Biomod/2017/StJohns
Presented by Team Toehold Conga Nanny
Project Title: Optimization of DNA Origami For Use in Electrochemical Virus Sensors
Abstract
In our long-standing attempt to create a virus sensor, we have improved the design of our virus-binding DNA origami - a “three-armed claw”. We deleted “hinge staples” to reduce stacking and produce tighter, clearer bands on agarose gels; in-gel FRET studies of virus binding are ongoing. MS2 Virus-Like Particle (VLP) degradation was decreased via cryo-protection with trehalose, and we studied the interaction between published aptamers and clinically relevant Norovirus VLPs using Fluorescence anistotropy. Psoralen cross-linking made origami more robust to denaturing conditions found in some sensor systems.
We remodelled and synthesized versions of our control origami (triangle) to display Methylene Blue dyes in different orientations. Once these origami are functionalized with thiols, we will study origami-VLP electrochemical interactions with gold electrode surfaces, paving the way for origami use in electrochemical virus sensors.
Introduction and Theory: DNA Nanotechnology
Watson and Crick Base pairing allows for a nucleotide sequence to assemble in a predictable manner. Used in combination with the DNA double crossover concept1, large macromolecular structures can be assembled into a variety of different patterns and shapes. All assemblies combine in aqueous solution, and rely on the strands to assemble into their thermodynamically most stable configuration.
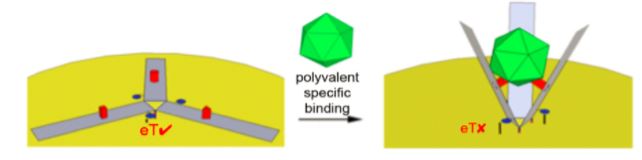
Having such high specificity, DNA nanotechnologists are now able to assemble DNA origami (DO) structures2 to have a set function. This lab sets out to design a DO Claw that would act as a virus sensor allowing for efficient and rapid detection, with a potential future application in diagnostics.
The overall goal of this project is to create a virus sensor, which can detect presence of viruses in solution. The virus sensor would be made up of our DNA origami, a D.O. claw based on Endo’s design3, coupled with a gold surface that would act as an electrode for electron transfer and signal production.
-
Figure 1. Our virus sensor design. Our D.O. claw (gray) is attached to a gold surface and would bind viruses (green).
-
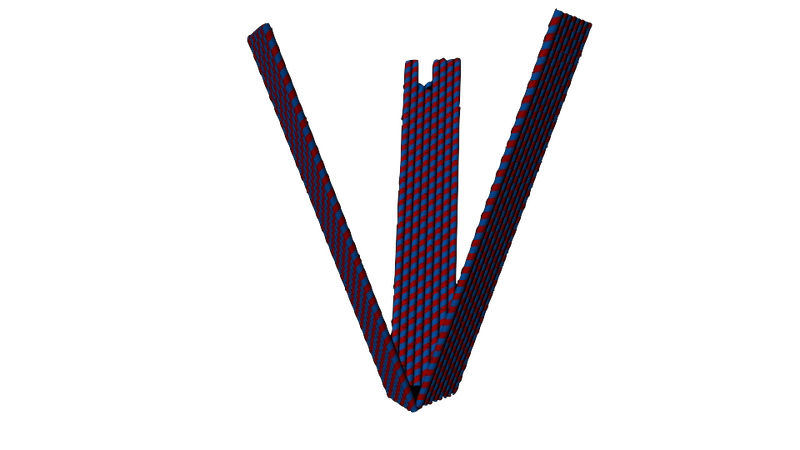
Figure 2. Our D.O. claw design, as seen when closed up. Its arms are 100 nm long and 20 nm wide.
-
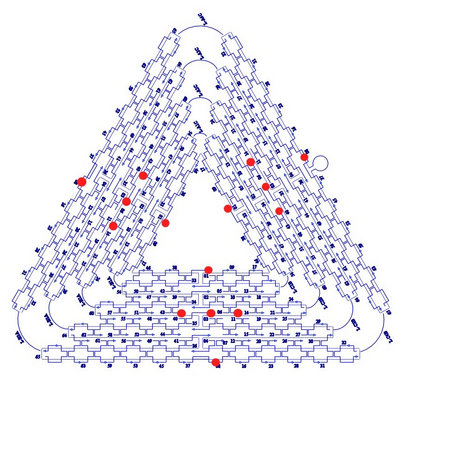
Figure 3. The Rothemund triangle we are using as a control to our D.O. claws.2 The length of the edges are roughly 120 nm. The red dots indicate binding sites.
