BME100 s2018:Group5 W1030 L1
| Home People Lab Write-Up 1 | Lab Write-Up 2 | Lab Write-Up 3 Lab Write-Up 4 | Lab Write-Up 5 | Lab Write-Up 6 Course Logistics For Instructors Photos Wiki Editing Help | |||||
|
OUR TEAM
LAB 1 WRITE-UPHealthcare IssueNasal congestion is one of the most common and frequent complaints that humans have. Various things cause the irritation or constriction of the nasal cavity such as (not all causes): >Colds, Influenza, Sinusitis >Nonallergic Rhinitis/ Vasomotor Rhinitis (VMR) >Allergies >Dry Air (Mayo Clinic Source)
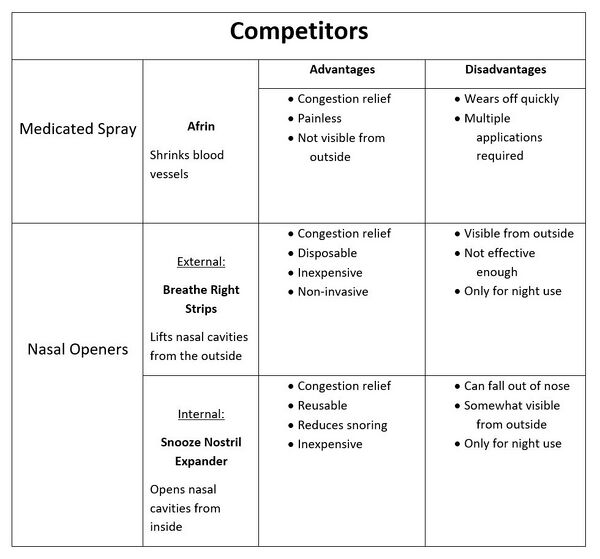
Competitors to Oxyspan
Customer ValidationPatients:
People suffering from Nasal Septum Deviation (80% pop.), Snoring (25% pop. habitually), Sinusitis (12.1% pop.), Nasal Turbinate Hypertrophy.
People with congestion from the Common Cold/Flu, allergies, and dry air.
Existing Patents1. Nasal spray composition and method for treating rhinitis, sinusitis or both Patent Number: WO2008070264A3 Url: https://patents.google.com/patent/WO2008070264A3/en?q=afrin&oq=afrin Original Assignee: Kent A Knauer Univ Case Western Reserve Original file date: 2008-06-12 Summary: This patent has claims on the use of a solution containing oxymetazoline in the treatment of nasal congestion, rhinitis, and sinusitis. The decongestant is administered through the nostril by means of a spray.
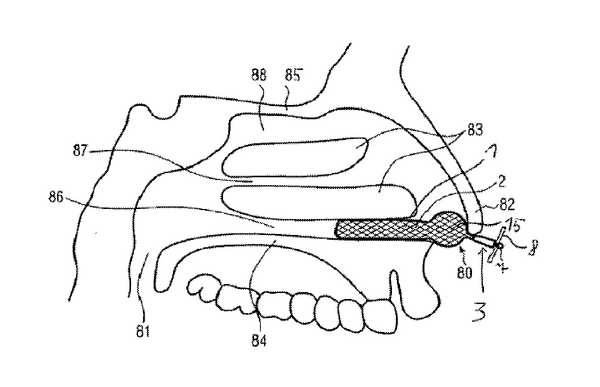
2. Stent for splinting a nasal passage Patent number: US20140018839A1 Url: https://patents.google.com/patent/US20140018839A1/en?q=nasaline&oq=nasaline Original Assignees: Peter RennerKlaus DüringJoachim Georg PfefferNasib Dlaikan-Campos Original file date: 2014-01-16 Summary: This patent makes claims to an insertable nose stent. Specifically it claims the elongation ability of the material and the wired braid lattice. It also claims the procedure for insertion. ReferencesC. (2018, January 03). Snoring and Sleep Apnea. Retrieved January 24, 2018, from http://www.entnet.org/content/snoring-and-sleep-apnea C. (2018, January 03). Stuffy Nose. Retrieved January 24, 2018, from http://www.entnet.org/content/stuffy-nose Chronic Sinusitis. (2017, March 31). Retrieved January 24, 2018, from https://www.cdc.gov/nchs/fastats/sinuses.htm Deviated Septum Relief - Nasal Aid Relieves Deviated Septum | Max-Air Nose Cones ® | Sinus Cones ® the best nasal cones to relieve deviated septum, stop snoring, remedy sinusitis, and relieve blocked noses. 1-800-797-036. (n.d.). Retrieved January 24, 2018, from https://www.maxairnosecones.com/deviated-septum-relief/ Nasal congestion: More than a stuffy nose. (2018, January 11). Retrieved January 24, 2018, from https://www.mayoclinic.org/symptoms/nasal-congestion/basics/causes/sym-20050644 RespiFacile® a reusable, Hi-Tech nasal dilator to improve your breathing. (n.d.). Retrieved January 24, 2018, from http://www.respifacile.com/reusable-nasal-dilator What is AIRMAX™ Nasal Dilator? (n.d.). Retrieved January 24, 2018, from https://airmaxnasaldilator.com/ “The AlaxoLito Nasal Stent.” Alaxo GmbH: New Stent- Based Therapies for Treatment of Snoring, Sleep Apnea and Decreased Nasal Breathing, www.alaxo.com/alaxolito_eng.html.
| |||||