Macrofluidics for Metastasis: Endothelial Cords
Overview of Cancer Metastasis
Cancer is the word that strikes fear and pain into the hearts of anybody who has lost a loved one to this globally relevant disease. The difficulty in eliminating this threat lies primarily in that, fighting it, is almost indistinguishable from fighting ones own body. Cancer and your body are made up of similar cell types, they reproduce similarly (though obviously cancer is doing it faster), and they even use the same blood stream to supply nutrients. That same blood stream is how cancer can spread throughout the body and that is exactly when cancer becomes lethal in most instances, as it spreads from a contained region to areas that are far less salvageable. So perhaps, if we could prevent the spread of cancer, we could effectively neutralize its lethality.

In metastasis, cancer cells from the initial tumor site spread to distant sites in the body. Tumors are made of billions of cells that each have the potential to spread to different tissue types in the body, but there is currently no effective simulation to study how these metastasizing cells may differ from A) non-cancerous cells or B) standard cancerous cells from a vantage point that is outside of the body in an environment that is controlled. In addition its also well documented that the vast majority of metastasizing cells do not in fact succeed in lodging onto another region of the body, which causes studies that intend to investigate this phenomena, to have to be able to run a large number of trials or trials that last for an extended period of time in order to observe successful metastasis on a quantifiable level.
The significance of metastasis grows continuously as early detection and aggressive treatments for isolated cancers continue to swing the percentage of cancer related deaths towards those tumours that have quickly or quietly reached a higher stage and metastasized.
Factors involved in Tumor Growth and Migration
Angiogenesis
An essential aspect of tumor growth and metastasis is angiogenesis. Angiogenesis is the development of new blood vessels. Studies have shown that tumors are unable to grow more than 2-3 mm in diameter without proper vasculature, as this vasculature is necessary to fulfill the energetic requirements of tumor cells. Thus, in order to keep growing, tumor cells precipitate formation of new blood vessels from existing blood vessels. Cancer cells can stimulate angiogenesis through a variety of cell-to-cell communications. Cancer cells also have the ability to induce endothelial changes by direct and indirect ways. They are able to directly target cells by adhesion receptors, soluble factors, gap junctions, and vesicles. Indirectly, they stimulate the signaling pathways of endothelial cells by secreting proteases into the extracellular space or activating stromal cells. Cancer cells are even able to change the pH, temperature, and availability of nutrients and oxygen to indirectly induce endothelial changes. [1]
Endothelial Cells
The entire vascular system is lined by an endothelium, which is composed of a single layer of endothelial cells. Endothelial cells form the inner lining of blood vessels, creating an anticoagulant barrier between the wall of the vessel and blood. This barrier acts as a selectively permable membrane. The endothelial cell also plays a significant role in basal and inducible metabolic and synthetic functions. They react with chemical and physical stimuli, regulate homeostasis, vasomotor tone, and immune or inflammatory responses of the body. In regards to metastasis and tumor growth, endothelial cells play a crucial role in angiogenesis. [2]
Tumors are comprised of both tumor cells and stromal cells. Endothelial cells are one type of stromal cell that is part of the tumor microenvironment. The cell-to-cell communication between endothelial cells and tumor cells can lead to tumor angiogenesis, and studies have shown that interrupting this signal exchange may result in reduced vasculature and reduced tumor size. Strategies to halt the communication signals between endothelial cells and tumor cells have become an area of interest in cancer research for this reason. It is believed that metastasizing cells interact with new regions far from the initial tumor via a number of possible mechanisms. One such theory is that the metastasizing cells use soluble factors such as the various vascular endothelial growth factors (VEGFs), that cause the new regions to accept and integrate it and allow it to reproduce there or in conjunction with a variety of ways of direct contact between cells such as with vesicles, or via adhesion contact and the receptors associated with that. Other theories are based more on indirect cell to cell communications such as through over stimulating the fibroblasts or using proteases to degrade the extracellular matrix which causes pro angiogenic factors to abound in the tumor microenivronment. [1]
Current Methods for Observing Metastasis
In vivo models
Transplantable Syngeneic Mouse and Human Xenografts
Transplantable models utilize cancer cell lines or tissues that are grown in mice or rats. These models can be categorized into two groups - syngeneic models and xenograft models. Syngeneic transplantable models generally deal with mouse or rat cancer cell lines or tissues that cause tumors in animals that have the same genetic background as the cell line or tissue. The transplanted tissues, tumor microenvironment, and host in a syngeneic model are all from the same species. This is the main advantage of using this kind of model due to the close interaction between the host and tumor in metastasis. A disadvantage of this model is that it lacks a myriad of the important characteristics of human tumors. Homozygously inbred mice, which are commonly used in syngeneic models, have tumors that lack the genetic complexity of tumors that occur in humans.
