BME100 s2014:T Group6 L3
| Home People Lab Write-Up 1 | Lab Write-Up 2 | Lab Write-Up 3 Lab Write-Up 4 | Lab Write-Up 5 | Lab Write-Up 6 Course Logistics For Instructors Photos Wiki Editing Help | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
OUR TEAM
LAB 3A WRITE-UPDescriptive StatisticsTable 1
All three devices failed the t-test when compared to industry standards for measuring vital signs. P-values were much lower than 0.05 which demonstrates that there was a significant difference between the data sets. Pearson R values were expected to be relatively close to 1.00. Aside from the devices measuring pulse, the values were significantly lower than 1.00. This shows that there is very little correlation between the temperature and blood pressure measuring devices.
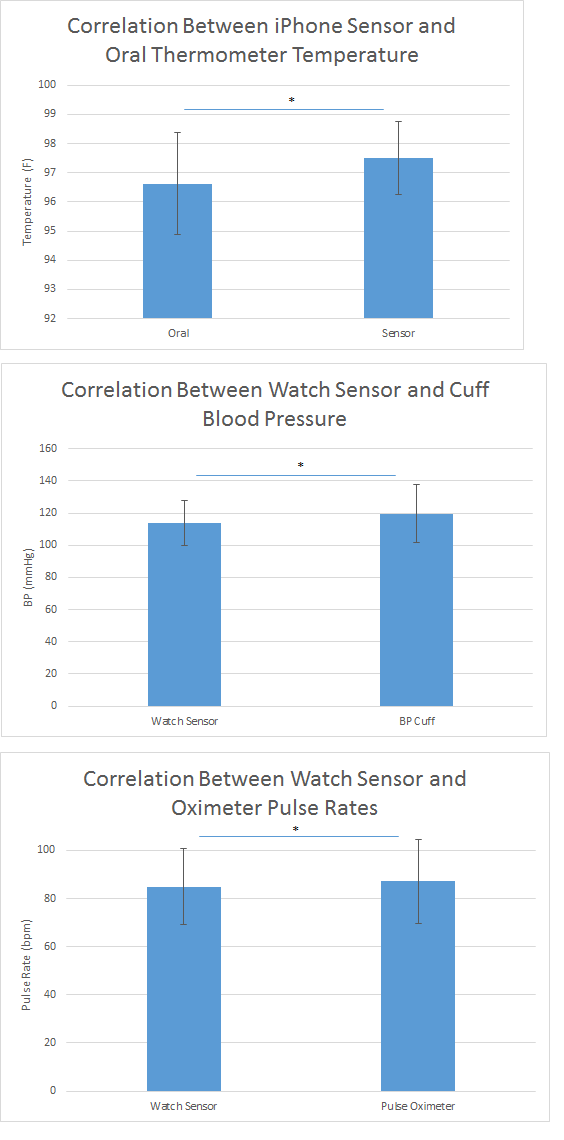
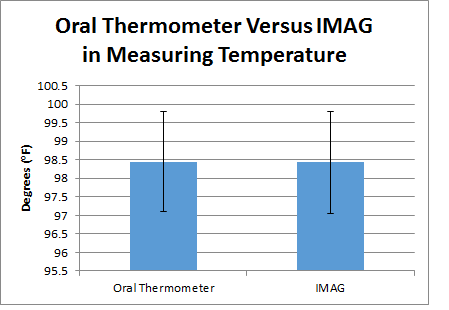
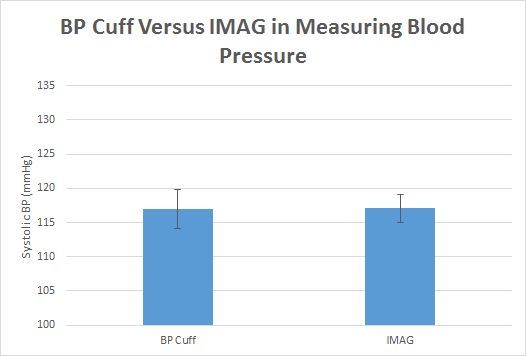
ResultsAsterix's have been placed between error bars to represent a significant difference between the data sets. Each bar in the graph represents the device in the caption below it. See the Table 1 above for appropriate data.
