Paper Microfluidics - Yiliang Zhou, Aditi Naik
Motivation for Paper Microfluidics
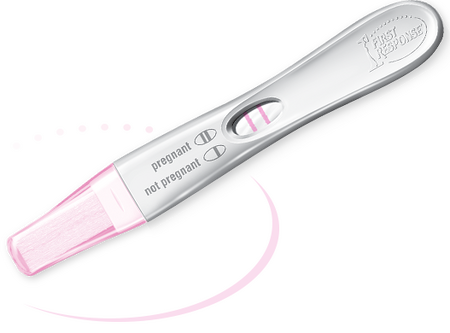
Paper-based microfluidic devices are devices that can control and manipulate small amounts of liquid on a cellulose substrate. Cellulose, a major component of all plant matter, is one of the most abundant biopolymers in nature.[1] Compared to traditional microfluidics, cellulose substrates provide the opportunity for applications to be low-cost, low-weight, and flexible.[2] Moreover, fluid flow can be easily driven by capillary forces within a microchannel, which requires no external power supply. The porous structure of cellulose provides a high-surface area to volume ratio, which can increase the sensor performance of these types of devices. Cellulose substrates also have the ability to store reagents in active form within the channel. All these advantages of using cellulose make it an ideal material for microfluidics devices. As shown in Figure 1, one of the most well-known paper-based microfluidics devices is a simple pregnancy test.
Challenges
Compared to traditional microfluidics, paper-based microfluidics still have the following limitations:
(1) Small sample retention
(2) For samples with low surface tension, some hydrophobic agents are not strong enough for patterning devices
(3) Detection limits are usually high via colorimetric detection
Fabrication Methods
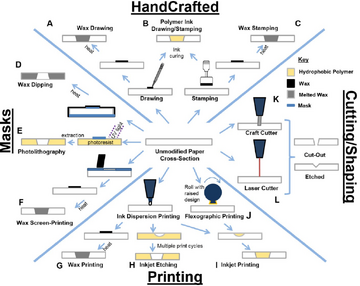
Cellulose paper is a hydrophilic substrate. In order to fabricate microfluidic channels, specific methods need to be used to tune the hydrophobicity of the cellulose substrate. Two mechanisms have been applied to pattern microfluidic channels. One is to selectively fabricate a hydrophobic surface onto cellulose film. The other mechanism modifies the cellulose film as a hydrophobic substrate first, and then, selectively tunes part of the surface back to a hydrophilic surface. Until now, different techniques have been proven to be feasible to create microfluidic channels on the cellulose substrate, such as wax printing,[4] inkjet printing,[5] photolithography,[6] flexographic printing,[7] screen printing,[8] laser cutting[9] and plasma treatments.[10] Researchers have divided these methods into four different categories, as shown in Figure 2.[11]
Wax Printing
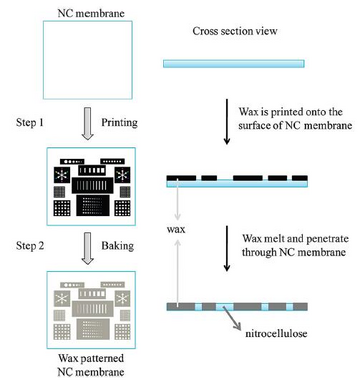
Wax printing is a technique that uses wax as a hydrophobic agent on the cellulose substrate.[4] The mechanism for it is illustrated in Figure 3.[4] Wax can be easily printed through a wax pen, inkjet printer or wax printer onto the surface of a cellulose membrane. Wax can then be melted, to allow it to penetrate through the cellulose under mild heat treatment. The wax surface becomes a hydrophobic surface. Wax printing is a simple, rapid, and environmentally-friendly method for patterning. A disadvantage of this method is that researchers need expensive wax printers and an extra heating process to complete the print.
Inkjet Printing
Inkjet printing is a new method of computer printing to transfer digital designs onto different substrates with droplets of ink. Specific biomolecules can be precisely printed onto a sensing zone of a paper-based microfluidic device.[5] As for patterning microfluidic channels, inkjet etching and inkjet printing have been developed. For inkjet etching, toluene has been used as an inkjet agent to selectively remove hydrophobic polystyrene that was pre-patterned on the paper. For inkjet printing, hydrophobic agents, such as alkyl ketene dimer, can be directly printed on a paper to form a hydrophobic barrier.[11] The advantages of inkjet printing include high resolution and a wide variety for choice of inks. Electrodes can also be printed by this method. A disadvantage of this process is that the speed of printing is not feasible for massive production unless roll-to-roll fabrication is used.
