Sysbio:Research
Home Contact Lab Members Publications Research Teaching Lab Calendar Lab Notebook Lab Blog
ResearchNonlinear Biodynamics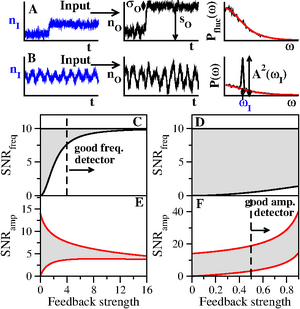 Biological networks control an organism survival and function both at the cellular level (genetic networks) and at a global level(neural networks). As such, they do not work in isolation but are highly interconnected and embedded in a continuously fluctuating and changing environment. Despite the wealth of signals and external variations they have to deal with, they seem to have been exquisitely tuned by evolution to reliably perform specific functions. This leads to some broad questions that we try to address in the lab at different levels: How can biological networks integrate and process information from different signals? How can they operate robustly in the presence of noise and undesirable fluctuations? What are the mechanisms underlying adaptation to environmental context? What is the relation between structure and function of simple biological networks? The advances in experimental techniques and the availability of vasts amounts of data is complementing the traditional molecular approach to research in Biology with a Systems perspective, in which one tries to understand the functioning of a whole decomposing it into simpler 'modules', similar to the engineer design of complex devices. Questions can here be answered at a more quantitative level, and the combination of experiment and mathematical modeling is being proven very fruitful. While this approach has been followed by some time in Neuroscience, it is only recently becoming widely applied in Molecular and Cell Biology. It turns out that one can trace interesting parallelisms in the ways neural and genetic networks process information. We use a combination of mathematical models, numerical simulation and theoretical techniques to investigate some of the above issues in relevant biological circuits. Since networks are not static entities, one of our main tools is non-linear dynamics, together with the theory of stochastic processes. Statistical physics, signal detection or information theory are also applied. We also establish collaborations with experimental groups where theoretical models can be supported by quantitative data. Signaling Networks and Cancer Small molecule inhibitors display significant potential as treatment for diseases and cancer progression involving deregulated signal transduction pathways. These inhibitors are developed based on their target specificity and binding affinity. We focus on the fact that the numerous signaling proteins and feedback loops in signaling pathway strongly influences the efficiency of combinatorial small molecule inhibitor treatments. The existence of several regulatory positive and negative feedback loops either creates complex dose-responses, desensitization to periodic treatments, or modulation of the drug effect in combinatorial treatments. Our experiments show that the effect of inhibitors strongly depends on the particularities of the architecture of the targeted pathway, which must then be taken into account when designing treatments to inhibit pathogen nonlinear pathways.The nonlinear characteristics of the interaction between the proteins ultimately dictates the dynamics and response to drug treatment. Regulation of Stem Cell Differentiation The cellular machinery is governed by interacting proteins, genes and metabolites that form complex and highly interconnected networks of interactions. This way, extracellular stimuli triggers pathways of biological events that regulate gene expression, protein activity, and ultimately, cell response. The transforming growth factor (TGF-ß) pathway is one of the most conserved and prolific of these signaling cascades, involved in a wide variety of both adult and embryonic processes. We use in vivo experiments and theoretical approaches to understand how the wiring of the pathways determines the role of the proteins that regulate neurogenesis. Therefore, to understand the regulation of neuronal formation it is not sufficient to understand the function of each of the proteins in the pathway. A deep understanding of the consequences of the nonlinear wiring of the pathway is required to understand stem cell fate decision and embryogenesis.
Ligand-Receptor Systems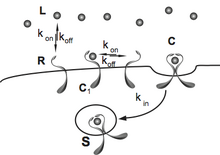 Cells translate extracellular information into internal responses using surface-receptors embedded in the plasma membrane. A cell surface-receptor is a highly specialized integral membrane protein that binds to a specific family of ligands, including cytokines, neurotransmitters, peptide hormones or growth factors. The interaction of ligand and receptor initiates a chain of intracellular events and biochemical reactions referred as signal transduction, leading to physiological changes and regulating essential cell processes, such as differentiation, development, proliferation or apoptosis. The understanding of the regulation of cell-surface receptors and the interaction with their corresponding ligands constitutes an extremely active area of research, mainly due to their pharmacological importance as selective targets for chemotherapeutic agents. At present, receptors-based drugs represent more than 60% of medicines in the market, designed to treat several diseases like autoimmune illnesses, infectious diseases or even cancer. We use a mathematical approach to unveil the consequences of the complex binding process and its relevance in the regulation of the signaling.
Synthetic BiologyWe use a synthetic biology approach to understand basic biological processes, engineering cells to exhibit a desired behavior that allow us to study regulatory mechanism such as cell division, transcription and translation. In detail, we implement nonlinear network motifs in mammalian cells to determine the dynamics of gene interaction networks by studying how introns affect gene expression by increasing the time required to transcribe the gene. We engineered negative feedback loop in animal cells to produces expression pulses, which have a broad time distribution that increases with intron length. These results in combination with mathematical models provide insight into what may produce the intron-dependent pulse distributions. Error in widget YouTube: Unable to load template 'wiki:YouTube'
|


