Macrofluidics for Metastasis: Carbohydrate Lattices
Introduction
Cancer is one of the leading causes of death throughout the world and cancer metastasis accounts for about 90% of cancer related deaths [1]. Better understanding of the complex process of metastasis is essential in creating improved treatment methods. One goal is creating an in situ microenvironment for the study of cancer metastasis and to improve current models. Cancer metastasis has an complex mechanism that occurs through a series of steps where the tumor cells migrates from their initial tumor location and then by invading surrounding tissues and infiltrating the circulatory system cause malignant tumor growth on a secondary organ [4].
Jordan Miller from the University of Pennsylvania led research in creating stiff carbohydrate lattices for the vasculation of synthetic tissues. Improving methods of creating channels is important in creating new synthetic materials for tissue engineering. There is a large need for organ donations. For example, in 2015 there were around 119,000 patients in need for an organ transplant, but only 30,970 organ transplants were performed [2]. In 2002 the number of people died on the waiting list exceed 6,000 [3]. Research in tissue engineering has promising applications from wound healing to the production of synthetic organs. One of the major challenges is most of the materials that are produced are avascular tissue. Fundamentally tissues are complex and composed of many vascular networks that can deliver nutrients, oxygen, hormones, etc to the cells that make up the greater system. The goal of many literature is to add 3D permeable structures in biomaterials to enhance regenerative and and engineered tissues.
The main focus of this project is to create a microenvironment for the study of cancer metastasis through the use of carbohydrate lattices. Molecular and genetic mechanisms underlying cancer have been widely studied in 2D models using cultured cancer cells. In vitro models are important in the study of cancer models because both animal testing and 2D layer models have drawbacks. For example, mice are usually injected with cancer cells that are not naturally metastatic which could lead to misinformation. Since the average cost to bring a drug to market is about 5 billion dollars it is advantageous to understand the metastatic process. This projects main goal is to use carbohydrate glass to create channels in hydrogels to mimic in vivo conditions.
Background
Cancer Metastasis Overview
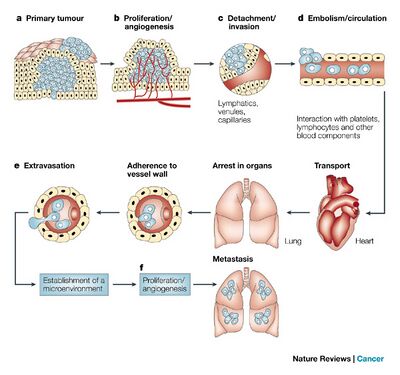
Metastasis occurs through a series of sequential steps in which tumour cells first migrate from the primary tumour, penetrate blood vessels and then colonize distant sites. It is generally characterized by the following steps:
- local infiltration of tumor cells into surrounding tissue
- transendothelial migration of cancer cells into vessels (intravasation)
- circulatory system infiltration
- etrasvascation
- proliferation in organs leading to tumor formation [5]
Figure 1: shows an overview of how metastasis occurs. First the malignant growth must have neoplastic cells with nutrients for the expanding tumor and extensive vascularization must occur to form capillary networks with the surrounding host tissue. After these steps the tumor cells invade the surrounding tissue. Thin venules are the easiest way for cancer cells to enter the circulation system due to their thin nature; however there are several different methods which this can occur. Most tumor cells entering the bloodstream are destroyed quickly, but once tumor cells survive circulation they become trapped in capillary endothelial cells on different organs. Extravasation then occurs inside the organ like intravasation this usually occurs through the penetration of thin venules but there are other modes of infiltration. Proliferation into the organs is the last step that is characterized by continued growth and destruction of immunological responses [6].
Carbohydrate Lattices

Carbohydrate lattices are created using 3D printing technology to serve a sacrificial structure that dissolve to form a flow channel in PEG which represents the tissue environment. The flow channel is used to circulate tumor cells through them which allows the flow and interaction of tumors cell in tissue environment to be studied. Miller et all produced carbohydrate lattices through thermal extrusion and fibre drawing with a 3D printer [7]. The glass reported is a brittle material with a young modulus at room temperature of 1 Gpa. Figure 2 shows the architecture of a lattice structure. By keeping the die diameter constant the diameter of the extrusion can be controlled by the translational velocity shown below.
