BISC209: Lab2
Lab 2: Begin Soil Microbial Communities & Diversity Project
Today you will work with your table group to sample soil from a greenhouse habitat of your choice. You will isolate DNA from that soil sample in pairs in order to begin 16S rRNA gene analysis of the bacterial community in that sample. You will also begin to culture and isolate bacteria from soil in that sample in order to compare the culturable and traditionally identifiable flora to a population identified through 16S rRNA gene sequencing.
Part A: Habitat Soil Sampling
Before going to the Greenhouse to make notes and observations and to sample the soil from the environment you and your group have decided to investigate, gather your equipment. Wash your hands. Take off your lab coat and leave it in lab.
Take with you the following equipment for each habitat (team of 4):
1-2 JMC 18in steel soil corers (looks like a metal hollow rod with the T at the top)
4 copies of the green house map downloadable from:[[1]];
2 small stainless steel garden markers to designate where you take your soil samples. Label them: BISC209 Micro Course S10 TUES (or WED) LAB - Soil Sample (A or B) Taken Here for Microbial Flora analysis- DATE)
lab notebooks and pen;
For Each Pair or Each Sampling Site in a Habitat: Make up a kit in a gallon size ZipLock bag that contains:
(1) clean spatulas or spoons,
(2) 50ml orange top sterile conical bottom plastic tubes labeled with your group's color code, initials, your lab section, the date, and the greenhouse room that you plan to sample- if there are to be two samples taken from this habitat (one per pair), distinguish your soil sample from the other taken at this site as A or B;
a black Sharpie,
several paper towels
(2) pairs of disposable gloves in the appropriate size for you and your partner
1 small sandwich bag (for collecting a few leaves from plants in your sampling area)
With your partner and your other team members, go to the greenhouse habitat you are to investigate. Before you and your partner take your soil sample (1 sample per pair -two samples per habitat), check your notes about your chosen location in the greenhouse and about your selected habitat. You should have recorded your impressions about this environment on your group or individual visit, but you may add to those notes now as you discuss the habitat with your group. Does it seem cool, warm, or cold; dry, moist, or average in humidity? Is there more sun, shade, or is it mixed? How would you describe the variety of plants in this room? What are the largest, most abundant, most interesting or most typical plants in this habitat? Today you will draw a scaled "map" in your lab notebook to show where you sampled with enough detail that someone else could easily locate the area even if the marker is removed. What else strikes you about this environment that you may want to add to your notes?
Remember that you will get more variety of bacteria from soil near plants, but it may make sampling the soil more difficult if the plants' roots are directly in the path of your soil corer. You will take two soil samples in this habitat, one sample per pair in slightly different locations. Avoid very wet soil and highly compacted soil that will be difficult to sample but get close to a plant and record it's name from the label on it.
The best area to sample in the cool temperate room (seasonal display house on the map) is around the Durant camellia. The Durant camellia is one, if not the most, prized plant in collection. This beautiful specimen blooms in winter, so it's possible that you will see it at its finest. It is over 130 years old and was donated by Mr. Henry Fowle Durant, founder of Wellesley College. In 1914, two similar plants from the original gift were lost during a fire and the remaining camellia was placed in this house in 1922. Since this plant is important to the history of Wellesley, we must be careful when sampling around it. Take your sample anywhere that is outside the camellia's "drip line" (where water dripping off the plant's leaves would hit the soil). If you sample outside the drip line you will avoid disturbing the plant's larger roots.
If you have chosen the water plant (hydrophyte) room with a warm temperate climate, there are fewer places to sample than in the other rooms since most of the plants are in pots. Probably the best site is around the bamboo over far against the far wall (don't confuse the the papyrus with the bamboo) . Ask your instructor to point out the bamboo if you don't recognize it.
In the tropical house you will have the best selection of sampling sites. Choose somewhere near a plant growing in the center circle rather than from the periphery where most of the plants are in pots.
TAKING YOUR SOIL SAMPLE:
When you and your partners have agreed on where each pair should take a soil sample, get your equipment laid out and ready. Place 2 or 3 paper towels down end to end in an open area near where you are going to sample and put on your gloves. The gloves are not sterile but be careful to avoid contaminating the exterior surface of the gloves with skin flora by touching your skin or anything other than the soil area you are going to sample.
