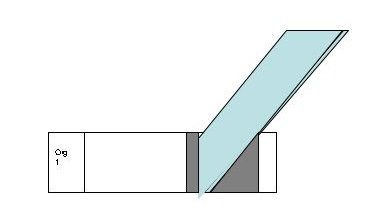
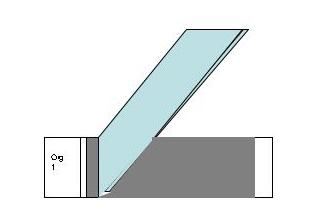
- Allow the slide to air dry. DO NOT HEAT FIX OR BLOT THIS SLIDE.
- Cover the smear with crystal violet for 1 minute.
- Drain and gently rinse the slide for a few seconds in a gentle stream of water that is directed above the smear.
- Drain the slide at a 45 degree angle with one end of the slide on a paper towel.
- Leave the slide to air dry; do not blot.
- Place a drop of oil on a section of the dried slide that is somewhat dark, but not opaque.
- Examine the slide with the microscope, looking for clear halos around a purple stained cells. Not all bacteria will have a capsule and even those that do, may not produce it when in exponential growth phase.
Materials for Capsule stain:
Any Negatively charged stain in a dropper bottle such as: 2% Congo Red, 10% Nigrosin, or freshly made aqueous undiluted India Ink (India ink is recommended by Claus, William G. 1989. Understanding Microbes. W.H. Freeman and Co. p 532. Higgins Black Magic Waterproof India Ink #4466 (Faber Castell, Corp.) Use 1:1 - 1 drop India Ink and 1 drop water inoculated with organism.
Gram's Crystal Violet
.
Endospore stain
Endospores are structures that are formed under adverse environmental conditions by certain Gram positive bacterial species such as Bacillus and Clostridium. Forming an endospore enables the organism to survive harsh environments, including lack of nutrients and moisture; thus, an endospore can remain viable for hundreds of thousands of years in dust or soil or in just about any environment. Endospores are the most resistant form of life known. Their presence in dust explains why Bacillus species are common laboratory contaminants. Their very thick spore wall is not easily penetrated by dyes, so they will appear, when Gram stained, as unstained areas within the cells. Malachite Green, when combined and heat, is driven into the spores, which then resist decolorization by acid-alcohol. Spores will will appear green when stained with an endospore stain. The ability to produce spores and the spores' location and size in the cell are characteristics used to differentiate some bacterial species.
Endospore Procedure
(Adapted from: Beishir, Lois. 1996. Microbiology in Practice. 6th edition. HarperCollins College Publishers. New York, NY. p.233-237.)
Prepare a bacterial smear slide (Find protocol at BISC209: Preparing a bacterial smear slide).

- Please only perform the spore stain during scheduled lab time. Place the slide with your smear(s) on a hot plate on the fume hood. Using a Pasteur pipet, apply enough malachite green primary stain to evenly cover all the smears.
The dyes used in this procedure are particularly messy, so be as careful as you can be, but some (or most) of us will end up with malachite green on our fingers. You can wear gloves ss long as you are careful while working with the hot plate if you are very concerned about stains on your fingers.
- Tear off a tiny piece of paper towel so it is just big enough to cover each smear area. Use forceps to place the paper towel piece over the smear so that it covers each smear and soaks up the stain. Be sure the pieces of paper towel don't overhang the slide surface.
- Apply more malachite green to the paper towel area(s) covering the smear(s) and watch for steaming of the stain (a desirable outcome). Do not allow the stain to dry out. Keep adding drops of malachite green as needed (when the smear just begins to appear to be drying). DO NOT add so much stain that it ceases to steam. Steaming is crucial!
- Watch over your slide carefully. Do not let the paper towel dry out. Keep the paper toweling moist with stain and the slide steaming hot for 3 minutes.