Photolithography
Photolithography exploits the properties of photoresists on paper. Photoresist-saturated paper is exposed to UV light through a photomask. After exposure, the uncured photoresist can be removed via organic solvent. Cured photoresist form as a hydrophobic barrier on the paper.[6] The fabrication process is rapid and allows for high resolution. A drawback is that photolithography requires the use of expensive photoresists as well as organic solvents which can be unsafe.
Flexographic Printing
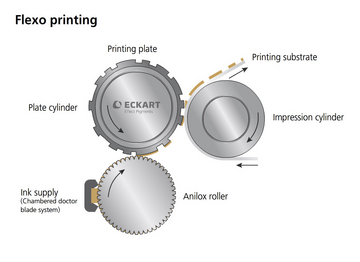
Flexographic printing is a high-throughput fabrication technique. An illustration demonstrating flexographic printing is shown in Figure 4. Olkkonen et al. used flexographic printing with polystyrene ink to fabricate a microfluidic channel on a paper substrate.[7] This method can be easily scaled-up, and the speed of printing can be greater than 300 m/min. A disadvantage of it is that different printing plates are needed. Meanwhile, it can only print one reagent at one time.
Screen Printing
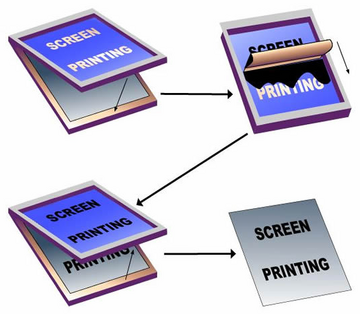
Screen printing is a printing method that uses a partially blocked mesh to selectively transfer ink on the substrate, using a blocking stencil. The detailed procedure is visible in Figure 5. The process of screen printing is simple, however, the resolution is low, and different blocking stencils are needed for different designs.
Laser Cutting
The laser cutting uses a computer to control a CO2 laser to cut paper substrate into a specific design. No additional chemicals are needed during this process. Some disadvantages of this method include the expense of the laser cutter being far more than a knife, and often the power of such a laser is strong enough to cause warping or tearing in these paper substrates.
Plasma Treatment
Plasma treatments exploit the chemistry of specific additives. Cellulose substrate can be pre-modified via octadecyl trichlorosilane silanization to fabricate a hydrophobic surface. Plasma treatment with a mask will revert the exposed substrate to a hydrophilic surface by the degradation of octadecyl trichlorosilane.[10] A drawback of this method is that the substrate under a mask is easily over-etched.
Applications
The major application of paper-based microfluidics is providing low-cost, user-friendly analytic platforms for assay diagnoses. Pregnancy tests are a well-known example of paper-based microfluidics. Researchers have used paper-based microfluidics for a wide range of applications, such as biochemical detection, immunological detection, and molecular detection.[13]
Biochemical Detection
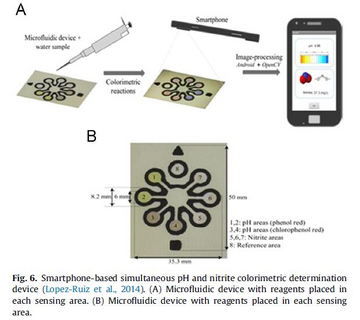
Many analytes have proven to be detectable with paper-based microfluidics. In the detection zone on the paper substrate, the analytes can have a chemical reaction with an immobilized reagent and develop a signal. This signal can be detected via different methods, some examples include colorimetric, electrochemical, fluorescent, and chemiluminescence (CL) signaling. Lopez-Ruiz et al. successfully developed paper microfluidic devices with applications in pH and nitrite colorimetric determination.[14 ]
Immunological Detection
Immunological detection is used to detect analytes with an immunoassay technique. Antibodies or proteins can be covalently bonded on a paper substrate with surface modification. One advantage of paper-based microfluidics in immunological detection is that paper substrates have the capability of storing reagents in the active form. This is crucial for applications in low-resource settings and point-of-care applications.