Before you contaminate your gloves with soil, one of you should take one or two leaves from one or two plants in your sampling area. Break off older, mature, living leaves from inconspicuous places on near the bottom of the plant (without touching the leaf surface) and put the leaves in your small sandwich bag. Before you leave the greenhouse (not while you have on your gloves), write down in your lab notebook the name of the plants and mark on the map from where you took the leaf samples.
To sample the soil, brush away any leaf debris or non-soil material that might end up in your sample.
Push the soil sampling device straight down, putting force with both of your hands until it is in about 15cm (6 inches). You may use your foot (if you can). It is best to twist the corer straight into the ground. Don't go deeper because we are not culturing in anaerobic conditions. You want to sample for, primarily, aerobic or facultatively aerobic bacteria.
Empty the corer on the paper towels you made ready nearby. Knock the side of the corer and the soil should emerge as an intact cylinder of soil. If it doesn't, you may pull it out with your gloved hands. It doesn't matter if you don't get an intact cylinder. Your goal is to sample equally from this soil from the lower, middle, and upper areas of the core (avoiding the very top 30mm [1 inch] of surface soil), collecting enough soil to fill two 50ml tubes from each sampling site.
Spoon or use the spatula you brought to get the soil sample into the labeled 50ml conical tubes. If you didn't get enough soil from your core to fill both tubes, use your corer to take another sample adjacent to the first sampling site. After both 50ml tubes have been filled, discard unused soil back into the sampled area and press down. Try to make the area look undisturbed. Mark the spot sampled by the team with your labeled garden marker.
Return as quickly as you can to the lab with your labeled soil sample tubes and all of your equipment. We have a lot to do today with these samples to start our analysis of the bacteria in this soil community.
BACK IN THE LAB
In the lab, you will mix your soil sample by sieving. Wear gloves and avoid contaminating your soil with skin flora or other lab environment organisms. There will be a piece of screening material and a large beaker at your bench. Wearing fresh gloves (with an uncontaminated finger surface), place the screen over the beaker and have your partner secure it with an elastic band, avoiding contaminating the center surface of the screen with human skin flora. Pour the soil from the tubes onto the screen in small aliquots. Your goal is to sieve out the large non-soil materials. This can be expedited by pushing gently on the soil with uncontamined gloved fingers. The mixed, seived soil will be your SOIL source for weighing aliquots for some of your enrichment protocols and for making your soil extract as described below.
TAKING THE DRY WEIGHT OF SOIL
Weigh three 1 gram samples of your mixed and sieved soil into properly labeled weighing boats (label each with a piece of your team tape color on which you have identified the soil sample). Leave these on the tray marked with your lab section found beside the top-loading balances. The instructor will dry these and return them to you for use in Lab 2 for a second Streptomyces enrichment.
SOIL EXTRACT preparation
Weigh 1 gram of soil and add it to 100 ml of sterile water in a sterile 250 ml flask with magnetic stirrer (on your bench). This will become your soil extract. Place the flask on a magnetic stirrer at medium speed for at least 15 min. After you stop the stirring, let the soil settle until the larger particulate matter settles to the bottom. Not all visible particles need to settle, just the big stuff. Pour off some of the supernatant, avoiding transfering the settled particulate matter, into a new labeled sterile 50 ml conical tube. This supernatant will be your SOIL EXTRACT and this extract will be used for some of the culturable bacterial enrichment protocols you will start today in Part C. Leave the rest of your soil extract in the flask for the time being. You will discard it at the end of the lab period.
GENOMIC DNA ISOLATION preparation
To start the culture independent bacterial identification (sequencing 16s rRNA genes), each student will need an additional 0.25 gram sample of sieved soil (take this sample from whichever soil sample you helped collect). Weigh out your aliquots using the top loading balance on wieghing paper that you are careful not to contaminate. Fold up the edges of the weighing paper to use as a crude funnel and pour the 0.25g of soil into a special PowerBead tube from the MoBIO PowerSoil DNA isolation kit (these tube will be at the instructor's bench). Make sure you use the right tube and that your soil isn't too wet to start the isolation. (See the Wet Soil option of the Soil DNA isolation protocol below.) Each student will do a genomic DNA soil isolation (Part B)in one Powerbead tube, using soil from the sample she helped collect.
