IGEM:IMPERIAL/2009/Encapsulation/Applications
Summary
Compare against list of specifications to determine feasibility and in order to choose our application
Table 1. Summary of different, targetable diseases of the small intestine
| Enzyme | Condition | Enzyme native location | Function | Genes | Chassis expression | References | Notes |
| Glutaminase | Deficient in Coeliac disease | Liver, Kidney, Small intestine | Deamination of glutamine | ? | Hypothetical sequences expressed? | [1]3 | Human glutaminase doesn't seem to have been expressed in E. Coli |
| Pancrelipase: amylase + trypsin + lipase | Cocktail of enzymes not present in cystic fibrosis, chronic pancreatitis and exocrine pancretitis deficiency | Produced in the pancreas, secreted in the small intestine | Degradation Amylase: Long carbohydrates; Trypsin:Peptides; Lipase:Fats | α-Amylase2A; α-Amylase2B Trypsin; Lipase | E. Coli? | Refs | Sequences don't seem to have been expressed in E. Coli |
| Lactase | Inability to process foods containing lactose. Severity very variable. | Small intestine | Turns lactose in glucose and galactose | LacZ in E. Coli K12;ProteinDataBase | E. Coli | Caltech 2008 | Already tackled by Caltech 08 |
| Anti-inflammatory compounds. Curcuminoids? | Crohn's disease: continuous inflammation of the bowel/ IBS - Irritable Bowel Disease | Found in tumeric. | Anti-inflammatory properties | Biosynthesis pathway expressed in E.coli | E. Coli? | Digestive diseases; Wikipedia; OMIM; Curcuminoid pathway | No cure, cause unknown, very difficult to treat. Curcuminoids are a hypothetical "solution" - no guarantee they will work |
| Phenylalanine hydroxylase (PAH) | Phenylketonuria (PKU) More Info |
Liver, kidney | Deamination of phenyalanine; breakdown to tyrosine | hPAH gene expressable in E.coli | E. Coli | Genetics Home Ref; OMIM; Wikipedia; Dietary info | Affects 1/2600 in Turkey; sufferers must follow a strict diet, managing phenylalanine intake. Any foods rich in protein (milk, fish, eggs, etc) are also rich in phenylalanine so must be avoided. Due to these foods being cut from the diet, special protein substitutes must be taken. This is extremely vital to the sufferer - substitutes must be taken regularly and evenly spread throughout the day. |
- Calderón J, Huerta-Saquero A, Du Pont G, and Durán S. Sequence and molecular analysis of the Rhizobium etli glsA gene, encoding a thermolabile glutaminase. Biochim Biophys Acta. 1999 Mar 19;1444(3):451-6. DOI:10.1016/s0167-4781(99)00026-3 |
Gut
pH in the gut
duodenum - pH 6 to 6.5 It is where most of the nutrients, vitamins, and drugs are absorbed. It is 6 inches long.
jejunum and ileum - pH 7.5
large intestine - pH 5.5-7
Time taken for food to travel
Radioactive labelling is used for this experiment.
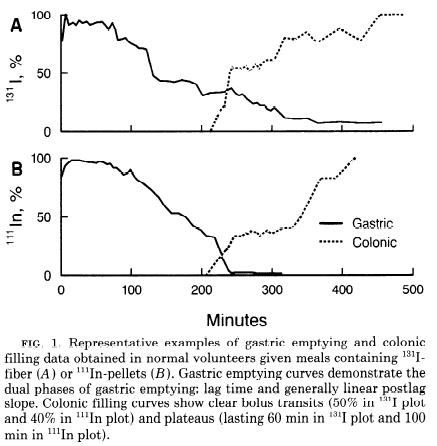 [2]
[2]
- Camilleri M, Colemont LJ, Phillips SF, Brown ML, Thomforde GM, Chapman N, and Zinsmeister AR. Human gastric emptying and colonic filling of solids characterized by a new method. Am J Physiol. 1989 Aug;257(2 Pt 1):G284-90. DOI:10.1152/ajpgi.1989.257.2.G284 |
Reference website Transit time of food
Application 1 - Lactose Intolerance
Lactose intolerance is the inability to metabolize lactose, a lack of lactase.
