Biomod/2011/TeamJapan/Tokyo/Project/Results
<html>
<style type="text/css">
/* ====================
主に全体に関わるCSS
==================== */
body {behavior: url(http://www.xs4all.nl/~peterned/htc/csshover3-source.htc);}
.clear {clear:both;}
body.mediawiki {
font-size: 14px;
background-color:#707070; background-position: center center; background-attachment: fixed; background-repeat: no-repeat; font-family: Calibri, Verdana, helvetica, sans-serif; }
h1 {
padding: 0px 20px 5px 20px;
font-size: 34px;
font-weight: bold;
}
h2 {
padding: 20px 20px 5px 20px;
font-size: 25px;
color: #0083eb;
text-decoration: none;
font-weight: bold;}
border: none;
h2 a {
color: #eb8300;
}
h3 {
padding: 20px 20px 5px 20px;
font-size: 20px;
color: #000;
font-decoration: none;
font-weight: bold;
}
h1.firstHeading {
display: none;
}
p { text-align: justify; } a:link { color: #00a5ea; text-decoration: none } a:visited { color:#00a5ea; text-decoration: none } a:hover {
color: #eb8300;
text-decoration: none } a:active { color:#f29400; text-decoration: none } #bodyContent {
width: 970px;
margin: 0px 0px 0px;
background-color:#ffffff;
border-width: 0px 1px 0px 1px;
border-color: #000000;
}
#content {
padding-left: 0px; width: 970px;
}
table#team_members {
text-align: justify;
border: 0;
}
table#team_members h2, table#team_members h3 {
clear: both;
}
#content * a:hover {
text-decoration:none;
}
#main_wrapper {
position:absolute;
left:0px;
top:20px;
margin-top: 0; width: 969px; height: 221px; align: center; border-style: solid;
border-color: white;
} /* ====================
メニューの画像を変更できる部分 ==================== */
#header {
position:relative;
left:0px;
top:0px;
margin-top: 0; width: 969px; height: 221px; align: left; background-color: #FFFFFF;
background-image: url(http://openwetware.org/images/e/e0/NEW_header.jpg);
} /* ====================
以下、特殊なclassに適用される ==================== */
#navigation { position:absolute;
left:18px; top:155px; width:1200px; height:69px;
z-index:100;
background-color: transparent;
float: left;
color: #0000FF; } #super_main_wrapper {
position:absolute;
left:0px;
top:227px;
width: 975px; align: center; background-color: #ede8e2;
heigth: auto;
}
#SubWrapper {
width: 645px;
padding: 0px;
border-left:4px solid #ede8e2;
float: left;
margin-top: 0px;
background-color: #ede8e2;
}
#SubWrapper * p, #SubWrapper p {
padding: 0 20px;
text-align: justify;
font-size: 12px;
}
#SubWrapper * h3, #SubWrapper h3 {
padding-top: 10px;
font-size: 18px;
}
#news {
width: 322px;
margin-top: 0px;
float: left;
background-color: #d8d5d0;
border-right:4px solid #ede8e2;
}
#news p {
padding: 0 20px 20px 20px;
text-align: justify;
font-size: 12px;
}
#news h3 {
padding: 10px 20px;
font-size: 18px;
}
#mission_box {
width:650px;
float: left;
}
#team_box, #heartbeat_box, #notebook_box, #parts_box, #gallery_box, #sponsors_box, .boxy {
width:215px;
float: left;
padding: 10px 0 0 0;
}
div.tleft {
border-width: 0px;
margin:0;
padding:0;
border-color:transparent;
}
/* ====================
ここからプルダウン周辺のデザイン ==================== */
- menu * {
margin: 0; padding: 0; }
- menu {
behavior: url(http://www.xs4all.nl/~peterned/htc/csshover3-source.htc); font-family: calibri, verdana, sans-serif;