- Remove the slides from the heat, CAREFULLY onto a nearby paper towel using a clothes pin. Do not burn yourself. Allow the slide to cool.
- Remove the little piece(s) of paper towel from the smear and discard them in the trash (not the sink).
- Rinse the smears forcefully with water into your staining tray until the water stream runs clear and any remaining paper toweling is removed.
- Counterstain with safranin (from your Gram stain kit) for 30 seconds.
- Rinse again and blot the slide dry in Bibulous paper.
- Observe your stained smear(s) using the 1000x objective and immersion oil. USE THE DIRECTIONS in the protocols section on Care and Use of the Microscope so that you don't get oil on the other objectives.
- If spores are present, and the stain is successful, this is a beautiful stain. The spores are bright green and the vegetative cells are red.
Materials for Shaeffer-Fulton Spore stain
Malachite green stain: 5% aqueous. Dissolve 5 g of malachite green in 100 ml distilled water.
Safranin (not Gram's safranin): 0.5% aqueous. Dissolve 0.5 g safranin in 100 ml distilled water.
Flagella stain
The bacterial motility structure is the flagellum. Flagella are fragile protein structures that are too thin to be resolved by the human eye using a brightfield microscope. However, if coated with stain and mordant, the diameter increases, sometimes enough to exceed the resolution limits of the human eye. Because motility is important functionally, it is worthwhile to determine if your bacterial isolates possess flagella. Since the flagella are quite fragile and often fall off the cell during the culture or staining process (or they may not take on enough stain complex to be thick enough to view) a negative flagella stain is not conclusive of non-motility. Use this stain in conjunction with other functional tests for motility such as the hanging drop and/or SIM test.
The advantage to staining the bacterial flagella, rather than simply relying on the functional tests for motility, lies in the stain's ability to reveal number and arrangement of flagella. It is common for a motile species to have more than one per cell. Flagella arrangement is quite variable among bacterial species. The most common arrangements you might observe are peritrichous (many flagella positioned all around the bacterial cell) and polar (one or more found at one or both ends of a cell). There are flagellated bacteria in many genera including: Aeromonas, Proteus, Bacillus and others.
In the flagella stain described below, it is important to use very clean slides. Wipe new slides with 95% alcohol on a Kimwipe and let the slide dry completly before starting the stain. Pass the cleaned, dry surface of the slide through a flame to remove any remaining dust. Avoid touching the upper surface of the slide. Cool the slide and you are ready to stain.
FLAGELLA STAIN Procedure:
(adapted from: Murray, R. G. E., R. N. Doetsch, and C. F. Robinow. 1994. Light microscopy. In P. Gephardt, R. G. E. Murray, W. A.Wood, and N. R. Krieg (ed.), Methods for general molecular bacteriology. American Society for Microbiology, Washington, DC. and MelliesReed, Jay. Bacterial Flagella Stain Protocol Resource Type: ASM Curriculum: Protocol Publication Date: 9/8/2008)
1. Clean a new slide with BonAmi cleanser to remove any film or oils that might be on the slide. You will need 1 cleaned slide for each bacterial isolate you wish to examine. Dry it and run it through the flame of your Bunsen burner to remove any oils from your fingers. Cool with out touching the slide.
2. Using a flame sterilized loop, transfer a small amount of growth from an agar plate or slant cultures into 2ml water in test tube (does not need to be sterile). Mix gently and check that the suspension is only slightly cloudy. Using too much inoculum will cause difficult in visualizing the flagella. Add more water if you need to dilute the innoculum.
3. Pipet 5 μL of the culture suspension to one end of the slide using your P20 micropipet .
4. Spread the suspension gently by holding the pipet sideways over the slide and rolling the pipet tip gently over the liquid once.
5. Discard the pipet tip in the autoclave bag.
6. Air dry thoroughly. Do not heat fix.
Presque Isle Cultures flagella stain
7. Flood the smear area of the slide with Presque Isle Cultures Solution I, the mordant. Incubate at room temperature for 4 minutes.