Occurance
The National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) in the United States estimates that between 30 to 50 million Americans are lactose intolerant. This includes an estimate of lactose intolerance affecting 75% of African-Americans and 90% of Asian and Native Americans.
Very high occurance in Chinese / Asians.
Production
E. Coli has three genes that code for lactase, lacZ, lacY, lacA. There is usually a repressor for the lactase gene in E. coli. However, when there is lactose, the repressor is removed and RNA polymerase binds to the promoter to begin translation and transcription
•lacZ encodes β–galactosidase (cleaves lactose into glucose and galactose)
•lacY encodes β–galactoside permease, a membrane bound transport protein that pumps lactose into the cell.
•lacA encodes β–galactoside transacetylase, an enzyme that transfers an acetyl group from acetyl-CoA to β–galactosides.
- James Chappell 03:40, 21 July 2009 (EDT): Last year Caltech looked into lactose intolerance, it would be interesting to see how they tackeled the problem. Link here to their wiki
Application 2 - Gaucher's Disease
Genetic disease in which lipids/glucocerebroside accumulates in a harmful way to excessive levels in the liver, spleen, lungs, bone marrow and, less commonly, the brain. This accumulation of fatty material in tissues interferes with the normal functioning of the body's organs, and may cause organ enlargement and bone pain. It is caused by mutations in the GBA gene.
Degradation of glycosphingolipids occurs in the lysosome, which contains digestive enzymes. The lysozome breaks down the glycosphingolipid into, fatty acids, sphingosine, and saccharide.
There are 3 types of Gaucher disease.
•Type 1 Gaucher disease most common. The brain and spinal cord are usually not affected. The patient could suffer from enlargement of the liver and spleen, anemia, easy bruising due to a decrease in blood platelets, lung disease, and bone abnormalities such as bone pain, fractures, and arthritis.
People who suffer from Types 2 and 3 Gaucher disease have problems that affect the central nervous system which cause abnormal eye movements, seizures, and brain damage.
•Type 2 Gaucher disease usually causes life-threatening medical problems beginning in infancy.
•Type 3 Gaucher disease also affects the nervous system.
It costs $550 000 annually for enzyme replacement therapy per patient. Another treatment method is bone marrow transplantation, which can cure non-neurological manifestations of Gaucher disease.
Currently, research is being perform on drugs that work on a molecular level, ie pharmacological chaperoning. A drug that has been approved by the FDA is Miglustat. Another drug is in development.
Occurance
It is a metabolic disorder which affects mostly Eastern and Central European (Ashkenazi) Jewish people. Worldwide, Gaucher disease occurs in 1 in 50,000 to 100,000 people.
1 in 10 people in the USA is a carrier for type 1 Gaucher's disease/ Production
Gaucher disease can be treated by enzyme replacement treatment with intravenous recombinant glucocerebrosidase. Studies have shown that glucocerebrosidase can be produced in yeast Pichia pastoris (with non-ideal results) and Chinese hamster ovary cells (CHO cells)
Application 3 - Anti-cancer Anthocynin
Here's a paper describing a study that showed that anthocyanin prevents esophageal tumors in rats, and colon cancer
Occurance
Colon cancer affects approximately 160000 Americans every year [1]. Throat cancer affects approximately 20000 Americans each year [2].
Production
Production of anthocyanin has been characterised in E. coli. paper
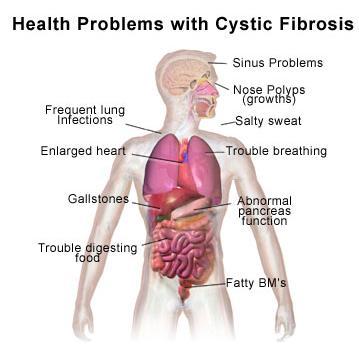
Application 4 - Cystic Fibrosis
Cystic fibrosis is a chronic illness caused by a autosomal recessive genetic disorder that affects the mucus and sweat glands. The patient will usually have thick and sticky mucus, which clogs the lungs, causing breathing problems and making it conducive for bacteria growth. The patient will also suffer from cause chronic coughing and inflammation as a result. Permanent lung damage and the formation of cysts in the lungs will result from the buildup of mucus and various infections. Mucus may also block pancreatic ducts and this prevents pancreatic enzymes from entry into the intestines.
