Mike Barnkob:Protocols/Cloning/Isolating mRNA from spleen
From OpenWetWare
Jump to navigationJump to search
Isolating mRNA from spleen
Protocol on how to isolate mRNA from tissue, such as the spleen.
General notes
Notes:
- A Mouse spleen is approximately 100-160 mg.
- The spin-column will be over-loaded if using too much materiale, which will result in low yield.
- For RNA extraction, use dedicated pipets, a clean lab coat, and filter tips. Spray pipets and surface with RNase Zap.
Lab practice:
- Wear a clean lab coat.
- Change gloves vigorously.
- Use dedicated (or at least very clean) pipets.
- Clean lab bench (it probably needs it too) with 1% SDS and rinse with 70% ethanol.
- Soak all utilities in 1% SDS overnight and rinse with 70% ethanol.
- Do not use microcentrifuge that is also being used for minipreps.
Reagents
For harvesting spleen:
- 5 ml of media in tube.
- Flask of 70% ethanol and empty pipet-tip holder.
- Keep media on ice.
For homogenization:
- 50 ml Falcon tube
- 70μM cell strainer
- D10 media
- 2.5ml syringe
- Red blood cell lysis buffer, Qiagen
For mRNA isolation:
- Qiagen RNeasy Mini Kit
- 70% ethanol
Protocol
Harvest spleen:
- Euthanize mouse by cervical dislocation.
- Dissect out spleen
Note: Wash fur with 70% ethanol to disinfect.
Note: Keep hair out of incision. - Transfer spleen into flask with media, keep on ice.
- In tissue culture hood, cut spleen into smaller pieces (2-4 in total), around 20-30mg per piece.
Homogenize tissue and lyse red blood cells:
- Work in tissue culture hood
- Add 1mL of R10 to 50 ml Falcon tube. Place 70μM cell strainer on tube and add part of spleen on top of this.
Note: The mRNA isolation kit can maximum handle 30 mg of tissue. Aim for 10-20 mg per part.
Note: Left-over spleen can be kept in RNALater media from Qiagen. - Take out plunger of 2.5ml syringe, and mesh spleen through strainer with blunt end of the plunger.
Note: Flush cells through with a little media afterwards.
Note: When done, add up to 5 ml R10. - Spin cells down at 1500 RPM for 5 min and remove supernatant.
- Resuspend in 2 ml of red blood cell lysis buffer and leave on ice for 5 min.
- Add 5 ml of media, spin cells down at 1500 RPM for 5 min and remove supernatant.
- Optional: Resuspend in 1 ml of media and count cells, otherwise continue to mRNA isolation.
Activate cells: (Optional)
- Plate out cells at 10^6 cells/mL in 25 μg/mL LPS to activate cells.
- Incubate cells for 24-48 hours at 37°C
mRNA isolation:
Follows Qiagen RNeasy Mini Kit instructions and presumes all reagents have been prepared.
- (Spin cells down at 1500 RPM for 5 min and remove supernatant)
- Disrupt and homogenize:
- Resuspend in 600μL buffer RLT and pipet up and down several times.
- Transfer to 1.5 Eppendorf tube
- Centrifuge the lysate for 3 min at full speed and transfer the supernatant to new 1.5 Eppendorf tube.
Note: The lysate is normally between 250-600 μL.
Note: When adding RLT, liquid can become viscus, so pipet slowly. - Add 1 volume of 70% ethanol to the lysate, and mix immediately by pipetting (don't vortex).
- Transfer 700 μL of sample to an RNeasy spin column placed in a 2 ml collection tube. Close lid gently, and centrifuge for 15 s at 10,000 rpm. Discard the flow-through.
- Add 700 μL Buffer RW1 to the RNeasy spin column. Close lid gently, and centrifuge for 15 s at 10,000 rpm. Discard the flow-through.
- Add 500 μL Buffer RPE to the RNeasy spin column. Close lid gently, and centrifuge for 15 s at 10,000 rpm. Discard the flow-through.
- Add 500 μl Buffer RPE to the RNeasy spin column. Close lid gently, and centrifuge for 2 minutes at 10,000 rpm. Discard the flow-through.
- Place the RNeasy spin column in a new 1.5 ml collection tube. Add 30–50 μL RNase-free water directly to the spin column membrane. Close the lid gently, and centrifuge for 1 min at 10,000 rpm to elute the RNA.
- Label and store at -20°C.
Quality control:
Using a NanoDrop
- A260/A280: ratio should be close to 1.8-2.0 [1].
- A260/A230: ratio should be close to 2.0 when isolating low amounts of RNA. Values < 1.0 indicates contamination with salts or rests of phenol or protein in the RNA solution.
Gel:
- Make 1% agarose gel
- Mix in Eppendorf tube:
- 4 μL dH20
- 5 μL Blue Loading Dye (6X)
- 1 μL RNA sample
- Run the gel at 100 V until the fastest dye has moved 2/3 of the gel length
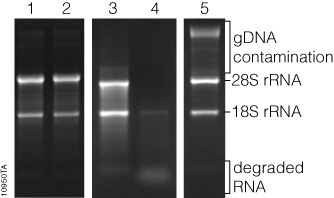
- Visualize gel using a UV transilluminator. A good extraction should have two clearly distinguishable bands, like below:
Storage:
- Purified RNA may be stored at –20°C or –70°C in RNase-free water. Under these conditions, no degradation of RNA is detectable after 1 year.
References
- RNeasy Mini Handbook, p37: http://www.qiagen.com/gb/resources/resourcedetail?id=14e7cf6e-521a-4cf7-8cbc-bf9f6fa33e24&lang=en
- RNA quality control: http://biomedicalgenomics.org/RNA_quality_control.html
- RNA qualitl control using gel electrophoresis: http://www.flychip.org.uk/protocols/gene_expression/rna_qc.php
- Bürgmann et al, 2003: aem.asm.org/content/69/4/1928.full