Tomlinson:Research
 Home
News
Research
Publications
Lab Members
Lab Openings
Internal
Home
News
Research
Publications
Lab Members
Lab Openings
Internal
Research GoalsSkeletal fracture is one of the most prevalent and costly medical issues in the United States, with direct costs exceeding $20 billion annually. In fact, some studies have shown that women over the age of 75 would rather be dead than suffer the consequences of a hip fracture. As a result, the overall goal of the Tomlinson lab is to determine novel mechanisms that augment bone strength and induce the rapid repair of skeletal fractures. In particular, the lab focuses on inflammatory signaling in the skeletal response to mechanical forces and damage using small animal models. The Role of NGF-TrkA Sensory Nerve Signaling in Bone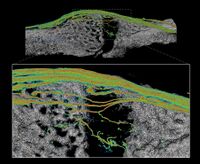 Although sensory nerves are known to be present in mature bone, relatively little is understood about their function in the skeleton, aside from pain. In this project, we are investigating the role of NGF-TrkA signaling in sensory nerves. Nearly all of the nerves in bone express TrkA, the high affinity receptor for nerve growth factor (NGF). Furthermore, sensory nerves blanket the surfaces of bone in a mesh-like network, a privileged location for the acquisition of mechanical signals. Using both in vivo and in vitro methods, we have demonstrated that NGF is robustly expressed by mature osteoblasts in response to non-damaging mechanical loads. Inhibition of NGF-TrkA signaling impairs load-induced bone formation whereas administration of exogenous NGF increases relative bone formation rates. These effects appear to be facilitated through altered Wnt/β-Catenin signaling, which we are currently investigating by using mice which lack NGF in the osteoprogenitor and osteoblast lineages. In addition, we have identified a compound which may provide long-term activation of TrkA to increase load-induced bone formation without the painful side effects of NGF. 1. Tomlinson RE, Li Z, Zhang Q, Goh BC, Li Z, Thorek DL, Rajbhandari L, Brushart TM, Minichiello L, Zhou F, Venkatesan A, Clemens TL. “NGF-TrkA Sensory Nerve Signaling Coordinates Vascularization and Ossification of Bone”, Cell Reports 2016; 16(10):2723-35. PMID: 27568565. PMCID: PMC5014649 2. Tomlinson RE, Li Z, Li Z, Minichiello L, Riddle RC, Venkatesan A, Clemens TL. “NGF-TrkA Signaling in Sensory Nerves is Required for Skeletal Adaptation to Mechanical Loads in Mice”, Proceedings of the National Academy of Sciences 2017; 114(18):E3632-E3641. PMID: 28416686. PMCID: PMC5422802 3. Li Z, Meyers CA, Chang L, Lee S, Li Z, Tomlinson R, Hoke A, Clemens TL, James AW. “Fracture repair requires TrkA signaling by skeletal sensory nerves”, Journal of Clinical Investigation 2019 Dec 2;129(12):5137-5150. PMCID: PMC6877307. 4. Fioravanti G, Hua PQ, Tomlinson RE. “The TrkA agonist gambogic amide augments skeletal adaptation to mechanical loading”, Bone 2021 Jun;147:115908. PMCID: PMC8097708. NSAIDs in Stress Fracture Risk and Repair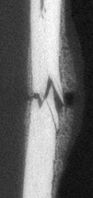 Non-steroidal anti-inflammatory drugs (NSAIDs) are the most commonly consumed medication in the world, with over 30 million daily users in the United States alone. NSAIDs are effective in reducing pain and inflammation by preventing the synthesis of prostaglandin E2 (PGE2) through the blockade of the cyclooxygenase (COX) enzyme isoforms, COX1 and COX2. However, PGE2 is part of an inflammatory signaling pathway that is known to be critical for load-induced bone formation. In collaboration with our partners at the US Army Research Institute of Environmental Medicine and the Rothman Institute, we have identified a clear link between NSAID usage and stress fracture susceptibility. Furthermore, we have observed in mice that NSAIDs may increase stress fracture risk through two independent mechanisms – diminished load-induced bone formation and decreased bone toughness. In addition, we have identified NSAIDs which can provide analgesia without affecting stress fracture risk or repair. We are currently analyzing the mechanisms by which NSAIDs affect the skeleton, with the aim of uncovering novel therapeutic targets for relieving musculoskeletal pain without affecting bone health. 