Yarn Microfluidics - Roger Dirth
Introduction
"Paper Microfluidics" offers an opportunity to create low-cost microfluidic devices that are straight forward to fabricate and can be used for diagnostics. Like "Paper Microfluidics", yarn microfluidics also uses capillary action to move liquid through material with no need for an external pump. However, using yarn offers some distinct advantages over using paper for microfluidic devices. [1]
Yarn is a continuous length of fibers that are interlaced to increase structural integrity and is used in applications such as textile production, weaving, embroidery, and sewing. Yarn can be broken down into two main categories, monofilament, and multifilament. Multifilament yarn is typically fabricated from five to one hundred fibers that are interlaced and can be categorized as either flat or textured. There are many different types of fibers that yarn can be composed of. Some are hydrophilic with high wettability, while others are hydrophobic with low wettability. The fibers in the yarn itself act as a channel that can contain flow without the added need for structures to act as barriers, as seen in paper devices. Yarn is also a medium that allows for three dimensional flow whereas three dimensional flow in paper devices would require complex stacking and containing methods. Additionally, flow can be directed in a yarn circuit by several methods such as weaving hydrophilic yarn into a hydrophobic substrate, interlacing hydrophobic and hydrophilic fibers, and adding surface treatment to increase hydrophobicity. Overall yarn offers a potentially simpler method for containing and directing flow for microfluidic systems. [1]
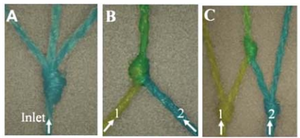
It was found that intersecting fibers could allow for mixing of different liquids carried by the fibers. Various knots have been tested in fibers to show various way to predictably control mixing rates by using different types of knots and tying knots at varying degrees of tightness. Adding knots into fibers can be used to combine, separate, or mix flows, as seen in Figure 1. [2]
Yarn microfluidics has the potential for creating low cost, scalable devices that can be used for application such as point-of-care systems that are highly needed by areas that have less industry for fabricating devices. Furthermore, the use of white fibers allows for colorimetric diagnostics. [3] Overall yarn offers an opportunity to create a low-cost device that can be used for diagnostics in areas that may be limited by cost.
Wicking Rate
The feasibility of using certain fabrics for a microfluidic device often depends on the material's wettability.“Wettability” is a measure of liquid and solid interactions and can be thought of as the wicking rate, or how much liquid moves through a material laterally over time. Factors that can affect a material’s wettability include fiber type, fiber surface properties, gaps between fibers as seen in Figure 2, pores in the fibers themselves, and weaving patterns.
Many commercially available fibers are treated with wax, for ease in sewing, that will give the fiber low wettability. Surface treatments on fibers can be removed to make the fiber more hydrophilic and increase the wicking rate. Methods such as plasma treatment or surface treatment with NaOH followed by immersing the material in an acid solution can remove surface hydrophobic treatments that were done. Plasma treatment was tested for both natural and synthetic fibers and was found to increase wettability for both types of fibers. [1]
Additionally, the wicking rate can be increased by twisting the threads. It was found that twisting the threads to a certain degree compresses the fibers in the thread and lessens flow resistance, therefore increasing the wicking rate. Surface evaporation has also shown to decrease wicking rate when local humidity is high and it is more prevalent when the wetted length is long. [2]
Washburn Equation
One of the challenges of working with fibers is that it can be difficult to model the characteristics of the material. Often many of the models rely on empirical data. However, the Washburn equation (Figure 3) does allow for a simple model to be applied for correlation of the fiber channels. The model however, is not exact, as some fibers will be fully enclosed while others will not. [2]
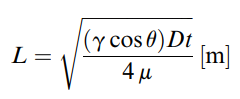
where,
L = wetted length
[math]\displaystyle{ \gamma }[/math] = interfacial tension
[math]\displaystyle{ \theta }[/math] = contact angle between the liquid and yarn surface
D = effective capillary diameter
[math]\displaystyle{ \mu }[/math] = viscosity of the liquid
Fabrication


For fabrication of microfluidic devices made from yarn there have been two common techniques that are often used. The first method uses hydrophilic fibers to create microfluidic channels. These fibers are able to create flow within a channel without the need for barriers as the yarn contains the flow. The yarn can be stitched into a substrate such as a polymer film and the stitch sites are often used as reaction zones. Collectively the loading areas of the reagents and the interlinking of different channels creates a microfluidic circuit out of yarn, such as seen in Figure 4 which shows a serial diluter composed of yarn. Furthermore, knots integrated in the system can act as splitters and mixers which both are demonstrated in the serial diluter (Figure 4). Additionally, altering the tightness of the knots enables to vary the degree of mixing by changing flow resistance within the knot. [2]
The second common method for fabrication uses a weaving pattern of hydrophobic and hydrophilic fibers to direct flow in channels. For example, the wettability of silk which can be increased by first boiling is woven with brass coated silk, that is hydrophobic (Figure 5). Collectively the weaving pattern of the hydrophobic and hydrophilic fibers creates a path that directs the flow.[1]
Applications
Threads and fibers have been used for over a millennium which makes their integration into microfluidic devices much simpler. Countries around the world have already have their unique materials, methods, and tools for creating fibers which gives an opportunity to use a medium that is already widely available. This overcomes the very large challenge of enabling countries restricted by industry or financial ability to create microfluidic devices independently.
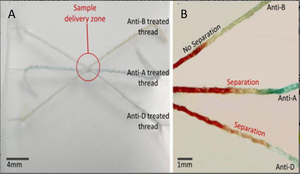
Yarn microfluidics offers a simple method for point of care testing. Research has actively been involved in using yarn for diagnosing diseases such as kidney infection. Tests have been done using colorimetric assays to analyze artificial urine for the presence of proteins, nitrites, and ketones. These can be indicating factors for complications from the kidneys. [4] Research conducted by Ballerini et al. demonstrated yarn as a substrate for a blood grouping test on human blood. This work allowed researchers to successfully determine ABO and RHD groups with only a 2-microliter sample of blood, as seen in Figure 6. [1] [5] Overall showing the potential for threads to carry out biomedical assays.
References
- Nilghaz, A.; Ballerini, D. R.; Shen, W. Exploration of Microfluidic Devices Based on Multi-Filament Threads and Textiles: A Review. Biomicrofluidics 2013, 7 (5), 051501. DOI: 10.1063/1.4820413
- Safavieh, R.; Zhou, G. Z.; Juncker, D. Microfluidics Made of Yarns and Knots: From Fundamental Properties to Simple Networks and Operations. Lab on a Chip 2011, 11 (15), 2618. DOI: 10.1039/c1lc20336c
- Li, X.; Tian, J.; Shen, W. Thread as a Versatile Material for Low-Cost Microfluidic Diagnostics. ACS Applied Materials & Interfaces 2009, 2 (1), 1–6. DOI: 10.1021/am9006148
- Reches, M.; Mirica, K. A.; Dasgupta, R.; Dickey, M. D.; Butte, M. J.; Whitesides, G. M. Thread as a Matrix for Biomedical Assays. ACS Applied Materials & Interfaces 2010, 2 (6), 1722–1728. DOI: 10.1021/am1002266
- Bhandari, P.; Narahari, T.; Dendukuri, D. ‘Fab-Chips’: A Versatile, Fabric-Based Platform for Low-Cost, Rapid and Multiplexed Diagnostics. Lab on a Chip 2011, 11 (15), 2493. DOI: 10.1039/c1lc20373h
