BME100 s2017:Group5 W1030AM L2
| Home People Lab Write-Up 1 | Lab Write-Up 2 | Lab Write-Up 3 Lab Write-Up 4 | Lab Write-Up 5 | Lab Write-Up 6 Course Logistics For Instructors Photos Wiki Editing Help | |||||||
OUR TEAM
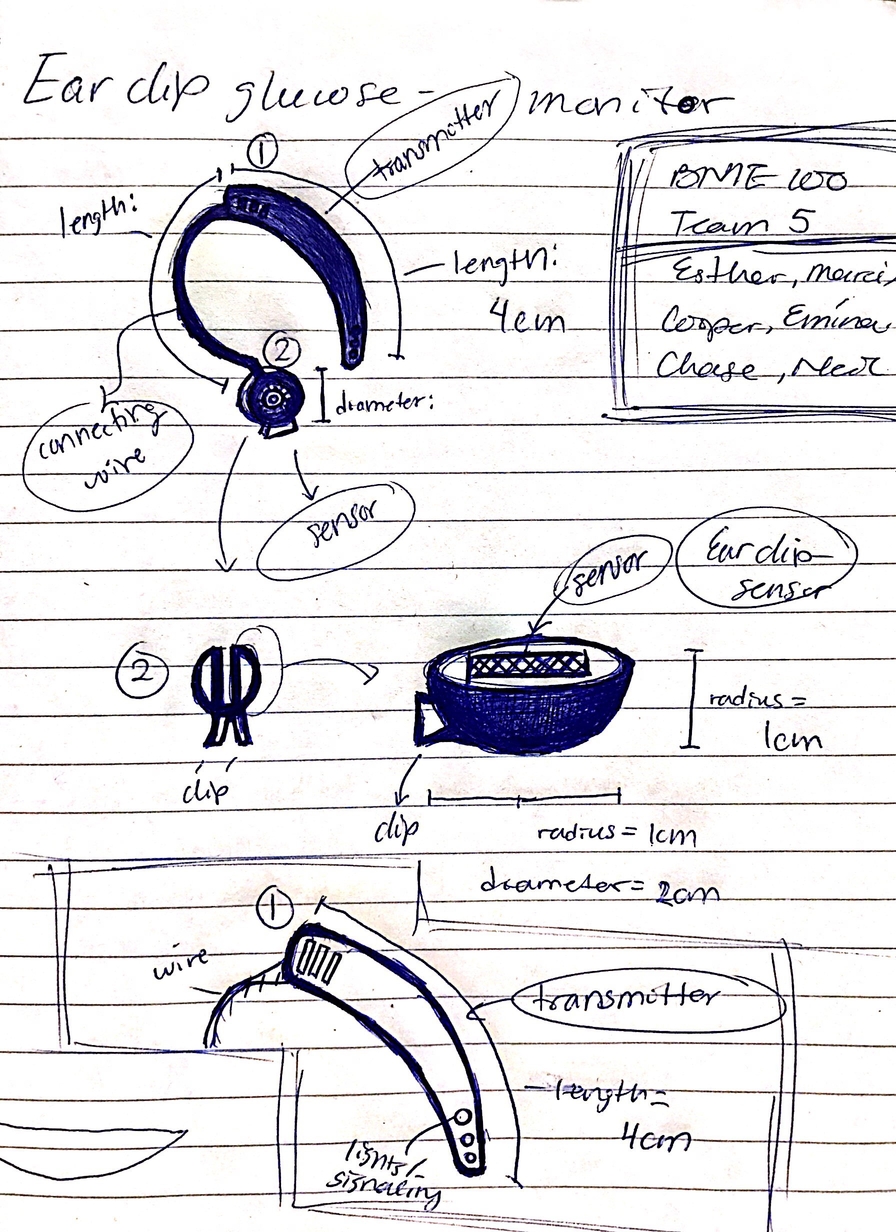
LAB 2 WRITE-UPDevice Image and DescriptionThe sensors on the inside of the clip monitor glucose levels continuously and discretely by attaching to the earlobe. The small wire carries the information to the transmitter hidden behind the ear like a small hearing aid. This transmitter then sends the data to the smartphone app to be interpreted. Furthermore, if glucose levels are critically low the app can send an emergency signal to get help for the patient Our device will address emergency issues related to low glucose levels. Because our device constantly monitors glucose levels and lets the user know when they need to increase their glucose levels, any healthcare issues related to low glucose levels, such as fainting and fatigue, will be reduced. Technical and Clinical FeasibilityTechnical Feasibility For our device to function properly, multiple technologies would need to be present. One such technology would be a very small, very sensitive sensor that would be able to detect changes in glucose levels within the ear. Another needed technology would be a small micro-transmitter that would be able to send the data to a smartphone app or other similar device. The material that the main body of the earpiece would also need to be flexible yet firm so that it can be easily adjusted without being uncomfortable to the wearer. What are the challenges? The challenge in this is that most developmental sensors and transmitters are rather bulky and would be uncomfortably noticeable to people around the wearer. One of the main goals of the device is to provide necessary functions to the wearer without appearing to be a hearing aid, as a stigma against hearing aids is currently present in our society. This makes compacting the device as much as possible a higher priority. Additionally, the development of a material that is firm enough to not fall off of the ear, without being uncomfortably rigid and tight, will be a complicated and tight. However, the payoff would be a much more comfortable alternative to finger pricking methods and other similar ear-based monitoring systems that are currently in development or in the market. What could go wrong? If the device is not accurate in measuring the body temperature, or the ratio of ear canal temperature to glucose levels for a specific person are off, the person could become hospitalized as they would not know the proper amount of glucose and insulin to intake. These errors can be amplified by the downsizing of the sensors and transmitters if they are not made properly, which is an issue that would need to be addressed before the product could move into the market. The device needs nearly perfect accuracy before it can be produced and shipped to the masses. Additionally, any sort of uncomfort caused by the device, or any judgements from people around the wearer will cause them to turn back to whatever method that they used in the past.
