User:Madeleine Y. Bee/Notebook/CHEM-571 2013F/2013/09/03
From OpenWetWare
 Experimental Biological Chemistry: Fall 2013 Experimental Biological Chemistry: Fall 2013
|
|||||||||||||||||||||||||||||||||
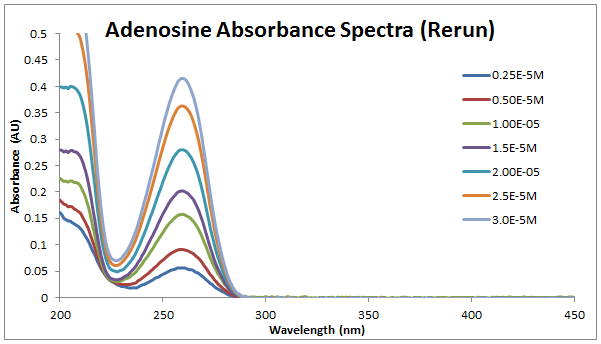
September 3, 2013ObjectiveExecute procedure for making several dilutions of adenosine and inosine solutions, measuring adsorption spectra, constructing calibration curves, performing statistical analyses, and determining concentration of 'unknowns' using calibration curves. Procedure PrepStock Solutions and DilutionsAdenosine MW: 267.24g/mol<br.> Inosine MW: 268.2g/mol<br.> <br.> Original stock solutions:<br.> Adenosine: (from 0.1091g in 50mL volumetric flask) 0.0082M<br.> Inosine: (from 0.1060g in 50mL volumetric flask) 0.0079M<br.> <br.> Dilutions to make, each with total volumes of 1mL:<br.>
DataAbsorption SpectraCalculations and AnalysisDetermine the standard deviation for your data points.<br.> Determine the confidence interval for 90% and 95% confidence.<br.> Determine if any data can be ruled out using a Q-test.<br.> Calibration CurvesMeasurement Notes
| |||||||||||||||||||||||||||||||||