BME100 s2018:Group7 W0800 L5
| Home People Lab Write-Up 1 | Lab Write-Up 2 | Lab Write-Up 3 Lab Write-Up 4 | Lab Write-Up 5 | Lab Write-Up 6 Course Logistics For Instructors Photos Wiki Editing Help | ||||||
OUR TEAM
LAB 5 WRITE-UPPCR Reaction ReportThe pre-lab write up was very helpful in getting the information for the lab ahead of time to make sure that the lab made sense and would be quicker to carry out later on. Yes, the first step involves sucking up the liquid with the pipettor, you have to push the button down and release once the tip is in the bottom of the liquid. The second step involves releasing the liquid from the pipettor, you have to push the button down as far as it goes to insure that all of the liquid was released. The final reactions had about the same amount of liquid as when they were placed in the machine. When taking the liquid from the tubes, the pipettor was placed just above 50 microliters to ensure that all of the liquid was removed. Our labeling scheme worked for the procedure, we made sure to label what number trial it was and what sample within both trials so they could be easily identified. Fluorimeter ProcedureImaging set-up First the phone being used was placed into the holder making sure it was propped up to stand straight. Then the sample holder was raised using a stand to make the sample parallel to the camera lens for accurate pictures. Finally, the box was placed on top of that making sure that it was facing the right way so that the flap on the outside could be easily opened when changing samples and easily closed when taking pictures.
Placing Samples onto the Fluorimeter
Data Collection and AnalysisImages of High, Low, and Zero Calf Thymus DNA   
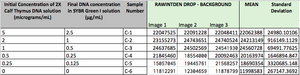 TABLE GOES HERE
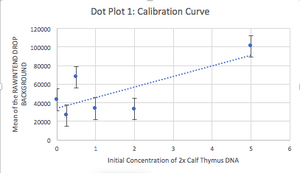 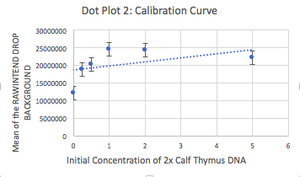 Images of Our PCR Negative and Positive Controls 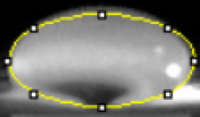 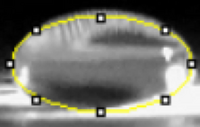 PCR Results: PCR concentrations solved TABLE GOES HERE 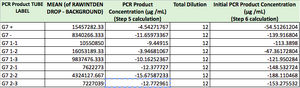 PCR Results: Summary
| ||||||


