Biomod/2012/DNA Maestros:Methods

Home Introduction The Project Materials & Methods Results and Discussion Conclusion Team Members Acknowledgements
DNA icosahedra were constructed from three distinct five way junction (5WJ) components V, Uapt and L, with programmed overhangs (see Table 1) [1]. Each 5WJ module V, Uapt and L was constructed from equimolar ratios of the respective five 5´ phosphorylated single strands. 5WJ V formed a 1:5 complex with Uapt to give VU5apt. The complementary module VL5 can be synthesized from components V and L in a 1:5 ratio. Post-annealing, the hybridized strands in VU5apt and VL5 give rise to our chemically responsive icosahedron. The ten identical overhangs present in VU5apt are also complementary to the overhangs in VL5. Post annealing, VU5apt and VL5 complex with each other in a 1:1 ratio to yield the chemically sensitive DNA icosahedron. Detailed synthesis protocols of each step are given below.
Phosphorylated Oligonucleotide Sample Preparation
Unmodified oligonucleotides were purchased from Sigma, India; whereas oligos with RNA aptamer or fluorescent tag were purchased from IBA, Germany. All oligonucleotides purchased were of HPLC purified standards. Phosphorylation reactions were carried out only for the unmodified oligonucleotides. 1mM stocks of all the oligonucleotides were prepared in milli-Q water. For phosphorlyation reactions, the following procedure was followed. To an eppendorf tube 2 µL oligonucleotide (from 1 mM stock), 10 µL MQ water, 2 µL 10X T4 PNK (Polynucleotide kinase) buffer (500 mMTris-HCl, pH 7.6 at 25°C, 100 mM MgCl2, 50 mM DTT, 1 mM EDTA, 1 mM Spermidine, Bangalore Genei, India) were added. 2 µL T4 PNK enzyme was added to this mixture. The concentration of enzyme used is 10 U/µL. 4 µL of 1 mM ATP was added to the above solution, vortexed for 1 min to mix the solution well and the tube was incubated at 37°C in a heat block for 1 h. Post incubation, the enzyme was deactivated by heating the mixture to 75°C for 10 min. The DNA was precipitated by addition of 80 µL absolute ethanol and 20 µL 3M potassium acetate, and incubated at -20°C for 8 h. Post incubation at -20°C, the tubes were spun at 14000 rpm for 40 min at 4°C. The DNA pellet was visible at the bottom of the tube. All supernatant solution was carefully removed and discarded. The pellet was washed with 100 µL of 70% ethanol and spun at 14 k for 2 min. All ethanol was removed carefully and the resultant pellets in the eppendorfs were air dried for 15 min to remove any remaining ethanol. The pellet was dissolved in 20 µL nuclease free water (Bangalore Genei, India) and the phosphorylated oligonucleotides were quantified by their UV absorbance at 260 nm.
Synthesis of NCI
For all the steps involving ligation of half icosahedra, VL5, VU5, VU5FAM, VU5apt etc we have used NCI as ‘chemical superglue’[2, 3]. NCI has been synthesised in the lab using the following procedure to: 5.5 g Cyanogen bromide (BrCN) and 3.2 g Imidazole are dissolved in 25 mL and 50 mL of dry benzene respectively. A solution of 5.5 g BrCN is added drop wise with stirring to a solution of 3.2 g imidazole in 50 mL benzene. The reaction mixture is warmed to 50°C during the addition and for 5 min after the addition is done. The reaction mixture is cooled at 4°C for 8 h. This may be preferably left for 12 h. The resultant yellow solid is filtered through Whatman filter paper and the supernatant solution is collected. The filtrate is concentrated to dryness under reduced pressure. White crystalline solid obtained is collected and purified by sublimation. The sublimate is pure N-Cyanoimidazole that is aliquoted in eppendorf tubes and stored at -20°C.
5 way junctions (5WJ)
From the stocks of phosphorylated oligonucleotides for V 5WJ, the oligonucleotides V1, V2, V3, V4, V5 were mixed together in a 1:1:1:1:1 ratio and the final volume was made up to 50 µL by addition of buffer (10 mM Phosphate buffer, pH 7.0, 100 mMNaCl, 1mM MgCl2). The final concentration of each oligonucleotide in 50µL of reaction volume was 20µM. In a similar manner, 5 WJ of Uapt and L were made from oligonucleotides U1, U2apt, U3apt, U4 and U5 and L1, L2, L3, L4,L5 respectively. Post addition of all solutions, the reaction mixture was heated to 90°C for 15 min. After 15 min, the sample was annealed from 90°C at the rate of 1°C/3 min till room temperature, incubated at room temperature for 2 h and then stored at 4°C for 48 h. The individual 5WJ were characterized by 15% PAGE stained with EtBr.