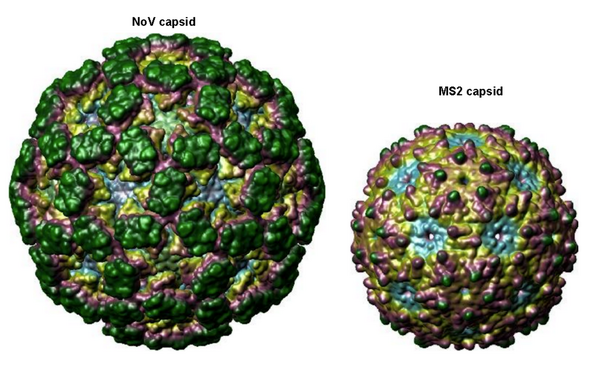
For proof of principle, we are testing the binding capabilities of our claws in solution using Virus-Like-Particles, empty protein shells of the viruses that have been made biochemically. The first of which are derived from bacteriophage MS2 that have been functionalized with T21 ssDNA on its surface by our collaborators (Figure 5). We chose MS2 VLPs as it has been shown to bind to D.O. triangle.4 We are also working with Norovirus (NoV) VLPs as they are more medicinally relevant and interesting; they are one of the leading causes of acute gastroenteritis. Other than being larger in size, NoV is topographiclly similar (i.e. similar suface roughness and charge density) to MS2 (i.e. similar surface roughness and charge density), as seen in Figure 4.
-
Figure 4. Norovirus VLP (left) compared to the MS2 VLP (right). The NoV capsid strains we have used include GI.7, GII.2 (Snow mountain), GII.4 (Grimsby & Houston) and GII.7; displaying a wide variety of clinically-relevant strains.5 The VLP we are using of NoV is based of the GII.4 strain.
-
Figure 5. A basic illustration of our ssDNA functionalized MS2 VLPs. The protein shell is roughly 22 nm in diameter, with the T21 ssDNA being 7 nm in length. Note that the ssDNA and VLP are not to scale.
With the NoV VLPs, we intend to bind a fluorescent aptamer to them, in conjunction with binding of MS2 VLPs by our D.O. claw. This will be done prior to integrating the aptamer into our claw design, to ensure that binding does occur. To detect binding between fluorescent aptamer and NoV VLP, we will use fluorescence polarization. Fluorescence polarization would be able to detect changes in molecular mobility, and as such, molecular weight, of our aptamer in bound and unbound states.
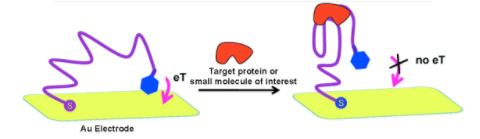
Looking back to our virus sensor design from Figure 1, we intend to use a signal-off mechanism as a binding signal for this sensor. Figure 6 illustrates the basic mechanism we want to incorporate-this shows an aptamer (purple) that has been functionalized with a methylene blue (blue) attached to a gold surface. Once bound to a protein (red), it is rigidified, and the MB is moved away from the gold surface, generating a signal-off response. We intend to implement this signal-off mechanism in our D.O. claw to detect conformational changes.
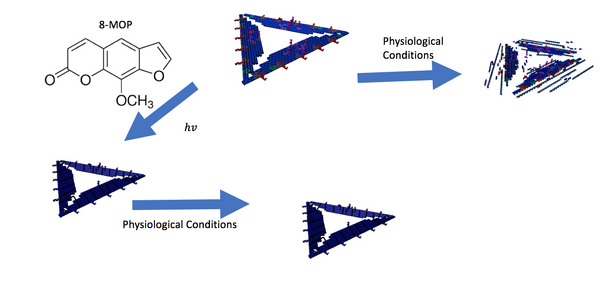
After considering the different conditions of D.O. structures would be subjected to, we intend to investigate how strengthening our structures with a crosslinker would make them more robust against denaturing conditions, illustrated by Figure 7.
Goals
Goals Achieved:
- Stop degradation of VLPs w/ cryoprotectant
- Improved band quality of claws by removing stacking
- Observed significant gel shift in binding using improved claws
- Make multiple versions of claw w/ differently positioned FRET dyes
- Observe FRET signal for unbound claw
- Validated fluorescence polarization with standard dyes
- Demonstrated reproducibility and concentration independence of polarization measurements using control molecules
- Detected the fluorescently labeled aptamer using agarose gel imaging
- Demonstrated a change in polarization between norovirus VLP and aptamer binding
- Demonstrated structural integrity of triangle origami using crosslink techniques
- Demonstrated functionality of sticky sites in crosslinked triangles
Near and Future Goals:
- Demonstrated a change in polarization with VLP and aptamer binding
- Reproduced polarization between norovirus VLP and aptamer binding
- Demonstrated VLP and aptamer binding with agarose gel shift
- Observe FRET signal w/ bound claws