font-color: #ffffff;
font-size: 19px; background-color: transparent; float:left; padding: 12px 0 0 0; }
- menu ul {
float: left; list-style: none; }
- menu ul li {
background-color:transparent;
position:relative;
float:left; list-style: none; padding: 10px 20px 0 0;
font-weight: bold;
width: auto;
}
- menu a {
color: #FFFFFF; display: inline; text-decoration: none; }
- menu a:visited {
color:#FFFFFF; text-decoration: none }
- menu a:hover {
color: #00a5ea; }
- menu ul li ul {
display: none; position: absolute; left: 0px;
width: 155px;
heigth: 1%;
font-size: 19px; opacity: 0.8; list-style: none;
top: 30px;
padding-top: 20px;
z-index:500;
}
- menu ul li:hover ul {
display: inline;
background-position: bottom;
}
- menu ul li ul li {
width: 100%; list-style: none;
background-color: #000;
margin: -1px;
padding: 0px 0 0 5px;
display: inline;
}
</style>
<script type="text/javascript">
/***********************************************
- CSS Vertical List Menu- by JavaScript Kit (www.javascriptkit.com)
- Menu interface credits: http://www.dynamicdrive.com/style/csslibrary/item/glossy-vertical-menu/
- This notice must stay intact for usage
- Visit JavaScript Kit at http://www.javascriptkit.com/ for this script and 100s more
- /
var menuids=new Array("verticalmenu") //Enter id(s) of UL menus, separated by commas var submenuoffset=-2 //Offset of submenus from main menu. Default is -2 pixels.
function createcssmenu(){ for (var i=0; i<menuids.length; i++){
var ultags=document.getElementById(menuids[i]).getElementsByTagName("ul")
for (var t=0; t<ultags.length; t++){
var spanref=document.createElement("span")
spanref.className="arrowdiv" spanref.innerHTML=" " ultags[t].parentNode.getElementsByTagName("a")[0].appendChild(spanref)
ultags[t].parentNode.onmouseover=function(){
this.getElementsByTagName("ul")[0].style.left=this.parentNode.offsetWidth+submenuoffset+"px"
this.getElementsByTagName("ul")[0].style.display="block"
}
ultags[t].parentNode.onmouseout=function(){
this.getElementsByTagName("ul")[0].style.display="none"
}
}
}
}
if (window.addEventListener)
window.addEventListener("load", createcssmenu, false)
else if (window.attachEvent)
window.attachEvent("onload", createcssmenu)
</script>
</html>
Experimental Results
|
<html><body>
</body></html>
|
<html><body>
</body></html>
|
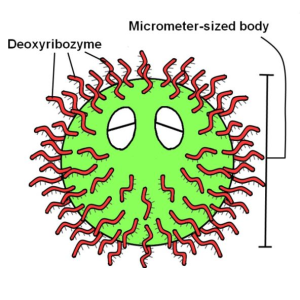
The construction of the body of the DNA ciliate
Method
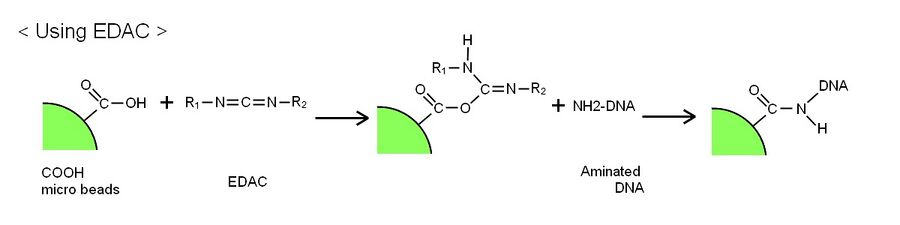
- The deoxyribozymes were attached to the polystyrene body of the DNA ciliate using the following chemical reaction by 1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide hydrochloride (EDAC). The polystyrene bead for the DNA ciliate body has carboxyl groups on its surface. The EDAC reacts with the carboxyl group and forms a reactive group (see the following figure). An amino-modified DNA reacts with the reactive group on the bead surface, and then the DNA is immobilized on the surface of the polystyrene bead body. We carried out this reaction using a chemical reagent kit, PolyLink Protein Coupling Kit for COOH Microspheres (Polyscience) (see Protocols).