The great part of our device is that it can provide continuous glucose monitoring at home and relay it to patients in a user friendly way through a smartphone. This also helps with the clinic since the data is stored on a smartphone for reference later. Or in more severe circumstances can be continuously uploaded sent to a healthcare provider via wifi or cellular data plan. What are the clinical risks? Because our device is noninvasive, there will not be many clinical risks. One of the potential risks that might occur is with the sensor, as the sensor on the clip could possibly damage the ear if the sensor is not designed to fit comfortably on the lobe. Have similar products been in a clinical trial? How long was the trial? There are two similar products to ours on the market today. The first is the Continuous Glucose Monitor(GCM) which works with a small electrode inserted under the skin. The two most popular are by Medtronic and Dexcom (http://online.liebertpub.com/doi/abs/10.1089/dia.2014.0238). There is a clinical trial comparing these two devices which lasted only 6 days. Another study determining how accurate GCM monitors are lasted 7 days (http://journals.sagepub.com/doi/abs/10.1177/1932296814559746). The second product similar to ours is the Glucotrack ear clip monitor which attaches to the ear and connects to a screen via a long cord. The measurement is not continuous and takes about one minute. According to the Glucotrack website clinical trials for this lasted just a few days since it is non invasive and only necessary to confirm the accuracy of the sensor (http://www.integrity-app.com/site/wp-content/uploads/2012/10/integrity_20071.pdf).
Market AnalysisValue Creation
Sales Price
Fundability DiscussionOur prototype should be funded because there are no zero scores on the fundability worksheet. There are scores of 3 in reimbursement, clinical feasibility, and IP position. Scores of two exist for competition, technical feasibility, and regulatory pathways. This leads to a score of 216. Customer Validation- Our device’s score would be a 1 as of right now because we have no specific data to support customer feedback. We are aware of the diabetic population wanting a better device, but we have no determined customers that have agreed to purchase. Market Size- Our device would receive a score of 1 in this criteria because our market size is 126 million in the US. Competition- This would receive a score of 2, there are multiple competitors but improvements are still needed on these devices. While there are many different existing kinds of glucose monitoring devices, like the blood test, CGM devices, and an actual ear lobe monitoring device, the diabetic population is still in need of a device that is non-invasive and easier to use. This still depends on whether the diabetics require continuous monitoring or not, but our device is a significant improvement because of its non-invasiveness, small and simple size, and discreteness. IP Position- This would be a score of 3 because there are numerous amounts of patents existing that are specifically for non-invasive ear lobe monitoring. Technical Feasibility- Our device’s technical feasibility would receive a score of 2, some challenges will be met but overcome with time and within a reasonable cost. Since a smaller and wireless device that monitors glucose on the earlobe hasn’t been made yet, our technology definitely requires research and trials, but it is feasible. Existing devices use wireless monitors that send information to an app, which is exactly what we aim to make but with a glucose monitor integrated with it. Additionally, there is an existing ear lobe glucose monitor, so that is possible; however it is big and not wireless, which is part of our challenge. Regulatory Pathway- Our device would be a score of 2, needing clinical data. This is because while there is approved clinical data for a glucose monitor sensor for the earlobe, our sensor would be the same but just smaller in size which could perhaps affect accuracy of the monitoring. So, clinical data would be needed for the device to make sure the accuracy is maintained. Clinical Feasibility- 3- Our device is similar in form to another non-invasive device that went through short and simple clinical trials only requiring patients coming in twice. The other device that continuously monitors glucose, that is similar in function to ours and is invasive, had relatively short clinical trials lasting up to approximately seven days. Therefore we could expect relatively simple clinical trials for our device. Furthermore, since it is non-invasive it just needs to be checked for accuracy. Reimbursement- This would receive a score of 3 because there are a large number of diabetics frustrated with the existing devices because there are many fallbacks with them like their invasiveness, size, or the amount of times they need to be changed out. They are willing to pay for a new device that would fix all of these problems and make monitoring their blood sugar easier.
|
|||||||