Save the rest of the soil you collected. You will use soil, rather than soil extract, later today in several of your culture enrichment protocols in Part C.
The most time efficient way to organize your work is to leave your soil extract stirring on the magnetic stirrer while you start the soil genomic DNA isolation described in Part B. It doesn't matter if your extract stirs longer than 15 minutes. When you and your partners are at an incubation point in the DNA isolation you can start organizing and dividing up the culture work for today described in part C that you will do as teams rather than as individuals.
Part B: Isolate Genomic DNA From Soil Sample
Protocol for Using the Power® Soil DNA Isolation Kit by Mo Bio Laboratories'
Manufacturer Information at [2]
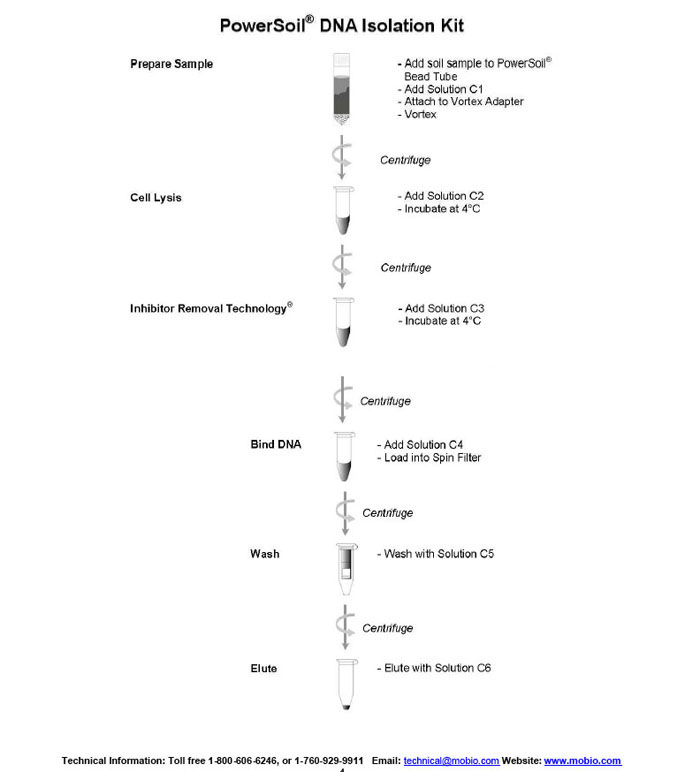
Please wear gloves during this protocol
Wet Soil Sample Option
If soil sample is dry or slightly damp you may skip this '"Wet Sample Option'" and start directly with step 1 below. If soil sample is muddy or really wet (this is unlikely), consult with your instructor. You may need to remove the contents the from PowerBead Tube (beads and solution) and transfer them into another sterile microcentrifuge tube. Add your soil sample to PowerBead Tube and centrifuge at room temperature for 30 seconds at 10,000 x g. Remove as much liquid as possible with a pipet tip. Add beads and bead solution back to PowerBead Tube and follow protocol starting at step 2.
Dry Soil Sample
1. Each student will do the DNA isolation. You will each use one 0.25 g soil sample that you weighed out in Part A. Add the soil to a PowerBead Tube and label your tube on the top only with your initials and a soil sample identifier (T-A or T-B for tropical; WT-A or WT-B for warm, wet temperate; CT-A or CT-B for cool temperate) on a piece of your team color tape.
What’s happening: After your sample has been loaded into the PowerBead Tube, the next step is a homogenization and lysis procedure. The PowerBead Tube contains a buffer that will (a) help disperse the soil particles, (b) begin to dissolve humic acids and (c) protect nucleic acids from degradation.
2. Gently vortex to mix.
What’s happening: Gentle vortexing mixes the components in the PowerBead Tube and begins to disperse the sample in the PowerBead Solution.
3. Check Solution C1 to see that it's not precipitated. If Solution C1 is precipitated, heat solution to 60C until the precipitate has dissolved before use.
