IGEM:IMPERIAL/2007/Projects/Biofilm Detector/Design
Infector Detector: Design
Overall Design
Possible Parts
Signaling System
BBa_F1610 RBS-luxI
No promoter - we need to put one on it!
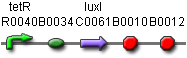
BBa_J23039 ptet-luxI
A better part has been found! This part will most likely be used.
Promoter for Signaling System
BBa_R0040
ptet, if uncontrolled
pLac, if constitutive secretion
pBAD, if controlled secretion
Detection System
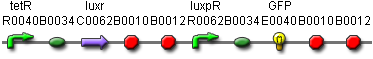
BBa_F2620 ptet-LuxR-pLux
Detection range: 1-1000 nM, highest sensitivity from 1-10 nM
Peak secretion: 400 GFP molecules cfu-1 s-1
Peak response time: 27 minutes
BBa_T9002 ptet-LuxR-pLux-GFP
With GFP attached
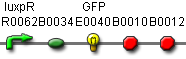
BBa_J37032 pLux-GFP
The same part, but without constitutive LuxR production
GFP Reporter
BBa_EI1354 GFP
Standard GFP, excited/emitted at 501/511 nm, with RBS and stop codons.
LacZ for β-galactosidase assay (LacZ is too big...)
Or possibly DsRed Express - this will be ordered from clontech!
Construct 1: Detector for AHL
Our first aim is to create a system that can accurately detect the concentration of AHL in solution. We found a well characterized detector system sensitive to 3OC6HSL which we will attach to a standard GFP reporter to determine its response to GFP.
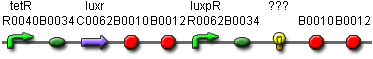
We would later test the promoter on a different reporter. The reporter that is chosen for the final comstruct needs to have a long half-life, be stable, be visible to the naked eye and should not have a response time longer than 3 hours.
It has been decided that DsRed Express will be the final reporter, since it has two important properties. A very long half-life (several days), and is visible to the naked eye. [The second property might not be true for small concentrations of DsRed].
Component: Detector for bacterial secreted AHL
To more accurately simulate a bacterial biofilm, we decided to create bacteria that would constitutively secrete AHL.
We might need to clone this part in if the biofilm E. coli strain doesn't produce AHL by itself...
Component: Amplifier for reporter production
Our detector plasmid would incorporate the Hrp system as an amplifier, to amplify a reporter gene yet to be decided.

The characterisation of the Hrp System will be carried out along with this project, and thus will only be incorporated into the infector detector once the system has been fully characterised.
It has now been shown that the amplifier system will most likely not work. This part of the project has been cancelled.
Construct 2: Detector for AHL
From our models, it has been shown that the construct 1 does not respond uniformly, since the activator protein LuxR is produced by a promoter and not maintained at a constant level. This makes it hard to relate the output of the system to the AHL input. We decided to add purified LuxR to the system instead, and simplify the construct by removing the constitutive LuxR production.