20.109(F14): Mod 2 Day 5 Assessing re-tuned system
Assessing Re-tuned System
Introduction
Today is a day when you'll start to collect information about the mutant candidates you've chosen. Over the next 3 lab sessions, you will look at
- the expression level of the mutant Cph8 proteins relative to the wildtype light sensor
- the DNA sequence of the region around the K+P- hotspot
- the β-gal activity of the mutant photography strains grown in the light and dark
- the resulting bacterial photograph taken with your mutant strain
The data should be informative, though preliminary.
Cph8 Library
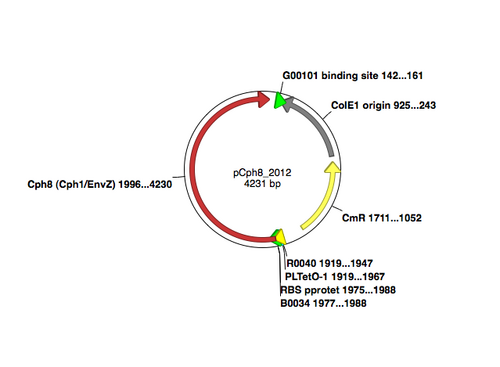
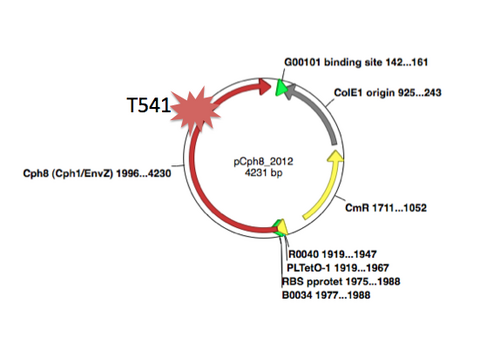
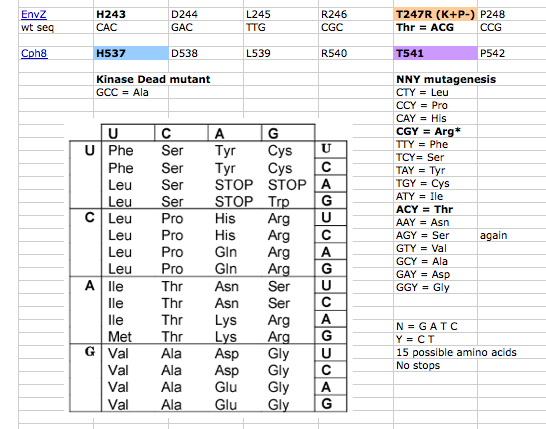
A map of the plasmid that encodes the light-sensor protein, Cph8, is shown. To make the library that we used last time, the plasmid was modified with a degenerate oligonucleotide. The degenerate oligo was synthesized with a mixture of nucleotides at the K+P- relevant position T541, as shown below. PCR was used to extend the degenerate oligos to incorporate them into the pCph8 backbone. Thus PCR products are a mixture of different sequences since the initial oligo template that was PCR amplified had a variety of sequences. Fifteen possible amino acids could be encoded at T541 if the oligo is responsible for the changes we see. It's important to remember that PCR itself has a low but detectable error rate, raising the possibility that mutations outside of the K+P- hotspot could lead to the phenotype you screened through. It's also possible that the plasmid itself is not responsible for the phenotype but rather there has been some random change in the cell's genome or in the pPCB plasmid that is responsible. As a class, though, we'll hope that there are enough interesting mutations to consider and learn from.
In advance of this experiment that you're performing, we have constructed a control for you. This control is a "kinase dead" mutant, H537A. Since the histidine that would normally be phosphorylated in the phosphorelay reactions is now an alanine, the amount of phosphorylated OmpR should be drastically diminished. The bacterial photography system that bears this mutation might have a lower value than wild type in the light and no induction of b-gal in the dark. You will test this candidate along with the wild type strain and the 2 mutants you have chosen.
DNA Sequencing
Though you don't know for certain which, if either, of your mutants exhibits detectably different activity in the context of the bacterial photography system, you will send plasmid DNA from both mutants for sequencing. The invention of automated sequencing machines has made sequence determination a fast and inexpensive endeavor. The method for sequencing DNA is not new but automation of the process is recent, developed in conjunction with the massive genome sequencing efforts of the 1990s. At the heart of sequencing reactions is chemistry worked out by Fred Sanger in the 1970’s which uses dideoxynucleotides.