What’s happening: Solution C1 contains SDS and other disruption agents required for complete cell lysis. In addition to aiding in cell lysis, SDS is an anionic detergent that breaks down fatty acids and lipids associated with the cell membrane of several organisms. If it gets cold, it will form a white precipitate in the bottle. Heating to 60C will dissolve the SDS and will not harm the SDS or the other disruption agents. Solution C1 can be used while it is still warm.
4. Add 60 microliters of Solution C1 to your PowerBead tube and invert several times or vortex briefly.
5. Give your labeled PowerBead tube to your instructor who will take all the PowerBead Tubes to FastPrep® Bead Beater. Your samples will "turbovortex" for 30-45 seconds (only 2-3 samples will fit in the BeadBeater at a time).
What’s happening: The BeadBeating step is critical for complete homogenization and cell lysis. Cells are lysed by a combination of chemical agents from steps 1-4 and mechanical shaking introduced at this step. By randomly shaking the beads in the presence of disruption agents, collision of the beads with microbial cells will cause the cells to break open.
6. Microcentrifuge your tubes at 10,000rcf for 1 minute at room temperature. Caution: Be sure not to exceed 10,000rcf or the tubes may break. Make sure the PowerBead tubes rotate freely in your centrifuge without rubbing!
7. Transfer the supernatant (don't transfer the beads!) to a clean 2 ml Collection Tube at the instructor's desk. If you don't know what a collection tube is, ask your instructor. Don't use a regular microfuge tube.
Note: Expect between 400 to 500 microliters of supernatant at this step. The exact recovered volume depends on the absorbancy of your starting material and is not critical for the procedure to be effective. The supernatant may be dark in appearance and still contain some soil particles. The presence of carry over soil or a dark color in the mixture is expected in many soil types at this step.
Subsequent steps in the protocol will remove both carry over soil and coloration of the mixture.
8. Add 250 microliters of Solution C2 to the collection tube and vortex for 5 seconds. Incubate at 4C for 5 minutes.
What’s happening: Solution C2 contains a patented reagent to precipitate non-DNA organic and inorganic material including humic substances, cell debris, and proteins. It is important to remove contaminating organic and inorganic matter that may reduce DNA purity and inhibit downstream DNA applications.
9. Centrifuge the Collection Tube at room temperature for 1 minute at 10,000rcf.
10. Avoiding the pellet, transfer up to, but no more, than 600 microliters of supernatant to a clean 2 ml Collection Tube (provided).
What’s happening: The pellet at this point contains non-DNA organic and inorganic material including humic acid, cell debris, and proteins. For the best DNA yields, and quality, avoid transferring any of the pellet.
11. Add 200 microliters of Solution C3 and vortex briefly. Incubate at 4C for 5 minutes.
What’s happening: Solution C3 is a second reagent (patented) to precipitate additional non-DNA organic and inorganic material including humic acid, cell debris, and proteins. It is important to remove contaminating organic and inorganic matter that may reduce DNA purity and inhibit downstream DNA applications.
12. Centrifuge the tube at room temperature for 1 minute at 10,000rcf.
13. Avoiding the pellet, transfer up to, but no more, than 750 microliters of supernatant to a clean 2 ml Collection Tube (provided).
What’s happening: The pellet at this point contains additional non-DNA organic and inorganic material including humic acid, cell debris, and proteins. For the best DNA yields, and quality, avoid transferring any of the pellet.
14. Shake to mix Solution C4 before use. Add 1.2 ml (do this by adding 600 microliters twice) of Solution C4 to the supernatant (be careful solution doesn’t exceed rim of tube) and vortex for 5 seconds.
What’s happening: Solution C4 has a high concentration of salts. Since DNA binds tightly to silica at high salt concentrations, this will adjust the DNA solution salt concentrations to allow binding of DNA, but not non-DNA organic and inorganic material that may still be present at low levels, to the Spin Filters.