8. Gently rinse with distilled water. Shake excess water from slide.
9. Flood with Presque Isle Cultures Solution II, the silver stain.
10.Use your clothes pin slide holder to pass the slide over, rather than in, a Bunsen burner flame by moving slide back and forth slowly, just until steam is emitted. Be careful not to overheat sample, as excess heat will destroy the flagella. Incubate at room temperature for 4 minutes.
11.Rinse in a gentle stream of distilled water. Carefully blot dry with bibulous paper or air dry.
12.View using oil immersion, at 1,000x magnification, by brightfield microscopy. Bacteria and flagella will appear golden brown. If too much stain was applied and the microscope slide was not really clean, it is difficult to find the cells and flagella.
The ingredients in the stain and mordant are proprietary reagents of Presque Isle Cultures, P.O. Box 8191, Erie, PA 16505; http://www.picultures.com
Acid-fast stain
WE WILL NOT DO THIS STAIN IN S10
The acid-fast staining procedure was developed by Paul Ehrlich in 1882. He found that the bacilli that are the causative agent in tuberculosis retain a water soluble dye reagent composed of crystal violet and aniline. These bacteria are able to retain the stain even after a wash treatment with a very strong acidified ethanol solution. Thus he named them acid-fast bacteria. Acid-fast bacteria have this resistant decolorization characteristic because of their cell wall composition. This type of cell wall contains a considerable amount of a waxlike lipid, mycolic acid, which combines tenaciously with the primary stain. Refinements in the initial Ehrlich method resulted in the currently used Ziehl-Neelsen procedure, which is better preserved than the original formulation. Only perform this stain if you are unsure if a bacterial isolate is Gram positive or negative
Acid Fast Procedure:
(Adapted from: Beishir, Lois. 1996. Microbiology in Practice. 6th edition. HarperCollins College Publishers. New York, NY. p.251-255.)
Prepare a bacterial smear slide (Find protocol at BISC209: Preparing a bacterial smear slide).

Use the staining trays and sink area. Be sure you evenly cover all the smears on the slide throughout this procedure.
The dyes used in this procedure are particularly messy, be as careful as you can be but some (or most) of us will end up with stain on our fingers.
- Cover the smear area(s) with carbolfuchsin. Add dye as needed for 5 minutes so that the smear remains covered with stain.
- GENTLY rinse the excess dye off the slide with water, try to avoid squirting the smear directly, into your staining tray. Rinse until the water stream runs clear.
- Gently tilt the slide at a 45degree angle and drip acid-alcohol decolorizer for 10 seconds.
- Rinse gently until no more red color runs off.
- Counterstain with methylene blue for 1 minute.
- Gently rinse and blot dry in Bibulous paper.
- Observe your stained slide at 1000x using the oil immersion lens.
- In this stain the acid fast organisms stain the red color of the primary stain (carbolfuchsin) and the non acid fast organism appear blue (methylene blue).
Materials:
Ziehl-Neelsen staining reagents:
Carbolfuchsin: 0.3 g basic fuchsin, 95% ethanol, 5 g phenol crystals, C.P., 95 ml distilled water. Dissolve basic fuchsin in the ethanol, In a separate container, dissolve phenol crystals in water. Mix the two solution together throughly.
Acid Alcohol decolorizer: 3ml hydrochloric acid, concentrated (37%), 97ml 95% ethanol. Add the acid to the ethanol and mix well.
Loeffler's methylene Blue: 0.3g methylene blue chloride, 30 ml 95% ethanol, 100 ml distilled water. dissolve methylene blue in the ethanol. Add the distilled water and mix well. Pour the solution through filter paper and funnel.
Links to Labs
Lab 1
Lab 2
Lab 3
Lab 4
Lab 5
Lab 6
Lab 7
Lab 8
Lab 9
Lab 10
Lab11
Lab 12