BME100 s2017:Group4 W8AM L3
| Home People Lab Write-Up 1 | Lab Write-Up 2 | Lab Write-Up 3 Lab Write-Up 4 | Lab Write-Up 5 | Lab Write-Up 6 Course Logistics For Instructors Photos Wiki Editing Help | ||||||||||||||||||||||||||||||||||||
OUR TEAM
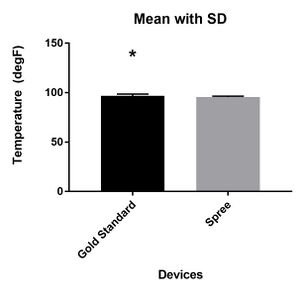
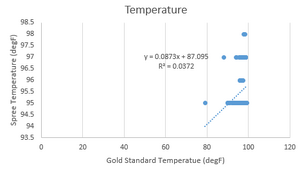
LAB 3 WRITE-UPDescriptive Stats and GraphTemperature Data
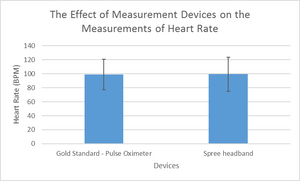
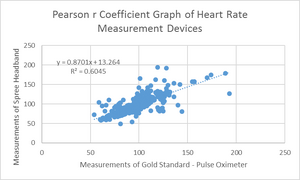
Inferential StatsHeart Rate
P-value: 0.6517 It is significantly different. The test is two-tailed. t= 0.4518 df=296 Number of pairs: 297 Standard Deviation for Gold Standard: 21.6796 Standard Deviation for Spree Band: 24.2612 Correlation coefficient, R: 0.7775
P-value: <0.0001 It is significantly different. The test is two-tailed. t=10.3 df=323 Number of pairs: 324 Standard Deviation for Gold Standard: 1.923 Standard Deviation for Spree Band: 0.8704 Correlation coefficient, R: 0.1929
Design Flaws and RecommendationsOne flaw in this clinical design was that it was not a blind trial. The test subjects knew what products they were testing, so the results could have been skewed. An improvement would be to make this trial blind and have the test subjects not know which device is being tested. This will ensure more accurate results.
Summary of ResultsThe results for the temperature of the Gold Standard and Spree Band are significantly different with a p-value that is less than 0.0001, which means that the null hypothesis that the two products will have the same measurements is rejected.
Experimental Design of Own DeviceBME 100 Experimental Trial Groups- One group with the standard VNS implanted, the second with our device implanted; subjects in both groups are epileptic, groups are at same risk for having a seizure. Sample Size- Group 1: 150 subjects implanted with VNS Group 2: 150 subjects implanted with our device Experiment- Hypothesis: We predict that group 2 (group implanted with our device) will have a decreased frequency and severity of seizures over the course of one year, compared to group 1 (group implanted with the original VNS). Experiment: In an experiment to test our device, a naturalistic long-term study would be necessary. 300 patients will be split into two groups, one group (group 1) of 150 patients is administered the standard VNS, another (group 2) of another 150 patients is administered our device. Both groups will be followed and observed for a year. Groups 1 and 2 will be compared to observe if our device lessens the frequency/severity of a seizure happening. Data will be compared after the year of observing to draw final conclusions Independent variable: Type of instrument implanted into a patient, or patients with no implantation. Dependent Variable: Severity/Frequency of seizures taken place by patients. Inferential statistics/ Rationale for Decisions- In the experiment to test our device, we have chosen 300 patients with epilepsy to fill the sample size. We have divided these 300 patients into two groups of 150 people each. This experiment is split into two to compare the two devices side by side, making two independent groups. This will make our experiment an independent t-test. Our experiment will last over the course of one year and will follow patients over this year, making it a longitudinal study experiment. The reasoning behind this is to compare to a seizure of patients as they happen over the year. Patients in both groups will know whether they have the original VNS or our device implanted, and the data is collected through seizures that randomly by patients, so survey bias will be minimal to none when data is collected. | ||||||||||||||||||||||||||||||||||||