Stanford/BIOE44:Module 1:Day2
M1: Day 2 - Gel Extraction and Ligation
Introduction
Last time we digested the color generator gene cassettes out of a plasmid. Now we need to separate the cassette fragment from the backbone fragment. To do this we will perform a gel extraction which has two steps. First we will run our digest reactions out on an agarose gel. This will separate the DNA fragments based on size. We'll cut out the band that corresponds the color generator genes. Then we'll use a kit to purify the DNA out of the the agarose piece.
After we purify the color generator gene cassette, we will insert it into a new vector that has an inducible promoter.
Background on Methods
Gel Electrophoresis
Electrophoresis is a technique that separates large molecules by size using an applied electrical field and a sieving matrix. DNA, RNA and proteins are the molecules most often studied with this technique; agarose and acrylamide gels are the two most common sieves. The molecules to be separated enter the matrix through a well at one end and are pulled through the matrix when a current is applied across it. The larger molecules get entwined in the matrix and retarded; the smaller molecules wind through the matrix more easily and travel further from the well. Molecules of the same size and charge migrate the same distance from the well and collect into a band.
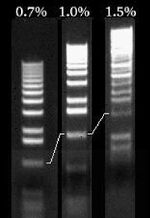
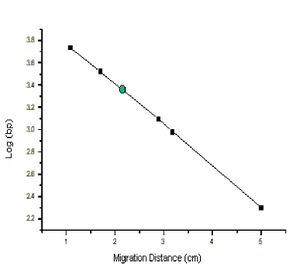
The distance a DNA fragment travels is inversely proportional to its length. Over time fragments of similar length accumulate into “bands” in the gel. Higher concentrations of agarose can be used to resolve smaller DNA fragments. This figure shows the same DNA fragments resolved with three agarose concentrations. The 1000 base pair fragment is indicated in each.
Ethidium Bromide is a fluorescent dye that is commonly added to agarose gels. This dye intercalates between the bases of DNA, allowing DNA fragments to be located in the gel under UV light and photographed. The intensity of the band reflects the concentration of molecules that size, although there are upper and lower limits to the sensitivity of dyes. Because of its interaction with DNA, ethidium bromide is a powerful mutagen and will interact with the DNA in your body just as it does with any DNA on a gel. You should always handle all gels and gel equipment with gloves.
Ligations
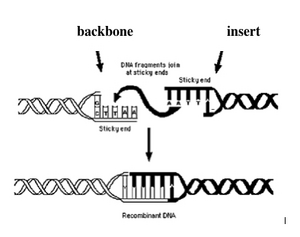
The following quote was taken from: How does DNA ligation work?
"DNA ligase (EC 6.5.1.1) covalently joins the phosphate backbone of DNA with blunt or compatible cohesive ends (see figure on top right) and it’s natural role is in repairing double strand breaks in DNA molecules. In molecular biology it is commonly used for the insertion of restriction enzyme-generated DNA fragments into vector backbones. Commercial ligases are supplied with a reaction buffer containing ATP and Mg2+, which are both essential for ligase activity. Since ATP can be damaged by repeated freeze-thaw cycles, it is advisable to make aliquots of the buffer (see my article “5 DNA ligation tips“).
The ligation reaction itself has two basic steps. Firstly the DNA ends have to collide by chance and stay together long enough for the ligase to join them. This is the most inefficient part of the reaction, but is easier at low temperatures. Why? Well, as you will probably know, all molecules move faster at higher temperatures so you can imagine that it is going to be easier for two DNA ends to collide and stay together if they are gently floating through the solution at low temperature, rather than whizzing about as they would be at higher temperatures. For cohesive ends, there is an additional reason; lower temperatures stabilize the hydrogen bonding between the complementary nucleotides, which really helps to keep things in place.
The second step is the enzymatic reaction, which is shown schematically in the figure on the left. DNA ligase catalyzes the joining of the 3′-OH to the 5′-phosphate via a two step mechanism. First, the AMP nucleotide, which is attached to a lysine residue in the enzyme’s active site, is transfered to the 5′-phosphate. Then the AMP-phosphate bond is attacked by the 3′-OH, forming the covalent bond and releasing AMP. To allow the enzyme to carry out further reactions the AMP in the enzyme’s active site must be replenished by ATP."