15. Load approximately 675 microliters of the C4 + supernatant mixture from the previous step onto a Spin Filter sitting in a Collection Tube (save the remainder of the supernatant!!) and centrifuge the spin filter at 10,000rcf for 1 minute at room temperature. Discard the flow through (NOT the spin filter!!!) and put the spin filter back in the Collection Tube. Add an additional 675 microliters of the Step 14 mixture to the same Spin Filter and centrifuge at 10,000rcf for 1 minute at room temperature. Discard the flow through and load the remainder of the Step 14 mixture onto the Spin Filter in the Collection Tube and centrifuge at 10,000rcf for 1 minute at room temperature.
Note: A total of three loads for each sample processed are required. You will using the same Spin Filter and Collection Tube for all 3 spins.
What’s happening: DNA is selectively bound to the silica membrane in the Spin Filter device in the high salt solution. Contaminants pass through the filter membrane, leaving only DNA bound to the membrane.
16. Add 500 microliters of Solution C5 to the Spin Filter in the Collection Tube and centrifuge at room temperature for 30 seconds at 10,000rcf.
What’s happening: Solution C5 is an ethanol based wash solution used to further clean the DNA that is bound to the silica filter membrane in the Spin Filter. This wash solution removes residual salt, humic acid, and other contaminants while allowing the DNA to stay bound to the silica membrane.
17. Discard the flow through (not the Spin Filter) from the 2 ml Collection Tube.
What’s happening: This flow through fraction is just non-DNA organic and inorganic waste removed from the silica Spin Filter membrane by the ethanol wash solution.
18. Centrifuge the Spin Filter in the Collection Tube again at room temperature for 1 minute at 10,000rcf.
What’s happening: This second spin removes residual Solution C5 (ethanol wash solution). It is critical to remove all traces of wash solution because the ethanol in Solution C5 can interfere with many downstream DNA applications such as PCR, restriction digests, and gel electrophoresis.
19. Carefully place Spin Filter in a clean 2 ml Collection Tube (provided). DO NOT transfer any liquid that may be on the bottom of the spin filter basket and avoid splashing any Solution C5 onto the Spin Filter.
Note: It is important to avoid any traces of the ethanol based wash solution in the elution that will be created in the next step.
20. Add 100 microliters of Solution C6 to the center of the white Spin Filter membrane.
Note: Placing the Solution C6 (sterile elution buffer) in the center of the small white membrane will make sure the entire membrane is wetted. This will result in a more efficient and complete release of the DNA from the silica Spin Filter membrane. As Solution C6 (elution buffer) passes through the silica membrane, DNA that was bound in the presence of high salt is selectively released by Solution C6 (10 mM Tris) which lacks salt.
Alternatively, sterile DNA-Free PCR Grade Water may be used for DNA elution from the silica Spin Filter membrane at this step (MO BIO Catalog# 17000-10). Solution C6 contains no EDTA. If DNA degradation is a concern, Sterile TE may also be used instead of Solution C6 for elution of DNA from the Spin Filter.
21. Centrifuge the Spin Filter in its Collection Tube at room temperature for 30 seconds at 10,000rcf.
22. Discard the Spin Filter. The DNA in the collection tube is now eluted and ready for freezing or for use as a PCR template.
Storing DNA:
DNA is eluted in Solution C6 (10 mM Tris) and must be used immediately or stored at -20to -80C to prevent degradation.
Make sure your DNA is labeled and give it to your instructor for freezing. She will use a nanodroper before she freezes it to find out the concentration. DNA at 100ng/microliter is desired for the pcr. You can concentrate or dilute the DNA to 100ng/microliter before attempting to amplify the 16s rDNA or you could adjust the volume of DNA template used in the pcr reaction. Your instructor will let you know which of those alteratives is the best way to proceed.
Part C: Starting the Isolation & Study of Culturable Soil Bacteria from your habitat
Background
There are large numbers of both beneficial and non-beneficial bacteria in soil. Often their roles are not well understood. The main antibiotic producing genera of soil microbes include the Bacillus, Cephalosporium, Penicillium, and Streptomyces. Nitrogen cycling bacteria (such as Azotobacteria, Berjerinckia, Cyanobacteria, Rhizobium, Frankia, Azospirillum, Clostridium, some Klebsiella), and/or sulfur utilizing bacteria (such as Disulfovibrio), as well as other types of recyclers are equally important to the soil microbial community. Since we are limiting our focus to bacteria, we will choose media that encourage growth of bacteria and/or discourage growth of fungi and other eukaryotic microorganisms. A wide variety of growth media and incubation conditions can be used to isolate bacteria from soil. In general, we can favor the growth of certain groups over others by altering the composition (e.g. pH, osmolarity) and/or nutrients available. Some bacteria will grow so fast on rich media (nutrient agar, TSA, etc.) that they will mask other slower growing genera. We will try not only enriched nutrient and dilute nutrient agar but also a variety of defined media that will favor slower growing genera.