- After the above reaction, we investigated whether the deoxyribozymes were actually attached on the surface of the polystyrene body of the DNA ciliate, using a deoxyribozyme activity assays because the deoxyribozyme cannot be recognized through an optical microscope. The deoxyribozyme activity is an RNA cleaving activity in a solution with a divalent ion, Zn2+.
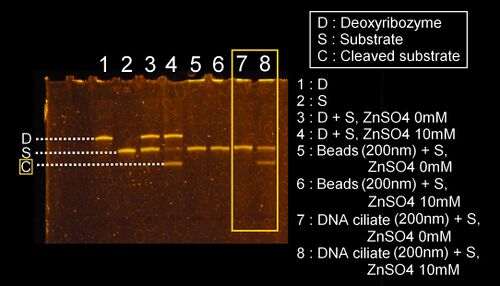
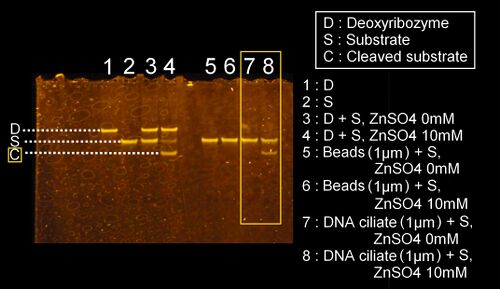
The results of the investigation of the deoxyribozyme activity on the DNA ciliate
Three independent modes of the DNA ciliate
1. Free moving mode
Method
- In the observation of the DNA ciliate, we used 1× saline-sodium citrate (SSC) buffer with 3% bovine serum albumin (BSA) (the materials for experiments are listed in Protocols). The sizes of DNA ciliate bodies were 200 nm and 1 μm. We put the solution including the DNA ciliates on a glass slide and covered by a cover slip. The solutions including the DNA ciliates were observed by a phase-contrast microscope and took videos (the equipment for experiments is also listed in Protocols).
Results
- Video (A) (the left two videos) shows the observation results of the DNA ciliates with a diameter of 200 nm under the optical microscope. Video (B) (the right two videos) shows the observation results of the DNA ciliates with a diameter of 1 μm. The lower videos are enlarged views of the upper videos.
<HTML><body>
| Video (A) <iframe width="450" height="259" src="http://www.youtube.com/embed/uGRn9Z8inW4?rel=0" frameborder="0" allowfullscreen></iframe> | Video (B) <iframe width="450" height="259" src="http://www.youtube.com/embed/-zzB6UeWKoM?hl=ja&fs=1" frameborder="0" allowfullscreen></iframe> |
| <iframe width="450" height="259" src="http://www.youtube.com/embed/E1vW6eaABcQ" frameborder="0" allowfullscreen></iframe> | <iframe width="450" height="259" src="http://www.youtube.com/embed/jKpgMfls3Kw" frameborder="0" allowfullscreen></iframe> |
</body></HTML>
- From these videos, we observed that the DNA ciliates were freely and randomly moving in the solution. By comparing the motion of the DNA ciliates with a diameter of 200 nm and 1 μm, the smaller DNA ciliates (200 nm in diameter) moved more strongly than the larger DNA ciliates (1 μm in diameter). We observed a very slow directional flow of the solution as an experimental artifact but the motion of the DNA ciliate was random independently of the flow. In addition, we observed some DNA ciliates that did not move at all; the DNA ciliates were probably crystalized one another or sticking at the surface of the glass slide.
- In conclusion, we achieved the free moving mode of the DNA ciliate. The motion of the DNA ciliate was based on the Brownian motion of the DNA ciliates. The random motion of the smaller DNA ciliates was stronger than that of the larger DNA ciliate. This result consistent with the theory of the Brownian motion described in Project page.
2. The track walking mode
 |
|
Confirmation of Deoxyribozyme activity
- We confirmed the cleaving activity of the deoxyribozyme for cilia attached on the DNA ciliate body using the polyacrylamide gel electrophoresis. Actually, the results have already been shown in the section of the confirmation of constructing the DNA ciliate body above (see the results) From the results, we conclude that the deoxyribozyme activity for the substrate DNA worked as we designed.