In Class
Gel Extraction
Before we begin our gel extraction, we need to know what size band we are looking for. Fill in the table below with the sizes of the parts you were given (and the other groups too). This info is on the Cambridge 2009 iGEM team wiki. You should read about how your genes are producing color too.
| Number | Color | Size (bp) |
| 002 | Violet | |
| 003 | Dark Green | |
| 004 | Light Green | |
| 100 | Red | |
| 200 | Orange |
Make a gel
The gene cassettes we are working with are relatively large so we will use a 0.8% gel. When doing gel extractions, it is good to keep the agarose percentage as low as possible because this will help improve the purification later.
- To make a 50ml gel with 0.8% agarose, how much agarose (g) must you add??? Check with one of the TAs to make sure you are correct before you proceed.
- Measure out the appropriate amount of agarose and put it into a 250mL erlenmeyer flask.
- Measure 50mL of TAE (in carboy in gel station, do not use water - any ideas why?) and add to the flask.
- Microwave until you see the solution start to bubble. Take it out and check to see if there are chunks by swirling it around. Continue to microwave until there aren't any chunks left. You want to use the minimum amount of heat to dissolve the agarose.
- Let your agarose solution cool to the point where you don't see whisps of steam coming off it. This is a good time to set up the gel tray and comb.
- Once cooled, add 10uL of the 5000x ethidium bromide (EtBr) solution. Swirl to mix.
- Pour the agarose/EtBr solution into the tray and let cool until solid (15-20minutes).
Load and Run the gel
Before loading your sample on the gel, you must first mix it with loading dye. Loading dye does two things: 1) it adds density to your sample so it will sink into and stay in the well and 2) it helps you visualize how far your samples have run down the gel.
Most often, loading dye is at 6x concentration. How much dye would you add to:
- A 10uL sample?
- A 50uL sample?
- your sample?
Protocol
- Move your solidified gel into a gel box. Make sure you have oriented your gel correctly - the wells should be at the negative pole. Why is this important? Add TAE until the chambers on either side of the gel are filled, the gel is covered and the wells of the gel are filled. Why do you think we use TAE instead of water?
- Add (whatever volume you calculated for your sample) loading dye directly to your sample. Pipette up and down to mix. For purposes other than gel extraction, you won't add the loading dye directly to your sample.
- Load 10uL of ladder into the first well of your gel.
- Load your sample into the next well. It helps to eject the sample very slowly - this allows the sample to settle into the well and maximizes the total volume you can fit in the well. If your sample is greater than 50uL, split it into two wells.
- Put the lid on the gel box and turn on the power supply. Run your sample at 95V.
Cut out the bands
Watch this video.
<html> <object width="425" height="350"> <param name="movie" value="http://www.youtube.com/v/K_tFVieCJcA"> </param> <embed src="http://www.youtube.com/v/K_tFVieCJcA" type="application/x-shockwave-flash" width="425" height="350"> </embed> </object> </html>
(also take note of how awesome the hand model is...)
Things to do differently from this video:
- Please take your gel out of the tray and lay it directly on the plastic wrap covered transilluminator.
- You do not need to use a new razor blade for each cut.
- If you cut your piece small enough, you don't need to cut it in half to put into the tube.
Extract the DNA out of the gel piece
For this part we will be using a Qiagen Gel Extraction kit. Please follow the instructions in this PDF.
Helpful hints:
- Gel extraction kits are located on the shelf near the sterile tips.
- Make sure one of the heat blocks is set to 50C before you start.
- Isopropanol is in a labelled 50mL tube below kit shelf.
- You do not need to do step 9.
- Elute in 30uL of water.
Ligation
Now that we have purified the gene cassettes, we will insert them into different vectors with inducible promoters. Next week we will characterize these composite parts. (FYI you guys will be making new composite parts that have never been made before... we may even submit these with our characterization data to the registry!)
You will have a choice of two inducible promoters:
- I0500
- R0011
Find these parts in the registry and write down:
- What are the promoter names and what they induced by?
- What are the concentrations of inducer relevant for induction?
- What plasmid are these parts on?
- What is the selection marker on the plasmid?
- The vectors with the inducible promoters you will get are precut, but at what sites were they cut?
- What is the size of cut vector?
Details of the ligation
Please read this DNA ligation protocol and determine what your ligation reaction components and volumes should be. This ligation calculator will be helpful.
Helpful hints:
- Don't forget to set up a ligation control. What would be a good control?
- Use a PCR tube.
- Set up a program on the PCR block that will incubate your ligation at room temperature for 30 minutes and then heat inactivate it at 65C for 10 minutes. (TAs will demonstrate how to write a program on the PCR block).
- Ignore the recommended dialysis step.