Human-mouse xenograft models deal with human cancer cell lines/tissues that are transplanted into animals with compromised immune systems. These human cancer cell lines or tissues then grow tumors within the animal. Although this model attempts to more closely replicate the genetic complexity of human tumors, evidence suggests that cancer cell-stromal cell interactions that are key in the progression of metastasis can not occur across species boundaries. The immunocompromised immune system of the animal also removes the possibility of studying the tumor interaction with the immune system. [3]
Genetically engineered mice
A myriad of genetically engineered models have been developed which produce genuine metastatic disease. The tumors in these models arise in their normal context, in a system that has a functional immune system, as opposed to xenograft models. These genetically engineered models include many different tissue types with varying degrees of penetrance and different latencies. Metastasis models based on genetically engineered animals allow important determinants of cancer to be investigated that are otherwise difficult to study using cell culture and transplant systems. For example, the effect of genetic heterogeneity on tumor phenotype can be investigated, which has shown to play a significant role in the expression of tumor mutations. This suggests that metastasis may be profoundly influenced by genetic background, but the evaluation of this genomic polymorphism is not possible using cell culture-based systems. A major disadvantage of genetically engineered models is the expense, though. The latency for metastases is still on the order of months, and the relative penetrance for metastatic disease is frequently much lower than that of tumor incidence. This necessitates large numbers of animals to be held for lengthy periods of time to have a large enough sample size for data analysis. [3]
Comparative models
Mice are often used for in vivo experiments given their short lifespans, availability, and well documented number of genetic alterations that enhance in vivo study. However, the biological comparison of cancer in mice versus humans isn't as close as it could be ideally. Interestingly, the canine genome is more similar to the human genome than mice is to either of them, and additionally since the the tumor is structurally and functionally similar and the likelihood of metastasis is also comparable between the two species. It is also worth noting the large number of canine pets that are diagnosed with cancer, and whose owners seek treatment, and therefore the considerable amount of research data to be had from the study of these canine cancers and from the administration of prospective novel treatments or drugs that are not yet cleared for human trials. In addition, after using this data to compare dog cancers to humans cancers, there are some interesting parallels that can be drawn. First off, environmental factors that predispose humans to cancer can also cause cancer in canines. This factor enables dogs to be good indicators of whether a human carcinogen is present. A distinction from human cancers though is that canine cancers will almost always manifest and metastasize faster, which cements their ability to forewarn of carcinogens. Fortunately, with the advent of the canine genome project, the major weakness of studying canine cancers was lifted in tha tagging important reagents is now a plausible task, and will lead to more thorough studies of these cancers. [3]
In vitro models
Several current methods exist for modeling metastasis that involve replicating angiogenesis to create effective in vitro simulations. One of the first things to understand when attempting to replicate angiogenesis, is that, in addition to getting all the growth factors and other stimulants correct, there are physical factors that heavily impact the proficiency with which vascularization operates. The kind of structure you give the veins to grow in will either help or hinder their ability to encourage the kind of blood flow you would expect from an authentically formed structure.