AnalysisDevices designed to measure the blood pressure, pulse, and temperature were compared with similar devices on the market that are known to be an acceptable industry standard. The pulse reading on a wrist mounted blood pressure cuff with a watch style sensor was compared to a pulse oximeter. A Pearson R plot was graphed and found that the two devices correlated to 0.79, and a subsequent t-test confirmed that the two devices differed significantly with a p-value of 0.0014. The same tests were used for blood pressure and temperature. Blood pressure was measured with a standard cuff manometer placed over the brachial artery, and with the wrist cuff from the pulse trial. The p-value was 1.20E-6, showing an even greater significance of variance between devices. The 0.28 Pearson value obtained confirms this variation. By far the greatest variance between devices was found between the iPhone temperature sensor, and the standard oral thermometer. The p-value was 8.09E-13 and the Pearson was 0.048. The same population group was used for each device tested, and as such the t-test performed was paired.
Summary/DiscussionThere are several changes that could be made to the RAIN sensor and the Omron wrist cuff, as well as the actual experimental design. First of all, the RAIN sensor could have been equipped with a better system of attaching to the subject's skin. Throughout the entire experiment, the subject had to hold the sensor to her skin, and therefore it is hard to say that the sensor was held onto the skin with constant pressure. This could have led to errors in the data. The part of the sensor that is attached to the skin could be smaller and easier to attach, and the actual body of the sensor could separate until smaller, lighter devices can be substituted. For example, the processing unit of the sensor could be detached and worn in a pocket, and wire leads could be used to capture temperature data. This would ensure that the sensors would stay in place without the body of the sensor pulling them off the skin. IR sensors placed in the module may also provide better readings than the metal studs that rely on thermal conductance. In regards to the wrist cuff, a possible design flaw could be that sometimes it tightened excessively around the subject's wrist, and other times it seemed to have barely tightened at all. This also could have led to discrepancies in the data since it had to have the same amount of constriction around the subject's wrist each trial in order to get reliable readings and maximize accuracy. A means to fix this error could be to incorporate a more sensitive pressure sensor in the wristband so the device is more apt to stop constricting when it reaches the optimal level of tightness. An experimental design flaw could be that in each group only one subject was tested. It could have been better to test more than one group member with the same device to provide a comparison for the device within the group. Even though different people have different vitals, if a device's readings seemed off from the rest of the class's readings, it would be helpful to know if it was the device or the person's abnormal vitals that caused the difference. In a perfect experiment, the same device would be used for every subject to reduce the sources of error.
LAB 3B WRITE-UPTarget Population and NeedOur company, Isentrotech will be providing internal monitoring equipment capable of sampling blood pressure, pulse rate, temperature, as well as many other chemical signatures derived from blood. An artificial arterial graft can be implanted inline with the brachial artery. An embedded processor sends data to a watch sensor, smart phone, or computer based software for real time continuous monitoring. The hope is that patients that require close monitoring of signals they may not be aware of will now have access to these signals and therefore greater freedom and mobility. Blood glucose, endocrine disorders, aids, and any other conditions threatened by fluctuations in blood chemistry could experience significant benefit from this technology. We hope to provide research platforms for real-time nutrition and metabolic studies, and enable professional scientists to catalog this information for future generations. The device has limited hardware capacity due to size constraints, but can easily be tailored to the individual needs of the patient.
Device DesignA vascular graft made of Dacron lined in collagen is mounted with a long sensor/transmitter module capable of wireless communication with a wrist watch, computer software, or smart phone. The sensor module is completely encapsulated in a layer of Dacron to avoid contact with body fluids. Sampling ports are allowed to extend into the blood stream. A PTFE compound covered in nano-scale protrusions ensures that protein formation does not occlude the sensors.
Inferential StatisticsTable 2
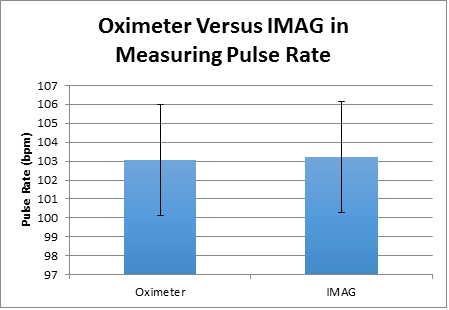
The results of the T-Test for all of the Pulse,Temperature,and the Blood Pressure were higher than 0.05, which indicates that our devices can be compared to industry standard devices. The Pearson R final results for the Pulse,Temperature,and the Blood Pressure were high, and almost reached one, which means that the results of our devices are almost the same as the results of the industry standard devices.
Graph
The final result that we can take out from the graphs is that the differences between our devices and the industry standard devices are too small and barely can be realized, so by comparing our devices and the original devices our devices are almost functioning similar to industry standard devices. See Tables 2-4 above for appropriate data. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||