Genetically speaking, cystic fibrosis is caused by a mutation in the CFTR gene. The CFTR gene controls a chloride ion channel responsible for the production of sweat, digestive juices and mucus. The absence of CFTR gene will cause chloride ion channels to malfunction, hence flow of chloride ions and water across cell membranes are disrupted. Cells lining the passageway of lungs and pancreas etc respond by producing a thick and sticky mucus, which clogs airways and glands. As it is an autosomal recessive disease, people who are heterozygous for the CFTR will just be carriers. Approximately 66% of cystic fibrosis is caused by the deletion of three nucleotides which results in the lost of phenylalanine.
This disease cannot be cured, hence management is necessary to prolong the lives of patients. Most patients take a cocktail of medication to fight against infections.
Lung infections are common, and usually antibiotics such as vancomycin and teicoplanin are taken to fight these infections. Teicoplanin is taken intravenously at home. This is not convenient for children as they are dependent on adults to inject them. Furthermore, the antibiotic teicoplanin require reconstitution before it can be used. This makes it extremely troublesome. It can be administered orally as well.
In the case of digestion, as the pancreatic ducts are blocked by thick and sticky mucus, pancreatic enzymes cannot be secreted into the small intestines, and patients will suffer from indigestion. Furthermore, chronic pancreatitis may develop in patients with cystic fibrosis. Patients must take enzymes orally. Currently, pancrelipase is available in the market for the cystic fibrosis patients, as well as patients suffering from other diseases that prevent pancreatic enzymes from being secreted. Pancrelipase is a cocktail of digestive enzymes including lipase, amylase and protease. It is available commercially in capsule, powder or tablet form. Click here for more info on pancrealipase. Digestive problems will lead to diarrhoea, malnutrition, poor growth, and weight loss, hence cystic fibrosis patients have to maintain a high calorie diet, and eat vitamin supplements. Additionally, pancreas produces insulin, pancreatic problems will result in diabetes.
Gene therapy is often used as well.
Occurance
Occurs in 1 in 3300 Caucasians
Production
Biosynthetic gene cluster isolated for E. coli to produce Teicoplanin [3]. 49 genes have isolated. Too complicated!!!
However, it will be much simpler for E. coli to synthesize common enzymes such as lipase, amylase and proteases.
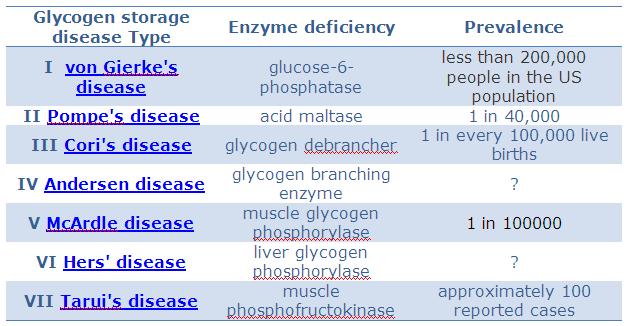
Application 5 - Glycogen storage disease IV / Andersen's disease
Andersen’s disease is caused by defective processing of glycogen synthesis or breakdown within muscles and liver etc. It belongs to a group of metabolic disorders caused by the absence or malfunctioning of lysosmal enzymes needed to breakdown the glucosamonoglycans. Suffers typically either do not produce enough enzymes necessary to break down the sugar chains into simpler molecules , or they produce non-functional enzymes. It is caused by a gene disorder.
Symptoms include early death, accumulation of abnormal polysaccharides, liver insufficiency, abnormal heart and nervous system. Death usually occurs around 5 years of age. Current treatment includes enzyme replacement therapy via intravenous injection.
Too rare. Forget it
Application 6 - Phenylketonuria
Description
Phenylketonuria (PKU) is an autosomal recessive genetic disorder where the body is unable to utilize phenylalanine. This is because of a deficiency in phenylalanine hydroxylase, which converts phenylalanine to tyrosine. Absence of phenylalanine hydroxylase causes phenylalanine to break down into phenylketone. A chronically high level of phenylalanine, called hyperphenylalaninemia will result. Phenylalanine then accumulates in blood and tissues and results in characteristic symptoms of PKU.