The vast majority of bacteria in soil (90-99%) will not grow on your plates at all. We will only find the ‘culturable’ bacteria that like the growth conditions you choose. The more types of media and growth conditions you use, the greater the variety and number of bacteria that you will find. Although most soils contain a rich and unbelievably diverse community of microorganisms, we will focus part of our investigation on a few of the culturable bacteria that contribute to this unseen world. To get a sense of the true diversity of the bacterial community in your habitat, in the other part of this two part project, we will isolate genomic soil DNA and id both culturable and unculturable bacteria by 16S rRNA gene sequencing.
Activity C-1: Plate Count of Soil Bacteria: Assessing Culturable Bacteria from Your Soil
To perform a standard plate count of your soil sample bacteria you must first serially dilute the soil so that the colony numbers will be manageable and so that you are more likely to have a countable number of cells. If you don't remember how to make a serial dilution, here is a link to a helpful animation for making dilutions http://www.wellesley.edu/Biology/Concepts/Html/serialdilutions.html.
Standard Plate Count of Soil Microorganisms done in pairs (one per soil extract)
The Soil Extract you prepared is now at a 1:100 dilution (1 gram/100 ml). This could also be called a 1% (w/v)solution.
Gather or find the following materials:
5 sterile 13 x 100 size culture tubes with caps
1 sterile disposable plastic individually wrapped 1ml pipet,
5 sterile dilute nutrient agar plates,
5 sterile plastic disposable spreaders
Setting Up a Standard Plate Count:
1. Label 5 tubes 10-3, 10-4 etc. through 10-7.
2. Label 5 destination plates of appropriate solid medium with dilution and identifying information. Because your goal is to obtain 30-300 well isolated colonies on a plate, generally only the 10-4 through 10-7 dilutions are plated. However, we are going to plate all of our dilutions. The 10 -3 dilution may be a potential source of aerobic spore forming bacteria (see the protocol for Aerobic spore forming bacteria enrichment on the Enrichment Media for Soil Bacterial from Mixed Populations protocol page of this wiki).
3. Slightly dehydrate the medium on each plate by cleaning the laminar flow hood, turning on the fan, placing the plates in the hood, and positioning the covers so they are ajar for 10 minutes or until the medium surface shows no visible moisture.
4. Pipet 0.9 ml of sterile water into the 5 tubes labeled in step 1. (You may use the same sterile 1ml pipet for all of them.)
5. Using your P200 micropipet, transfer 100μL of the 1:100 dilution to the tube labeled 10-3, mix well by vortexing.
6. Using a new tip, transfer 100 μL ml of the 10-3 dilution to the tube labeled 10-4. Mix well. Mixing 0.1ml of the 10-3 dilution with 0.9ml of sterile water makes a 10-4 dilution.
7. Continue to transfer 100μL aliquots (after mixing well) from each dilution to the next tube of water until you have carried the dilution to 10-7.
8. Use your P200 micropipet and a new tip to transfer 100μL of the 10-3 dilution and dispense it to the center of the dilute nutrient agar plate labeled 10-3. Use a sterile plastic disposable spreader to gently push the dispensed sample two or three times clockwise around the dish, and then several times counterclockwise. Make sure all of the surface area of the plate has been inoculated. Don't press too hard as force will cause the microorganisms to collect at the edge of the spreader, resulting in uneven distribution.
9. Repeat step 8 to inoculate the rest of your prelabeled dilute nutrient agar plates with the 10-4 through 10-7 dilutions of bacteria.
10. Allow the moisture to be absorbed into the agar before inverting the plates and putting a labeled piece of your team color tape around the set. Incubating the set of standard plate count plates at RT until next week in a rack designated by your instructor.