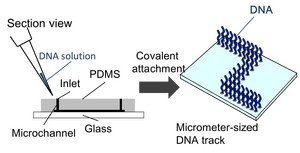
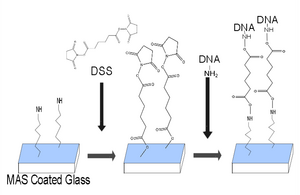
Construction of DNA tracks
Method of the construction of DNA tracks
Results of the construction of DNA tracks
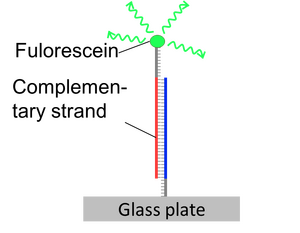
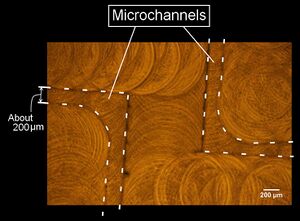
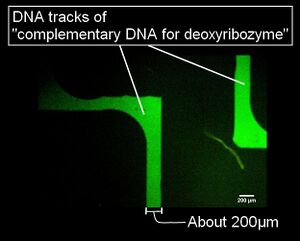
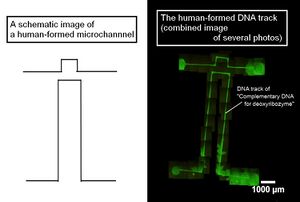
- Figure 1 shows a picture of the polyacetal resin mold (the mold for microchannels are shown by broken lines). This is a part of large microchannels we used to array DNAs. Figure 2 shows a fluorescence microscope image of the DNA tracks. We observed the fluorescence of the DNA hybridizing with the DNA track arrayed on the glass plate. In addition, Figure 3 shows the whole picture of the large DNA tracks with a human like shape (observed similarly).
- In conclusion, we successfully constructed DNA tracks using microchannels, and we confirmed the ability of hybridization between the immobilized DNA as the tracks and its complementary DNA. Thus, we believe that the deoxyribozyme on the DNA ciliate also hybridizes with the track DNA, which has a complementary DNA sequence of the deoxyribozyme.
- In conclusion, we observed the DNA ciliates stayed at the spot of the substrate DNA on a glass plate.
Investigation of the directional walking by simulations
- We investigated the directional walking of the DNA ciliate by numerical simulations (see Simulations)
3. Light-irradiated gathering mode
UV-switching system
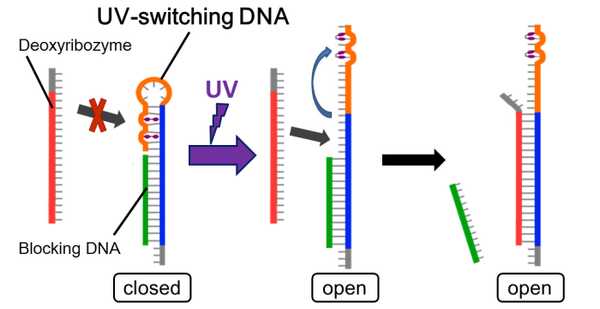
- The UV-switching DNA has a stem-loop structure and short blocking DNA, which blocks hybridization of deoxyribozyme. After UV irradiation, this loop becomes open, and hybridize with the deoxyribozyme. (more detail...)
Confirmation of UV-switching
Method
- We did native-PAGE to check UV-switching system. (more detail...)