In a healthy individual, blood phenylalanine level is about 1 mg/dl. In a PKU patient, levels may range from 6 to 80mg/dl, but are usually greater than 30mg/dl.
More information can be found here.
Genetics
Abstract:
Mutations (>500 alleles) occur in the phenylalanine hydroxylase gene (PAH) on chromosome 12q23.2 encoding the L-phenylalanine hydroxylase enzyme (EC 1.14.16.1). The PAH enzyme converts phenylalanine to tyrosine in the presence of molecular oxygen and catalytic amounts of tetrahydrobiopterin (BH4), its nonprotein cofactor.[3]
- Scriver CR. The PAH gene, phenylketonuria, and a paradigm shift. Hum Mutat. 2007 Sep;28(9):831-45. DOI:10.1002/humu.20526 |
Occurrence
It occurs in 1 in 10,000 to 20,000 Caucasian or Oriental births. Though rare, its symptoms are chronic and patients have to be on a strict diet for the rest of their lives.
Symptoms
Most symptoms of PKU are avoided by screening newborns to ensure early identification, and management.
Symptoms include subtle signs of nervous system function problems, for example, increased muscle tone and more active muscle tendon reflexes. High concentration of phenylalanine in the blood will cause severe brain problems for instance, mental retardation and seizures. If not discovered early, children could develop a small head, prominent cheek and upper jaw bones with widely spaced teeth, poor development of tooth enamel, and decreased body growth.
Since blood phenylalanine levels are dependent on diet. A basal phenylalanine levels is necessary for normal growth and body functions. Low levels can cause mental and physical sluggishness, loss of appetite, anaemia, rashes and diarrhoea.
Management
In the US, blood phenylalanine levels are done on all newborns in every state.
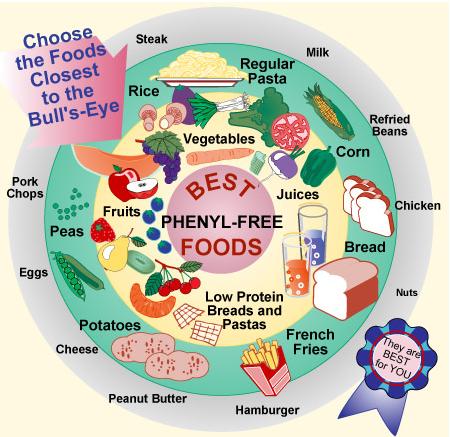
We hope to maintain the blood level of phenylalanine between 2 and 10 mg/dl as phenylalanine is essential for normal growth. The diet of a PKU patient must contain lower than normal amounts of phenylalanine. Foods such as meat, fish, poultry, eggs, cheese, milk, dried beans, and peas, that are rich in proteins are avoided. Instead, measured amounts of cereals, starches, fruits, and vegetables, along with a milk substitute are usually recommended.
Info - Glycogen Storage Disease
Application 7 - Xylanase & Cellulases
Ruminants survive on a diet high plant matter through the breakdown of cellulose and xylan by their gut microflora. Non-ruminants are unable to do this and therefore rely on a diet rich in non-plant matter (e.g. they feed on ruminants).
Of course, the transfer of energy between trophic levels is notoriously poor. If non-ruminants were able to obtain more energy from plant matter, the net effeciency of energy transfer would be raised reducing the volume of food required in a diet. In addition, polysaccharides such as cellulose form viscous gel-like structures that trap starch, proteins and fats which would otherwise be accessible to the animal's digestive enzymes and transport systems.
The following efforts have been made to facilitate digestion of plant matter in non-ruminants.
1) Cellulases are commonly added to bird feed to increase nutritional value however this is expensive.
2) To depolymerise cellulose, the protease resistant endoglucanase E of Clostridium thermocellum was expressed in the exocrine pancreas of transgenic mice.[4] 600ng of the protein would be required to faciliate a 90% reduction in beta-glucan viscosity in mice.
There are two theoretical ways in which these polymers could be used to provide additional nutrients to the consumer.
1) Delivery of acid-stable enzymes to the stomach. Spec: Must be able to withstand pH of 1-3
2) Delivery of alkaline & protease stable enzymes to the intestine. Spec: Must be protease resistant and withstand pH range 6-8
Different enzymes fitting both of these requirements are well characterised.