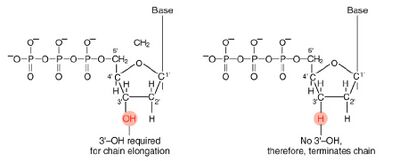
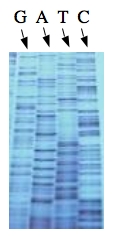
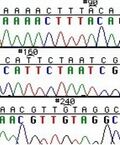
These chain-terminating bases can be added to a growing chain of DNA but cannot be further extended. Performing four reactions, each with a different chain-terminating base, generates fragments of different lengths ending at G, A, T, or C. The fragments, once separated by size, reflect the DNA’s sequence. In the “old days” (all of 30 years ago!) radioactive material was incorporated into the elongating DNA fragments so they could be visualized on X-ray film (image on left). More recently fluorescent dyes, one color linked to each dideoxy-base, have been used instead. The four colored fragments can be passed through capillaries to a computer that can read the output and trace the color intensities detected (image on right). Your sample will be sequenced in this way on an ABI 3730 DNA Analyzer.
Today you will isolate plasmid DNA from the cells to send for sequencing. You'll also measure the β-gal activity associated with your mutant candidates. Finally, you will set up new cultures of your mutants for next time, to isolate protein from the cells you grow.
Part 1: DNA analysis and sequencing
You will isolate plasmid DNA from the overnight cultures of the 2 mutant candidates. The miniprep protocol we'll follow is already familiar to you from Module 1. Each sequencing reactions requires 800 ng of plasmid DNA, and 25 pmoles of sequencing primer in a final volume of 15 μl.
- Resuspended your plasmid DNA pellets in 40 ul of sterile H2O. The miniprep'd plasmid should have ~1 ug of nucleic acid/ul but that will be a mixture of RNA and DNA, so we will guess at the amount of plasmid DNA.
- Next you should prepare two eppendorf tubes, one to sequence each of the two mutants. Each tube should have
- 4 ul of your plasmid DNA, mutant candidate or wild type
- 10 ul of sequencing primer NO296 (seq = TCG TCA ACC TCA TTT TGC GCC AG, this has already been diluted 1:20 from the stock).
- 16 ul sterile water
- Pipet this sequencing solution up and down to mix and then transfer 15 ul to one of the tubes in the 8-tube strip we will turn in at the dropbox in the hall for Genewiz. The data will be available for you to examine < one week from today. Keep your fingers crossed.
Part 2: β-galactosidase Assay
You will measure the β-gal activity associated with the wild type, the kinase dead, and the mutant candidates grown in the light and the dark. You should also set up 5 ml overnight cultures for your samples in the dark to measure protein concentrations next time. Review the protocols here and here for details and ask the teaching faculty if you need help. The overnights can be set up in sterile glass tubes using 5 ul of your dark grown overnights to innoculate the fresh media. Calculate the b-gal activity of your samples before you leave today.
Part 3: Writing Faculty
We'll end the day with a discussion about the format of scientific articles, and good writing.
DONE!
For Next Week (Due M2D7)
Please note: this FNW assignment is due on Thursday, 10/30 or Friday, 10/31
- Calculate the length of the Cph8 protein using the DNA sequence that's here. If you don't already have a favorite program for doing this calculation already, try to use the suite of DNA analysis programs that are here.
- Draft your Materials and Methods section for your Module 2 research article. At a minimum, you should describe the strains and plasmids you've used, the mechanics of taking a bacterial photograph, the b-gal assay and the library screen through to the electroporation. You have the option to update the M&M with your experiments from M2D6 if you'd like to, but it is not required. Drafting this section well now will help you write a knockout research article when it's due.
NOTE
If you are giving a journal club talk on M2D8, then the slides for your presentation must be uploaded to the Stellar website that is associated with our class. The presentation order will be determined by the order that your finished slides are uploaded.
Reagents
- for β-gal assay
- Z-buffer
- 0.06 M Na2HPO4
- 0.04 M NaH2PO4*H2O
- 0.001 M KCl
- 0.0001 M MgSO4
- 4 mg/ml ONPG
- 1M Na2CO3
- Z-buffer
- for alkaline lysis miniprep
- Solution I
- 25 mM Tris pH8
- 10 mM EDTA pH8
- 5 mM Glucose
- Solution II
- 1% SDS
- 0.2M NaOH
- Solution III
- 3M KAc, pH 4.8
- Sequencing primer NO296
- 5'- TCG TCA ACC TCA TTT TGC GCC AG -3'
- starts at ~3500 in pCph8
Navigation Links
Next Day: Mod 2 Day 6: Readout DNA Previous Day: Mod 2 Day 4: Journal Club I