Results
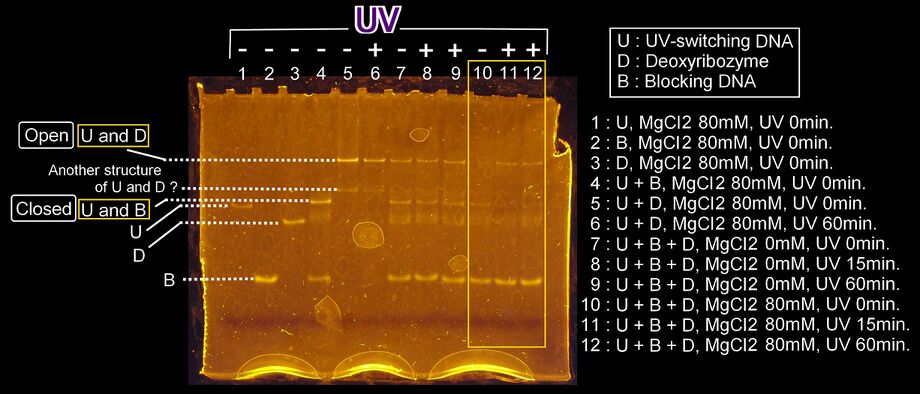
- The control bands were appeared in lane 1 to 6. Lane 4 means the bands when the loop is stable and hybridization U and B . Lane 5 means the bands when the loop is open and not spotted UVhybridization of U and D. Lane 6 means the bands when the loop is open and spotted UV (band U-D).
- In the presence of Mg2+, the switching was caused clearly (lane 10 to 12) because of the stable effect of Mg2+.
- From the picture, in lane 10, there is the band B+U and not the band U+D, so it is confirmed that when UV is not spotted, UV-switching DNA is close the loop. On the other hand, in lane 11 and 12, there is the band U+D and not the band B+U, so it is confirmed that when UV is spotted, UV-swithching DNA is open the loop.
- In lane 4,5, and 6, there is a band which is neither monomer nor hybridized. In lane 1, 2, and 3, there is only a band, so the band is not dimer, so we thought the band is another hybridizing structure. The density of these bands is much weaker than the band of normal hybridizing structure, so we thought this band is little effect to UV-switching system.
- Comparing lane 5 and 6, the density of the bands in lane 6 is weaker than in lane 5. We thought the density of the band U+D becomes weak by spotting UV. We thought that is a reason the band of Lane 11 and 12 are weak.
- Comparing lane 11 and 12, the density of the band U+D is much the same, so we thought UV-switching finished in 15 minutes in this experimentation.
- In conclusion, we confirmed UV-switching DNA changes the structure and branch migration happens when UV is spotted.
Gathering at the specific area
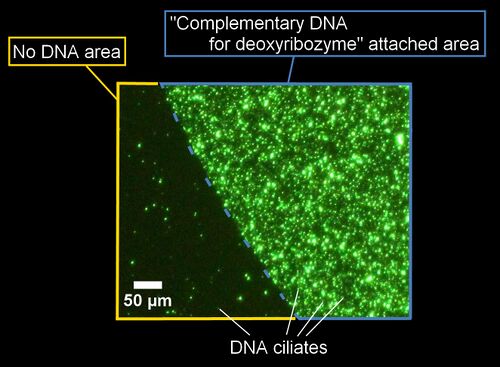
Method
- Attaching complementary DNA of deoxyriboazyme on a glass plate
- Making the situation which deoxyribozymes hybridize with complementary DNA on the glass plate
- How to make the situation for hybridization is here
- Putting DNA ciliates on the glass plate
- Waiting for 2 hours
- Observing the DNA ciliates under an fluorescent microscope
Results
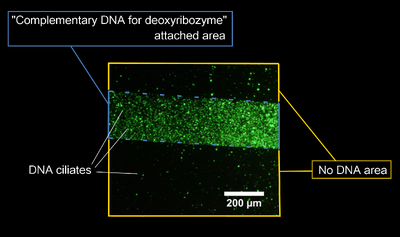
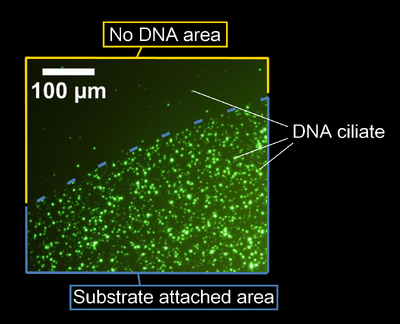
- A fluorescent image of the DNA ciliates gathering at the spot of complementary DNA is here.
- Complementary DNA was attached on upper-right area in this image.
- There was no DNA in lower-left area in this image.
- DNA ciliates gathered at the spot of complementary DNA, and didn't gather at another area. Following this result, it was confirmed that DNA ciliates can gather at the specific area after UV irradiation.