Protease resistant xylanase:
Gene: SfXyn10
Origin: Streptomyces fradiae var. k11
Expressed in: E.coli
pH stability: 4-10
Protease resistant: Yes
Reference: [5]
Endoglucanase E:
Application 8 - Alternatives
- Plant growth.
- Title:Encapsulation of Plant Growth-Promoting Bacteria in Alginate Beads Enriched With Humic Acid [6]
- Abstract:The key to achieving successful, reproducible results following the introduction of beneficial microbes into soil relies on the survival rate of the inoculated bacteria in a heterogeneous soil environment and hence an improved encapsulation method was developed. Owing to the constraints associated with the inoculum formulation, in this study, encapsulation of a plant growth promoting bacteria (PGPB) isolate Bacillus subtilis CCpg104 was attempted with alginate by enriching the bead microenvironment with humic acid. High viability of the encapsulated bacteria was observed with minimum cell loss upon storage for 5 months. Steady and constant cell release from the bead was observed for 1 week at different pH. Encapsulated cells remained active as evidenced by their ability to solubilize calcium phosphate in vitro. Successful plant growth promotion of lettuce by the encapsulated bacteria under gnotobiotic and sterile environment was also achieved. Feasibility of this improved encapsulation technique is mainly due to the dual benefits of humic acid to microbe and plant and its chemical properties allowing an easy mixing with alginate without interfering in the formation of the alginate gel beads by cross-linking with Ca2þ ions. Thus, the encapsulation method described in this study can be effectively used to protect the PGPB inoculum from adverse conditions of the soil for their successful establishment in the rhizosphere.
- Comments: Just food for thought on alternative applications.
- Nuri Purswani 11:10, 23 July 2009 (EDT):
- General reviews:
- Title:Therapeutic uses of microencapsulated genetically engineered cells [7]
- Abstract:Microencapsulated genetically engineered cells have the potential to treat a wide range of diseases. For example, in experimental animals, implanted microencapsulated cells have been used to secrete growth hormone to treat dwarfism, neurotrophic factors for amyotrophic lateral sclerosis, β-endorphin to decrease pain, factor XI for hemophilia B, and nerve growth factors to protect axotomized neurons. For some applications, microencapsulated cells can even be given orally. They can be engineered to remove unwanted molecules from the body as they travel through the intestine, and are finally excreted in the stool without being retained in the body. This application has enormous potential for the removal of urea in kidney failure, ammonia in liver failure and amino acids such as phenylalanine in phenylketonuria and other inborn errors of metabolism.
- Bacterial microbiology (includes part on capsules)
PKU
Entrez Gene information | Gene information
Glycerol Increases the Yield and Activity of Human Phenylalanine Hydroxylase Mutant Enzymes Produced in a Prokaryotic Expression System |
Paper
PAH expression vector for E. Coli | Paper
Biochemical characterization of recombinant human phenylalanine hydroxylase produced in Escherichia coli | Paper
Prokaryotic expression analysis of I269L and R270K mutations of the phenylalanine hydroxylase gene | Paper
Lactose intolerance
Entrez Gene information | Gene information
A randomized trial of Lactobacillus acidophilus BG2FO4 to treat lactose intolerance | Paper
References
- Calderón J, Huerta-Saquero A, Du Pont G, and Durán S. Sequence and molecular analysis of the Rhizobium etli glsA gene, encoding a thermolabile glutaminase. Biochim Biophys Acta. 1999 Mar 19;1444(3):451-6. DOI:10.1016/s0167-4781(99)00026-3 |
- Camilleri M, Colemont LJ, Phillips SF, Brown ML, Thomforde GM, Chapman N, and Zinsmeister AR. Human gastric emptying and colonic filling of solids characterized by a new method. Am J Physiol. 1989 Aug;257(2 Pt 1):G284-90. DOI:10.1152/ajpgi.1989.257.2.G284 |
- Scriver CR. The PAH gene, phenylketonuria, and a paradigm shift. Hum Mutat. 2007 Sep;28(9):831-45. DOI:10.1002/humu.20526 |
- Error fetching PMID 18388456
-
Encapsulation of Plant Growth-Promoting Bacteria in Alginate Beads Enriched With Humic Acid Chiu-Chung Young, P.D. Rekha, Wei-An Lai, A.B. Arun