20.109(F15):FACS analysis (Day7)
Introduction
FACS stands for "Fluorescence Activated Cell Sorting." The FACS machine has revolutionized biology by allowing researchers to isolate cells based on their spectral qualities. For example, if you have a fluorescently tagged antibody that preferentially binds to a certain cell type, you can isolate a pure sample of this cell type from a complex mixture by using a FACS machine. In addition to purification, the FACS machine can count the number of cells that have a certain spectral quality. If a FACS machine is used just for counting and not for separating subpopulations of cells, then the procedure is called "flow cytometry," and this is what you will be doing today.
Before there were FACS machines, there were Coulter counters. Coulter counters are automated cell counting machines developed in the 1950s that count cells as they flow in a liquid stream. In an ingenious conceptual leap, Mack Fulwyler combined the technology of ink jet printers with that of Coulter counters to develop the first FACS machine. The ink jet printer head works by vibrating a nozzle so that a spray of discrete droplets is formed. Similarly, in a FACS machine, a liquid suspension of cells is forced at high pressure through a vibrating nozzle to create tiny charged droplets, each containing a single cell. The stream of droplets passes in front of a laser beam, and the scattered light is analyzed by a series of filters and photomultiplier tubes that convert the light signal into electrical impulses. Thus, each cell is "interrogated."
For FACS, the spectral qualities of the cell are analyzed nearly instantaneously and compared to your desired spectral qualities. For example, if you have a mixture of green fluorescent cells and non-fluorescent cells, you can ask the machine to isolate the green cells. If a cell registers as green, an electrical charge deflects the cell to make it fall into a collection chamber.
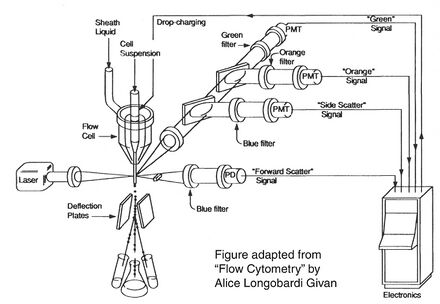
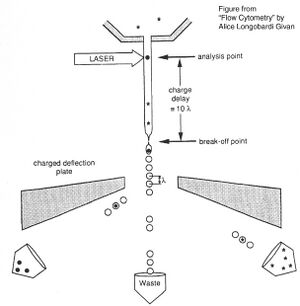
FACS is technically challenging and most FACS machines are only run by experts. In contrast, biologists are often trained to perform flow cytometry in order to analyze the proportion of their sample that has particular spectral qualities. You will be using flow cytometry to measure the percentage of cells that are fluorescent. You have lipofected cells with two non-functional EGFP genes. Recombination between these two genes can restore the full length EGFP coding sequence so that cells express EGFP. By measuring the percentage of cells that fluoresce green, you will have some measure of the frequency of homologous recombination within mammalian cells.
Protocols
Preparing your cells for flow cytometry
The following protocol should be performed in the sterile hood through step 6. Step 7 can occur partially outside the hood, depending on your workflow.
While one partner performs steps 1 and 2, the other partner may label 17 eppendorf tubes and put them on ice. Or, the other partner may simply observe and make reminders about the protocol.
- Aspirate the media from your 17 wells of cells, tipping the dish slightly and applying vacuum at the edge of each well until the liquid is just gone.
- Be sure to clean the Pasteur pipet between independent samples, either by briefly dipping in 70% ethanol, or by putting a fresh yellow tip on the end.
- You do not need to clean the pipet between duplicate wells.
- Finally, be sure not to over-aspirate and start sucking up cells.
- Gently wash each well with 1 mL PBS, aliquotted with a 10 mL pipet.
- In other words, don't blast the liquid right at your cells.
- Add 200 μL of trypsin to each well with a P200. Toward the last addition (4ish wells left), the partner who is not aspirating should start a 1’ timer. After the last addition, briefly rock the plate in each direction to distribute the trypsin over the cells.
- When the timer goes off, aspirate the trypsin, starting with the first well to which you added trypsin.
- Only a trace sheen of liquid, all but imperceptible, should remain.
- Now incubate the cells at 37 °C for precisely 10 minutes, according to your timer.
- Label and ice your tubes now, if you haven't already.
- For simplicity of later data collection and analysis, please label your tubes numerically according to scheme below.
- Quickly distribute 200 μL OptiMEM to each well with a P200 to keep the cells "happy."
- Now resuspend each well and transfer it to a labeled eppindorf tube, one at a time. Here use your P200, thoroughly pipetting in order to break up aggregates and make a homogeneous cell suspension. After you fill each tube, transfer it to ice.
- Take a moment to figure out a good workflow with your partner here.
- Keep your tubes on ice as you walk to the flow cytometer in 16-378.
- Be sure to take 17 FACS tubes (with strainer caps) when you go to the flow cytometer.
Sample scheme
- mock
- EGFP
- Δ5
- uncut Δ3
- cut Δ3
- Δ5+uncut Δ3-1
- Δ5+uncut Δ3-2
- Δ5+uncut Δ3-3
- Δ5+cut Δ3-1
- Δ5+cut Δ3-2
- Δ5+cut Δ3-3
- Exp A-1
- Exp A-2
- Exp A-3
- Exp B-1
- Exp B-2
- Exp B-3
Flow cytometry analysis of transfected cells
Carefully observe the flow cytometer during your run. Be sure to ask questions if you don't understand the purpose of any step. In particular, you want to understand all four axis parameters and both gates (R1 and R2). Ultimately, you want to compare the percentages of green fluorescent cells in different samples.
Navigation links
Next day: Module 2 starts: Evaluate mutations and site-directed mutagenesis
Previous day: Lipofection