Half icosahedra (VU5 and VL5)
In an eppendorf tube, 50 µL of U5WJ (20 µM) and 10 µL of V 5WJ (20 µM) were mixed to form half icosahedron VU5. The eppendorf tube containing this reaction mixture was heated at 45°C for 4 h, and then the temperature was decreased at the rate of 1°C/3 min till room temperature, where the samples were incubated for 2 h and then stored at 4°C for 72 h. VL5 was similarly made by mixing V and L 5WJs. The half icosahedra were ligated using N-Cyano imidazole (NCI) to increase their stability. The excess residual NCI and nickel phosphate (by-product) was washed by passing through a CentriconTM with 10 kDa cutoff till all the solid precipitate disappears. The half icosahedra were characterized by 8% PAGE stained with EtBr.
Construction of the FAM-labeled half-icosahedron(VU5FAM)
We used the standard half icosahedron, VU5 that carried ten identical single stranded overhangs 10 bases long. VU5 (10 μM) was annealed with a complementary 10 base oligonucleotide carrying a Fluorescein label at its 5´ end (FAM-10-mer-oligo, 100 μM) in a total reaction volume of 50 μL. This results in the duplexation of the ten single stranded overhangs of VU5 with ten FAM labelled oligomer forming VU5FAM. This was then used for fluorescence assays described in section 10.1.
Construction of the aptamer modified half-icosahedron (VU5apt)
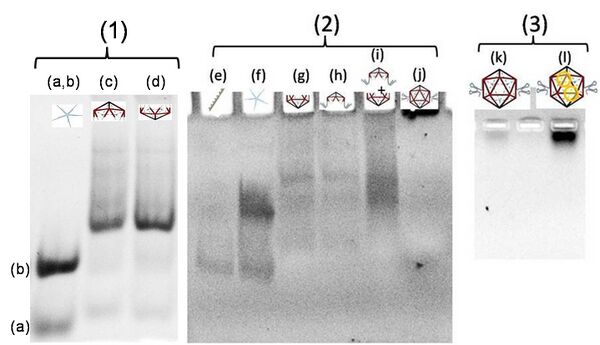
VU5apt is assembled as similarly to standard VU5, except for a slight modification. We use V 5WJ and a modified U 5WJ (Uapt5WJ) for the construction of VU5apt. The V5WJ was assembled by annealing equimolar ratios of V1, V2, V3, V4 and V5) as published [1]. The Uapt5WJ was formed by annealing equimolar ratios of U1, U2apt, U3apt, U4 and U5 (final concentration 20 µM in a reaction volume of 50 µL). Here, the 5’ ends of U2apt and U3apt have been modified to incorporate the RNA aptamer to cdGMP. The sample was annealed from 70°C to 20°C (for Uapt 5WJ) and 90°C to 20°C (for V 5WJ) at the rate of 0.33°C/minute, followed by incubation at room temperature for 8 h, and subsequent incubation at 4°C for 48 hours. The formation of 5WJs was confirmed on EtBr stained 15% polyacrylamide gels (Figure 2.1,a-d). The annealed 5WJs were then mixed in 1:5 ratio of 5WJs V:Uapt such that the concentration of VU5apt finally is 3.33µM in 60µL reaction volume. The mixture was then annealed from 45°C to 20°C at the rate of 0.33°C/minute, equilibrated at room temperature for 8 h, followed by incubation at 4°C for 48 h. This reaction mixture was then subjected to NCI ligation as previously described [1]. Post ligation, the residual NCI and by products were dialyzed out and the ligated VU5apt was purified and concentrated in a CentriconTM with a 10KDa cut off, and the final concentration of VU5apt was determined. The formation of VU5apt was confirmed on EtBr stained 8% polyacrylamide gels (Figure 2.2,g-j).