Scaffolding
Scaffolding is prominent due to its ability to provide multilayered structural support during implantation as well as the the ability to adjust porosity of the scaffold which allows it to accommodate the space requirements of many cell types and other biologically relevant molecules. Often the structure is 3D printed and depending on the intended use, simply implanting the scaffold itself is sufficient, or the the scaffold can be seeded with cells before implantation, or a culture can be performed on the surface of the scaffold in a bioreactor if necessary. [4]
Prevascularization in Culture
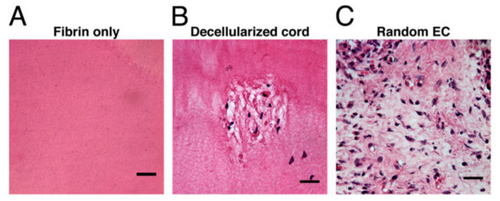
This method, alternatively called "random arrangement", proposes a co-culture of the necessary growth factors in combination with the mural cells. This combination allows the vascularization to occur alongside the formation of the structure and encourages a thorough integration. It isn't microfluidics-based which makes it a simpler method since post culture, the only step is to implant the structure to see how it performs. However, because of the lack of direction given to the forming veins, they tend to form extremely tortuous shapes which lead to poor blood flow and subsequently leaky veins. [5]

Challenges of Current Methods
Unfortunately however, most of these current methods in modeling metastasis are little more than reasonable theories with some supporting evidence. Although a multitude of ways for modeling metastasis have been attempted, each method does not account for all factors that contribute to tumor growth and migration. It is unlikely that any one model can account for the complexities of human cancer cells and metastasis. In order to combat metastasis we would first need to cement our understanding of cancer metastasis and growth through conducting more precise experiments about these theories that encompass many of the complex factors that contribute to human tumor growth and migration. Normally, to conduct more rigorous experiments, you need to implement greater levels of control over the experiment so as to isolate factors or interactions. Looking at how most experiments are conducted, either A) in vitro or B) in vivo, its understandable that in vivo studies are not nearly as helpful given the difficulty with imaging cells individually, along with the lack of control that is applicable to an in vivo environment. This is precisely what makes it difficult to assess the potency of individual factors. So the main focus on cancer studies these days, is in how to create an in vitro experiment, with only the factors of interest that is also simultaneously an effective simulation of the environment that cancer cells typically operate in. [6],[7]
Microfluidics for Modeling Metastasis

There are several advantages in regards to transitioning to the microscale for modeling cancer metastasis. For starters, reducing the scale of the environment is often a good place to start when attempting to implement greater control in a system. In particular the utilization of microfluidic devices for cell culture is advantageous for several reasons in and of itself. Microfluidic devices exhibit greater experimental regulation and flexibility due to the use of polydimethylsiloxane (PDMS), the material which is the "cement" of nearly all device molds. This allows the device design to be adapted to the needs of specific individual cell types. The small-scale of the device also enables a lower number of cells and reduced consumption of reagents, which is a significant economic benefit.
Methodology
The overall objective of this project is to create macro-channels that allow the flow of circulating tumor cells through biomaterials representative of specific tissue sites that cancer cells commonly spread to. Our device serves as a microfabricated template with the purpose of generating patterned assemblies of endothelial cords that have been proven to be effective at stimulating angiogenesis; Therefore, they will also be effective at simulating an in vivo environment where we can study the interactions between metastasizing cells and other individual cells and/or factors. [8]
Endothelial Cell Cords
Forming patterned endothelial cell (EC) cords are a newer method that employs the use of microfluidic processes to develop a device that can be molded into almost any shape, that is easily replicable, and has a surface that is customizable. A material known as PDMS is used to make the mold (which is custom designed through the processes of photo- and soft- lithography). PDMS has become well established over its ability to serve as a suitable cell culture surface. In regards to EC cords however, PDMS is preferred for its nonpolar surface which allows the PDMS to be peeled away from the cords after they have been polymerized within the mold. Once made, the EC cords not only provide a clear cut path for the veins to progress down, but also the distance cords have from each other can be predetermined so that angiogenesis between veins is promoted to support superior blood flow. This method of bolstering the vascularization process makes EC cords one of the more attractive options for creating vascularized tissues to study metastasizing cells in. EC cords also only need a fraction of the cells that a prevascularized random arrangement would need. [8]
Materials
This section is to discuss some of our material choices and potential modifications that can be made.