[math]\displaystyle{ D(v) = \frac{A}{\sqrt{v}} }[/math]
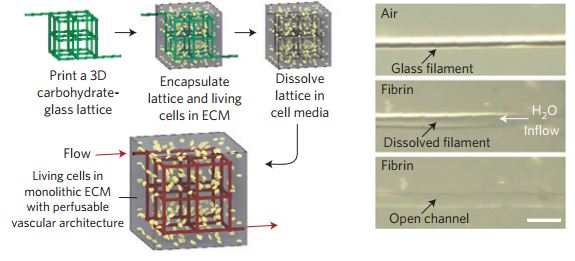
Figure 3 is a schematic overview of carbohydrate lattice used by Miller et al where the carbohydrate lattice is encapsulated by PEG hydrogel ECM which represents the tissue environment. As shown in the figure living cells were introduced together with the PEG hydrogel ECM and the cell media dissolves the carbohydrate lattice in within minutes while not damaging the cells. This process creates a monolithic tissue construct that matches the carbohydrate lattice that was dissolved in the earlier step.[3]
Current metastasis study
A common method to study cancer metastasis is to transplant cancer cells to small animals such as rats and mice and study how the cancer cell spread throughout the animal’s body. The transplant model can be separated into two different models the syngenic models and xenograft model. In addition, there are a variety of ways to create devices for the study of cancer metastasis including 3D printing, soft lithography, photographically,and microcontact printing. Projects like Recreating the Tumor Microenvironment deal in microfluidics where an automated pneumatic valve system controls laminar flow flow for the packing tumor spheroids. Macrofluidics for Tumor Metastasis creates devices using 3D printing for the purpose of creating metastasis models. In addition endothelial cords is another method used in researching metastasis models, Macrofluidics for Metastasis: Endothelial Cords.

Hydrogels have been extensively studied to mimic ECM and are an integral part of creating realistic in vitro environments. Hydrogels allow for the diffusion of different components like oxygen and nutrients. They are sally bicompatible materials like collagen and polyethylene glycol. In addition hydrogels are adjustable to different in vivo systems. Hydrogel stiffness can range several orders of magnitude like body tissues [8]. Hydrogels play an important role in creating cancer models because they cam mimic the ECM.
Many methods are going to be used to get a full picture of cancer metastasis. Our project hopes to use carbohydrate glass as a novel way to make a device for the study of cancer metastasis. We are the first group to use carbohydrate lattices for this purpose, so there is no directly comparable method besides creating a channel with the use of a needle which may disrupt the structure of the hydrogel.
Benefits of Transitioning to the Microscale
By modeling the microenvironment of the tissues using PEG-hydrogels the interactions and flow pattern through the PEG-hydrogels become much more reliable since the environment is very similar to a real tissue. PEG-hydrogel can model the microenvironment because the pore size of the hydrogels are very similar to vein size. Also the flow of tumor cells can be easily studied because at microscale only laminar flow exists.
Experimental Procedure and Methods
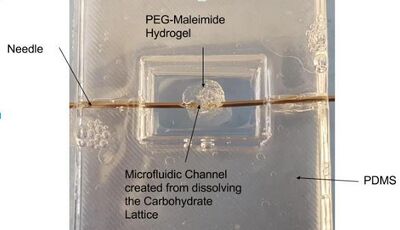
To help study the metastasis of breast cancer a sacrificial carbohydrate lattice will be used to create a microfluidic channel within PEG maleimide hydrogel which used replicate the tissue environment. PDMS skeletal structure was used to help contain PEG-maleimide hydrogel. The center of the mould shown in figure [3] was used to create the chamber for the hydrogel. After the PDMS is complete 28 gauge catheter inserted with needle into both sides of PDMS to meet at the center of hydrogel chamber.(figure [4]) Afterwards, the carbohydrate lattices is placed between needles and enough PEG-maleimide 8 arm hydrogel was added to engulf the entire carbohydrate lattice. Phosphate buffered saline(PBS) was added to induce the hydrogel to swell which increases the microscopy resolution. After waiting overnight for the hydrogel to fully swell the needle is removed from the catheter and fluorescent microbead solution is inserted into the catheter which dissolved the carbohydrate lattice on contact. Then the microfluidic device is placed under a 5X magnification microscope to determine whether the microfluidic channel was created or not.
References
[1] Marius, L.; Hofman, V.; Long-Mira E. Circulating Tumor Cells Allow Early Diagnosis of Lung Cancer in Patients with Chronic Obstructive Pulmonary Disease. PLoS ONE. 2014, 9
[2] Mehlen, P.; Puisieux, A. Nature Reviews Cancer 2006, 6 (6), 449–458. DOI: http://dx.doi.org/10.1038/nrc1886
[3] Organ Donation Statistics https://www.organdonor.gov/statistics-stories/statistics.html (accessed Apr 7, 2017).
[4] Zijl, F. V.; Krupitza, G.; Mikulits, W. Mutation Research/Reviews in Mutation Research 2011, 728 (1-2), 23–34. DOI: http://dx.doi.org/110.1016/j.mrrev.2011.05.002
[5] West, J.; Moon, J. Current Topics in Medicinal Chemistry 2008, 8 (4), 300–310. DOI: http://dx.doi.org/10.2174/156802608783790983
[6] Fidler, I. J. Nature Reviews Cancer 2003, 3 (6), 453–458. DOI: http://dx.doi.org/10.1038/nrc1098
[7] Jordan S. Miller et al. Nature Materials 2012;11:768-774 DOI: http://dx.doi.org/10.1038/NMAT3357
[8]Sunyer, R.; Jin, A. J.; Nossal, R.; Sackett, D. L. PLoS ONE 2012, 7 (10). DOI: https://doi.org/10.1371/journal.pone.0046107