Construction of the FAM-labeled VU5apt (eVU5FAM)
10 µM of VU5apt was incubated with 100 µM complementary FAM labelled 10-mer DNA strands (FAM-10-mer) in a total volume of 50µL at 45°C for 4 h and annealed to 20°C at 0.33°C/minute, incubated at room temperature for 8 h and 4°C for 48 h to give VU5apt carrying ten FAM labels (eVU5FAM). This was then used for fluorescence assays described in section 10.3.
Regular Icosahedron (I)
25 µL of ligated VU5apt and VL5 (3.33 µM each) were mixed in an eppendorf to form icosahedra. The reaction mixture was heated in a heating block at 45°C for 4 h, and the temperature was decreased at a rate of 1°C/3 min till room temperature followed by incubation at 20°C for 2 h. Then the sample was transferred to 4°C to equilibrate for 72 h. The half icosahedra and full icosahedra were characterized on 0.8% agarose gel (Figure 2 (2)).
Formation of FD10 loaded engineered Icosahedron: eIFD10
A typical procedure to make an FD10-loaded, full icosahedron engineered to be sensitive to cdGMP (eIFD10), was done on a total sample volume of 50 µL that contained 1.65 µM VU5apt, 1.65 µM VL5 and 2 mM FD10. The sample was incubated at 40 °C for 15 min, annealed to 37°C at the rate of 0.3°C /min, held at 37°C for 2 h, annealed till 20°C at the rate of 0.3°C /min, incubated at 20°C for 8 h, and then equilibrated at 4°C for 48h.
Two types of fluorescently labelled eIFD10 complexes were made. One of these, eIFD105AX had five Alexa 647 acceptor fluorophores on the icosahedral scaffold, and made from VU5apt where the strand U1 was labelled with Alexa 647 at 5´ end. The second was eIFD1011AX carrying eleven Alexa fluorophores on the icosahedral scaffold. Here the V 5WJ used to construct both VU5apt and VL5 had strands V1, V2 and V3 with 5´ Alexa 647 labels. In addition, VU5apt was made from U 5WJ, that had U1 labelled with Alexa 647 at 5´ end.In both cases, eIFD1011AX and eIFD105AX were not subjected to ligation and the mixture was purified from free FD10 using a CentriconTM of 30 kDa molecular weight cut-off in order to separate free FD10 from the loaded eIFD1011AX or eIFD105AX as relevant.
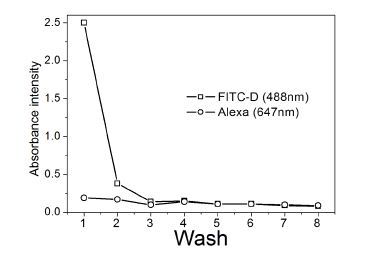
To 50 µL of eIFD105AX containing 2 mM FD10, 150µL of phosphate buffer was added and centrifuged at 3000 rpm at 4°C. The flow-through fractions obtained after subjecting to a CentriconTM, that contained eluted free FD10 was monitored spectroscopically to determine the efficiency of FD10 separation. To this, 200 µL of phosphate buffer was added and the procedure repeated till three consecutive washes showed negligible fluorescence (Figure 4). The wavelengths monitored for each component were 488nm as a measure of FD10, and 647 nm as a measure of any disintegrated DNA capsule. The purification traces showed efficient purification and stable nanocapsules.eIFD1011AX or eIFD105AX were then used further for fluorescence investigations outlined in section 10(iii).
Fluorescence Assays
Strand displacement reporter assay
A sample of VU5FAM prepared as described in section 5, was equally divided into two tubes (25 μL each). In one tube, equimolar amount (20 μM, 25 μL) of lower half icosahedron (VL5) was added and in other tube an equal volume (25 μL ) of buffer (10 mM Phosphate buffer, pH 7.0, 100 mM NaCl, 1mM MgCl2) was added. Both samples were annealed using step wise temperature reduction at the rate of 0.33°C/minute from 45°C till 37°C , incubated at 37°C for two hours and then further reduction in temperature at the same rate till about 20°C. It was further incubated at 20°C for two hours, and then kept at 4°C till further use. Post-annealing and incubation, and before recording of data, the sample was made upto 400 μL with phosphate buffer such that the final concentration of FAM-10-mer-oligo was maintained at 200 nM. To each tube free TMR was added to a final concentration of 100 nM as an internal control. The spectra of each sample were recorded using Fluorolog 3L instrument (Horiba Jobin Yvon) by exciting at 488 nm and 547 nm for FAM and TMR respectively and collecting the intensities at 515 nm and 596 nm respectively. For all samples, intensity at 515 nm was normalized to the intensity at 596 nm.