Molecular Detection
Sequence-specific detection of nucleic acid hybridization has been proven to be detectable with paper-based microfluidics. A sequence with a tag or change in the concentration can be targeted by a capture probe, allowing a reaction to being visible and potentially measurable.
References
1. Klemm, D.; Heublein, B.; Fink, H.-P.; Bohn, A. Cellulose: Fascinating Biopolymer and Sustainable Raw Material. Angewandte Chemie International Edition 2005, 44 (22), 3358–3393. https://doi.org/10.1002/anie.200460587
2. Zhou, Y.; Fuentes-Hernandez, C.; Khan, T. M.; Liu, J.-C.; Hsu, J.; Shim, J. W.; Dindar, A.; Youngblood, J. P.; Moon, R. J.; Kippelen, B. Recyclable Organic Solar Cells on Cellulose Nanocrystal Substrates. Scientific Reports 2013, 3 (1). https://doi.org/10.1038/srep01536
3. PNG all. https://www.pngall.com/pregnancy-test-png/download/46678 (accessed Feb 28, 2022).
4. Lu, Y.; Shi, W.; Qin, J.; Lin, B. Fabrication and Characterization of Paper-Based Microfluidics Prepared in Nitrocellulose Membrane by Wax Printing. Analytical Chemistry 2009, 82 (1), 329–335. https://doi.org/10.1021/ac9020193
5. Abe, K.; Suzuki, K.; Citterio, D. Inkjet-Printed Microfluidic Multianalyte Chemical Sensing Paper. Analytical Chemistry 2008, 80 (18), 6928–6934. https://doi.org/10.1021/ac800604v
6. Martinez, A. W.; Phillips, S. T.; Butte, M. J.; Whitesides, G. M. Patterned Paper as a Platform for Inexpensive, Low-Volume, Portable Bioassays. Angewandte Chemie International Edition 2007, 46 (8), 1318–1320. https://doi.org/10.1002/anie.200603817
7. Olkkonen, J.; Lehtinen, K.; Erho, T. Flexographically Printed Fluidic Structures in Paper. Analytical Chemistry 2010, 82 (24), 10246–10250. https://doi.org/10.1021/ac1027066
8. Renault, C.; Scida, K.; Knust, K. N.; Fosdick, S. E.; Crooks, R. M. Paper-Based Bipolar Electrochemistry. Journal of Electrochemical Science and Technology 2013, 4 (4), 146–152. https://doi.org/10.5229/JECST.2013.4.4.146
9. Nie, J.; Liang, Y.; Zhang, Y.; Le, S.; Li, D.; Zhang, S. One-Step Patterning of Hollow Microstructures in Paper by Laser Cutting to Create Microfluidic Analytical Devices. The Analyst 2013, 138 (2), 671–676. https://doi.org/10.1039/C2AN36219H
10. Cai, L.; Xu, C.; Lin, S. H.; Luo, J.; Wu, M.; Yang, F. A Simple Paper-Based Sensor Fabricated by Selective Wet Etching of Silanized Filter Paper Using a Paper Mask. Biomicrofluidics 2014, 8 (5), 056504. https://doi.org/10.1063/1.4898096
11. Cate, D. M.; Adkins, J. A.; Mettakoonpitak, J.; Henry, C. S. Recent Developments in Paper-Based Microfluidic Devices. Analytical Chemistry 2015, 87 (1), 19–41. https://doi.org/10.1021/ac503968p
12. Schofield Printing. http://www.schoprint.com/screenprinting.html (accessed Mar 26, 2023).
13. Xia, Y.; Si, J.; Li, Z. Fabrication Techniques for Microfluidic Paper-Based Analytical Devices and Their Applications for Biological Testing: A Review. Biosensors and Bioelectronics 2016, 77, 774–789. https://doi.org/10.1016/j.bios.2015.10.032
14. Lopez-Ruiz, N.; Curto, V. F.; Erenas, M. M.; Benito-Lopez, F.; Diamond, D.; Palm a, A. J.; Capitan-Vallvey, L. F. Smartphone-Based Simultaneous Ph and Nitrite Colorimetric Determination for Paper Microfluidic Devices. Analytical Chemistry 2014, 86 (19), 9554–9562. https://doi.org/10.1021/ac5019205