Next week you will count the number of colonies on these plates, observe the variability in colonies on each plate and, possibly, select one or more of these colonies for isolation and further study.
Calculating the number of bacteria per gram of soil
If you divide the number of colonies you find by the amount of diluent used times the dilution factor, you will obtain the number of cultivatable bacteria per gram of soil. Only plates that with between 30 and 300 colonies per plate give accurate calculations.
CFU = number counted on plate/(diluent plated*dilution of plate counted)
Activity C-2: Streaking for Isolation on enrichment media and dilute nutrient agar
How did your first attempt at streaking for isolation turn out? Have you checked your plate that you streaked in LAB 1. If you didn't get single well isolated colonies of two different bacteria on that plate, ask your instructor to watch you prepare your first streak plate today so she can offer suggestions for improving your technique.
C2a: Each student should use the 1/100 dilution of Soil Extract (the supernatant in the conical tubes) from each soil sample collected. Streak one plate of Dilute Nutrient Agar(dNA) and one plate of full strength Nutrient agar. Both Nutrient and Dilute nutrient agar will supply general nutritional needs of most bacteria and discourage fungal growth (because of the neutral pH). We are using both media to see if, in the dilute form, the growth of fungal organisms will be reduced without negatively impacting variety of bacteria. Using liquid SOIL EXTRACT as your inoculum is analogous to using a broth culture source. You will follow the steps in Streaking for Isolation in the protocol section of this wiki.
The formulation of enrichment media supplies specific nutrients that encourage the growth of bacteria types that grow too slowly or not at all in media missing these nutrients. Selective media is selective because it contains one or more ingredient(s) that inhibits the growth of competitor microbes (such as cycloheximine, a drug that prevents fungal growth but does not affect bacteria negatively). Media can be both selective and enrichment. In some cases growth on enrichment or selection media will take up to 2 weeks. Thus, you will have to keep track of the progress of your isolation of different bacterial species as not all of your organisms will be ready for the same steps at the same time.
C2b: There are 11 groups of bacteria described in the Isolation and Enrichment Media for Soil Bacterial from Mixed Populations in the protocol section of this wiki. Your goal today is to consult with your partner and the other members of your team and divide up the available media so that each soil extract from your habitat gets plated onto as many different enrichment media as possible. We want to find culturable bacteria from all 11 of the enrichment groups, if possible.
We hope that, eventually, in the weeks to come, each student will be working with a unique subset of the 11 possible bacterial groups. Make sure, when you divide up the work, that all bacterial groups are covered by at least one of the soil samples from your habitat. (You can set up both samples for each enrichment if you and partners feel really ambitious, but, at minimum, both soil extracts from each habitat should be enriched for Actinomyces and all enrichment groups should be covered by one sample from each habitat.) Notice that the enrichment protocols are divided with subheadings based on whether you use soil directly or soil extract as the inoculum. Distribute the options so each group member tries a variety of methods.
You are beginning an investigative project that is, increasingly, uniquely your groups'. Although the culture-independent molecular techniques will be done by all of you at the same time, a lot of the work that you will do each week on characterizing your culturable bacteria depends on the unique properties and metabolic capabilities of the bacteria you choose to isolate. The goal is for your group to culture and characterize a diverse population of bacteria from a defined habitat. You will present your findings in a poster presentation to the class at the end of the semester. This presentation will be more interesting if you and your group members work together to cover as wide as possible a selection and enrichment strategy now and in the next few weeks.
Come to the lab and check on your cultures often over the next few days. Make observations about the number, size, color and shape of the various colonies that appear and draw the growth you observe in your lab notebook. There is a data table in the form of an Excel spread sheet that you can download from the Data folder in your First Class Lab conference that will help you keep track of the characteristics of each of your isolates and allow you to share your data with your team.
This project is an investigative one that will require outside of lab time. Because bacteria have widely varying generation times, they will form colonies at different rates. It is your responsibility to check your cultures often and subculture or move them to your lab section's designated rack in the walk-in cold room to alt growth before the isolated colonies we seek become a mess of overgrown lawn growth on your plates and the isolation must be started all over.