Strand displacement by the Aptamer in presence of cdGMP
A sample of eVU5FAM as described in section 7 (total volume 50µL) was divided equally into two tubes (25µL each). Into one tube, cdGMP was added and made up to a total volume of 50µL and final concentration of 100 µM cdGMP.In the other tube equal volume of phosphate buffer was added. The tubes were incubated at 37°C for 2 h for efficient completion of the cdGMP binding to aptamer. For fluorescence measurements, the samples were made up to 400 µL. This also gives a final concentration of FAM close to 200 nM. In both the tubes, an equal volume (1 µL) of free TMR to give a 100 nM concentration was also added as internal control. Fluorescence spectra of each sample were recorded using Fluorolog 3L instrument (Horiba Jobin Yvon) equipped with a Peltier temperature controller. Samples were excited at 488 nm and 547 nm for FAM and TMR respectively and intensities were collected at 515 nm and 596 nm respectively. For all samples, intensity at 515 nm was normalized to the intensity at 596 nm.
FRET assay to monitor cargo release from eIFD1011AX or eIFD105AX
The purified sample of eIFD1011AX or eIFD105AX prepared as described in section 9 was divided in two tubes, made up to 400 µL with phosphate buffer, which should typically give a FAM label concentration near 100 nM. In one tube cdGMP was added to give a final concentration of 100 µM and in other tube an equal volume of phosphate buffer was added. Both the tubes were incubated at 37°C for 2 h, and then the fluorescence from both the samples was measured by exciting FITC at 488 nm and collecting the donor fluorescence intensity (D) at 515 nm as well as acceptor intensity (A) at 570 nm. The data are presented as the ratio between fluorescence emissions at 515 and 570 nm as a measure of Försters resonance energy transfer.
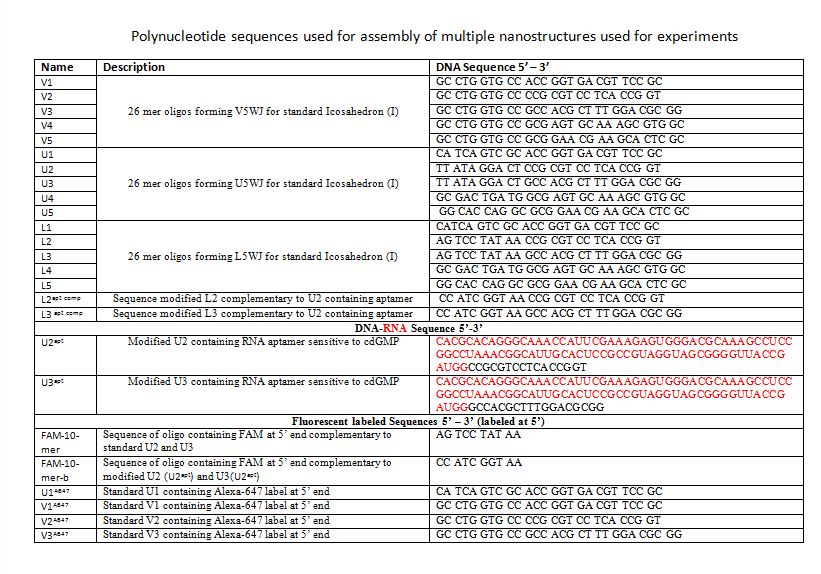
Polynucleotide sequences used for assembly of multiple nanostructures used for experiments
References
-
Bhatia, D., Mehtab, S., Krishnan, R., Indi, S. S., Basu, A., Krishnan, Y. Icosahedral DNA nanocapsules by modular assembly. Angew. Chem. Int. Ed. 48, 4134-4137 (2009).
-
Luebke, K. J., Dervan, P. B. Nonenymatic ligation of double-helical DNA by alternate-strand triple helix formation. Nucleic Acids Res. 20, 3005 –3009 (1992).
-
J. Qi, R. H. Shafer. Covalent ligation studies on the human telomere quadruplex. Nucleic Acids Res. 33, 3185 – 3192 (2005).