1. Park J, Fertala A, Tomlinson RE. “Naproxen impairs load-induced bone formation, reduces bone toughness, and diminishes woven bone formation following stress fracture in mice.” Bone 2019; 124:22-32. PMID: 30998998 2. Johnston TE, Dempsey C, Gilman F, Tomlinson R, Jacketti A, Close J. “Physiological Factors of Female Runners With and Without Stress Fracture Histories: A Pilot Study.” Sports Health 2020;12(4):334-340. PMID: 32525466 3. Staab JS, Kolb AL, Tomlinson RE, Pajevic PD, Matheny RW, Hughes JM. “Emerging evidence that adaptive bone formation inhibition by non-steroidal anti-inflammatory drugs increases stress fracture risk”, Experimental Biology and Medicine 2021; Feb 27. PMID: 33641442 NSAIDs in Dental Implant Osseointegration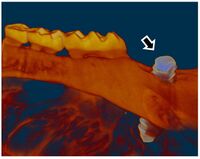 The use of dental implants has transformed the practice of dentistry over the last few decades. The success of this procedure relies on osseointegration, the process by which bone forms a structural and functional connection with the implant over a period of weeks to months. A significant portion of implants fail to osseointegrate, requiring additional surgical intervention. As a result, identifying factors that delay or prevent the osseointegration of dental implants is a top research priority. We hypothesize that use of NSAIDs in the immediate post-operative period may inhibit osseointegration of dental implants. This hypothesis may help explain idiopathic early failure of dental implants, since standard post-operative care often includes immediate and continuous use of high-dose NSAIDs. To directly test our hypothesis, we have assembled a research team of oral surgeons and orthopaedic basic scientists to generate quantitative data from newly placed dental implants as well as analyze bone tissue to ascertain the events that lead to early implant failure. This project is supported by an Osteo Science Foundation grant, in collaboration with our partners in Oral and Maxillofacial Surgery. Bioprinting Vascularized Bone Grafts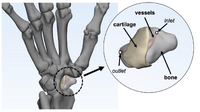 The main cause of large bone graft failure is poor vascularization which leads to inner graft necrosis. Although this issue has been recognized for some time, developing bone grafts with adequate vascular function has proven to be a difficult problem to address and has prevented the widespread clinical use of engineered bone constructs. In this research area, we utilize 3D bioprinting to generate vascularized bone graft using patient-specific imaging data. First, we are developing a new computational tools to provide semi-automated generation of vascular structures within volume generated from standard clinical imaging. Next, we are optimizing the parameters used for extrusion bioprinting to improve outcomes and reproducibility using a type II fuzzy system. Our ultimate goal is to use bioprinting to generate patient-specific vascularized bone grafts for repair of carpal bones and other small bones with complex geometry and articulating surfaces. 1. Sedigh A, DiPiero D, Shine KM, Tomlinson RE. "Enhancing Precision in Bioprinting Utilizing Fuzzy Systems", Bioprinting. 2022 March. doi: 10.1016/j.bprint.2021.e00190. 2. Morrison DG, Tomlinson RE. “Leveraging advancements in tissue engineering for bioprinting dental tissues”, Bioprinting. 2021 Aug;23. doi: 10.1016/j.bprint.2021.e00153. Epub 2021 Jun 13. PubMed PMID: 34268456; PubMed Central PMCID: PMC8276871. 3. Sedigh A, Akbarzadeh-T MR, Tomlinson RE. "Comparison of Type-1 and Type-2 Fuzzy Systems for Mineralization of Bioprinted Bone", bioRxiv. 2021 March. doi: 10.1101/2021.03.31.437908. 4. Sedigh A, Tulipan JE, Rivlin MR, Tomlinson RE. "Utilizing Q-Learning to Generate 3D Vascular Networks for Bioprinting Bone", bioRxiv. 2020 October. doi: 10.1101/2020.10.08.331611. Other Research AreasCheck out our Publications. |