These enrichment protocols vary in the length of time between steps. We can't make this become regular once a week work to fit our lab schedule. You will need to be highly organized and remember when you need to come to lab to do the next part of the isolation or characterization tests. Fortunately, much of what you will need to do outside of lab time is not time consuming. Usually, it will amount to taking a well-isolated colony and subculturing it onto new media (a few minutes of work); however, your lab instructor can't keep track of all the isolations in progress and remind you that it is time for the next step. The success of this project depends on your organizational skills and your commitment to time-sensitive attention to the task at hand.
CLEAN UP
1. All culture plates that you are finished with should be discarded in the big orange autoclave bag near the sink next to the instructor table. Ask your instructor whether or not to save stock cultures and plates with organisms that are provided.
2. Culture plates, stocks, etc. that you are not finished with should be labeled on a piece of your your team color tape. Place the labeled cultures in your lab section's designated area in the incubator, the walk-in cold room, or at room temp. in a labeled rack. If you have a stack of plates, wrap a piece of your team color tape around the whole stack.
3. Remove tape from all liquid cultures in glass tubes. Then place the glass tubes with caps in racks by the sink near the instructor's table. Do not discard the contents of the tubes.
4. Glass slides or disposable glass tubes can be discarded in the glass disposal box.
5. Make sure all contaminated, plastic, disposable, serologic pipets and used contaminated micropipet tips are in the small orange autoclave bag sitting in the plastic container on your bench.
6. If you used the microscope, clean the lenses of the microscope with lens paper, being very careful NOT to get oil residue on any of the objectives other than the oil immersion 100x objective. Move the lowest power objective into the locked viewing position, turn off the light source, wind the power cord, and cover the microscope with its dust cover before replacing the microscope in the cabinet.
7. If you used it, rinse your staining tray and leave it upside down on paper towels next to your sink.
8. Turn off the gas and remove the tube from the nozzle. Place your bunsen burner and tube in your large drawer.
9. Place all your equipment (loop, striker, sharpie, etc) including your microfuge rack, your micropipets and your micropipet tips in your small or large drawer.
10. Move your notebook and lab manual so that you can disinfect your bench thoroughly.
11. Take off your lab coat and store it in the blue cabinet with your microscope.
12. Wash your hands.
13. See you next time!
Assignment
Turn in at the beginning of lab next week, a summary of the relevant morphologic (shape, arrangement, Gram stain or other appropriate stain, motility, etc.) and useful metabolic characteristics (antibiotic producers, cellulose or nitrogen recyclers, etc.) of 1 or more desired or expected soil bacterial genera from each of the 11 groups that you are attempting to find in your habitat. (These 11 bacterial groups are listed in purple font in the BISC209: Enrichment section.). This summary can take the form of a table or a narrative organized into sections. If you don't think you will find any bacteria of a certain group in your particular habitat, list some genera included in that group and explain, briefly, why they are not likely to be found in your soil community. You can find a lot of this information in the e-book version of The Prokaryotes or Bergey's Manuals. Links to the Wellesley Library's e-copies of these Reference books is found in the Resources section of this wiki and in the Enrichment section in Protocols.
Make sure you have read all of LAB 3 carefully before coming to lab next time. In your lab notebook (not to turn in), organize, outline or make flow diagrams of all the procedures that you will perform in Lab 3, including a clear hierachial schema of where those procedures fit in the two separate aspects of our soil communities identification and analysis project (I. id and characterization of culturable bacteria and II., id of soil community bacteria by 16s rDNA sequencing of soil genomic DNA). You can get a broad sense of the progress of the two parts of our project from the narrative and the two schematics found on the Introduction to the Project page at BISC209:Project1. There is a more extensive description of one of the two main parts of the project: the multi-week process of 16S rDNA sequencing of the soil genomic DNA isolate that you prepared this week, found at the beginning of LAB 3. Make sure you read and understand what we are doing at each step and how that step fits into our overall goal of getting a sense of the diversity, role, and relationships among the bacterial flora in a particular soil community.
Through the week check on and perform any relevant steps on your cultures following the relevant protocols.
Links to Labs
Lab 1
Lab 2
Lab 3
Lab 4
Lab 5
Lab 6
Lab 7
Lab 8
Lab 9
Lab 10
Lab11
Lab 12