A photoresist mask was designed in Adobe Illustrator and sent to Fineline Imaging so that they could produce a relatively cheap and high resolution mask. The PDMS used in for the device was Sylgard 184 because it has shown to be more biologically friendly than RTV 615. HUVECs are used over other endothelial cell types for their low cost and that the technique for isolating them is relatively simple. The surface of PDMS is naturally nonpolar which is acceptable for this device design because the EC cords should be removed with as little difficulty as possible, but if this were not the case and cell adhesion was preferable, coating the surface with fibronectin or Poly-D-Lysine are effective ways to encourage cell adhesion to the surface. The EC cord is seeded with only HUVECs, but there are a good variety of additional cells or beneficial molecular factors that could be added to potentially increase angiogenesis such as Fibroblast Growth Factor which directly aid in the replication of endothelial cells and in their ability to form cylindrical structures, Vascular Endothelial Growth Factor to encourage the growth of new blood vessels in areas that are oxygen deprived, or even Matrix Metalloproteinase which destroys vein walls in order to allow for branching of the vein.
[8]
Procedure

- Culture HUVECS
- Feed with F12K media every 2-3 days
- Split cells roughly every 10 days
- Photolithography
- Clean wafer with HDMS
- Spin at 2000 RPM for 60 seconds
- Apply SU-8 2000.5 to enable adhesion of photoresist
- Spin at 2000 RPM for 60 seconds
- UV exposure for 10 seconds
- Bake for 1 minute at 95 degrees Celsius
- Apply SU-8 2050 photo resist
- Spin at 1500 RPM for 60 seconds
- Bake for 5 minutes at 65 degrees Celsius, then for 20 min at 95 degrees Celsius
- UV ray exposure of wafer while its covered with photoresist mask
- 25 seconds of exposure
- Bake for 5 minutes at 65 degrees Celsius and then 12 minutes at 95 degrees Celsius
- Develop with SU-8 developer
- Immerse for 10 minutes
- Wash with fresh SU-8 developer
- Clean wafer with HDMS
- Soft Lithography to create PDMS mold
- Thoroughly mix Sylgard 184 pdms and respective curing agent in 1:10 ratio
- Place in degasifier for 1 hour
- Pour mixture onto design wafer
- Heat at 100 degrees Celsius for 30 minutes
- Cut and peel PDMS mold from wafer
- Seed cultured cells into collagen gel
- Centrifuge gel into PDMS mold
- Polymerize gel to fibrin
- Peel off PDMS
- Implant polymerized gel to test efficiency
[8]
References
[1] Lopes-Bastos, B. M.; Jiang W. G.; Cai J. Tumour-Endothelial Cell Communications: Important and Indispensable Mediators of Tumour Angiogenesis. Anticancer Research 2016, 36, 1119-1126.
[2] Sumpio, B.E.; Riley, J.T.; Dardik, A. Cells in focus: endothelial cell. The International Journal of Biochemistry & Cell Biology, 2002, 34, 1508-1512.
[3] Khanna, C.; Hunter, K. Modeling metastasis in vivo. Carcinogenesis 2005, 26, 513-523.
[4] Pereira, R. Bartolo, P. 3D Photo-Fabrication for Tissue Engineering and Drug Delivery. Engineering 2015, 1 (1), 90–112.
[5] Laschke, M.; Menger, M.; Prevascularization in tissue engineering: Current concepts and future directions Biotechnology Advances 2016, 34, 112-121.
[6] Halldorsson, S.; Lucumi, E.; Gomez-Sjoberg, R.; Fleming, R. Advantages and challenges of microfluidic cell culture in polydimethylsiloxane devices Biosensors & BioElectronics 2015, 63, 218-231.
[7] Zhang, J.; Nagrath, S.; Microfluidics and Cancer: Are we there yet? Biomed Microdevices 2013, 15, 595-609.
[8] Baranski, J. D.; Chaturvedi, R. R.; Stevens, K. R.; Eyckmans, J.; Carvalho, B.; Solorzano, R. D.; Yang, M. T.; Miller, J. S.; Bhatia, S. N.; Chen, C. S. Proceedings of the National Academy of Sciences 2013, 110 (19), 7586–7591.
