BME100 f2017:Group2 W0800 L5
| Home People Lab Write-Up 1 | Lab Write-Up 2 | Lab Write-Up 3 Lab Write-Up 4 | Lab Write-Up 5 | Lab Write-Up 6 Course Logistics For Instructors Photos Wiki Editing Help | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
OUR TEAM
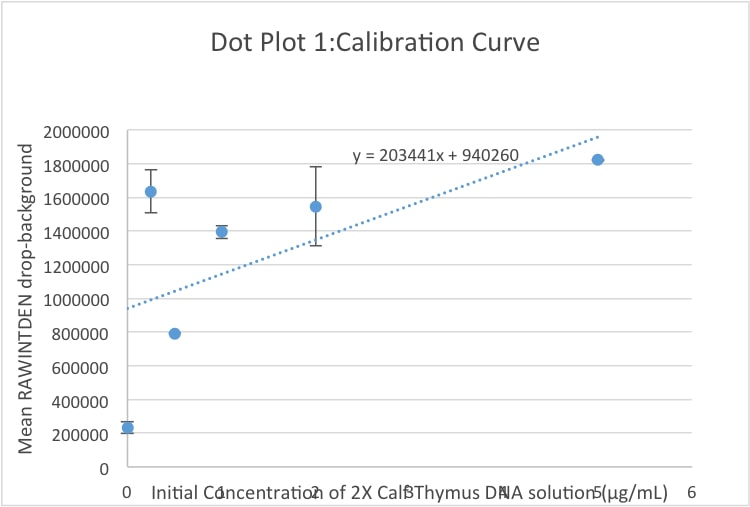
LAB 5 WRITE-UPPCR Reaction ReportThe purpose of this PCR reaction lab experiment was to measure different concentrations of DNA and use our smart phone to complete a calibration. We used SYBR Green dye, Flourimeter, calibration samples, a positive control, negative control, 3 DNA samples of patient 1, and 3 DNA samples of patient 2. We used our 2X calf Thymus DNA and SYBR Green solution as our calibrator data to measure how much DNA was produced by PCR. We did this by using the calibrator data to create a calibration curve, then we collected the data from the positive control, negative control, 3 DNA samples from patient 1, and 3 DNA samples from patient 2. Combining this Data on an excel spreadsheet, we found that each patient had different values for the initial PCR Product Concentration (µg /mL). This led to the conclusion that Patient 1 was closer to the negative control, and Patient 2 was inconclusive. Fluorimeter ProcedureImaging set-up Obtain all of the needed materials, and set the fluorimeter on top of two closed boxes that contains extra replacement tips for the micropipette(two boxes were used due to the size of the phone used). Turn on the blue laser light on the fluorimeter, and fit the glass slide, with the rough side facing up, onto the designated area. The designated area has a width that is a few millimeters wider than the slide itself, and can perfectly fit the slide. Find a stand that can set up the phone at an appropriate angle that slightly looks down on the slide being used; the phone should be set up about 4 centimeters from the base of the boxes to create an optimal camera focusing distance. The pictures taken should have the drop being the main focus of the picture, and this distance to the drop should stay constant. Following this setup the phone should be taking pictures that are perfect for the additional steps that are taken with the pictures using image J. The phone used in the experimentation was an IPhone 6s. The phone needs to have special conditions in order to make the photos come out clear. These special conditions include turning off the live feature and shutting flash mode on the camera. The camera also needs to be in a stationary state to maximize the clearness and preciseness of the photos.
Placing Samples onto the Fluorimeter 1. First, the micropipette should be set to 80mL, then the micropipette should be fitted with a tip firmly so the tip will not move. Proceed to press the top all the way down, while holding the top down insert the tip into one of the eight solutions. Release the top once the tip is at the very bottom of the container making sure all of the liquid is sucked up into the micropipette. 2. Release the entire contents inside one of the bigger capsules with the red marking on top, then close the red marked capsule, and turn upside down to mix the two solutions. Change the tip of the micropipette to a new one, and then press the tip all the way down. Insert the tip into the capsule containing SYBR Green I, then release the top to suck up 80mL of contents. 3. With the tip full of liquid place the tip in between the first two middle circles on the glass slide, slowly press down the top to release the liquid into a "beach ball" on the glass slide. Change the tip on the micropipette once again to a clean one, proceed to press the top all of the way down and place the tip into the red marked capsule previously mentioned in step 2. Release the top sucking up 80mL of the liquid, and move the micropipette tip over to the SYBR Green I already on the glass slide. 4. Press down the top slowly to release the liquid in the tip onto the ball already on the slide. Be careful that the liquid does not splash and therefore disperses, and try to keep the droplets as centered on the slide as possible. Center the beam into the middle of the droplet for best results. Put the cover on the fluorimeter to shelter out as much light as possible, and take 3 pictures of the droplets using the setup. DO NOT MOVE THE PHONE FROM THIS POINT ON. Take off the cover, and set the micropipette to 160mL. Press down the top of the micropipette all the way, and stick the tip into the droplet. Release the top to suck up all of the 160mL drop on the glass slide, once again discard the tip. Move down the glass slide to the next two unused middle circles for the next incoming droplets. 5. Repeat steps 1-4 for all solutions. *Exception- For the large capsules with numbers on the lid, treat them like already mixed red big capsules. No contents need to be mixed with them.*
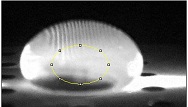
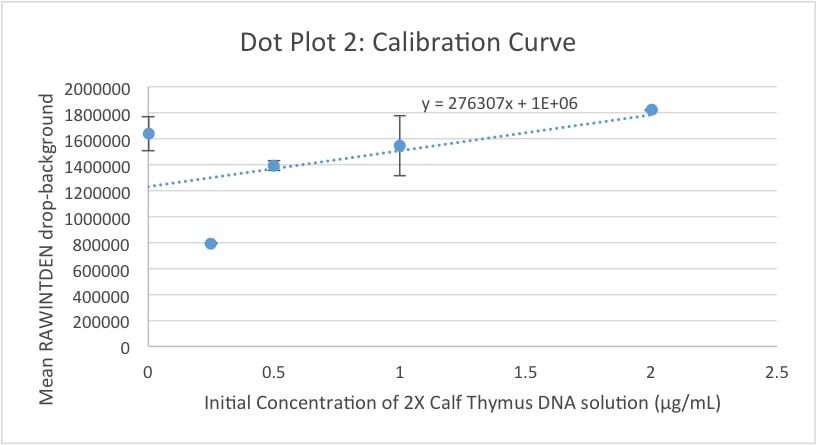
Data Collection and AnalysisImages of High, Low, and Zero Calf Thymus DNA 5 μg/mL sample 0.5 μg/mL sample zero DNA Calibrator Mean Values
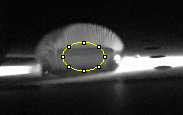
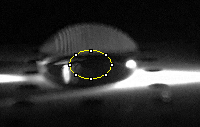
Calibration curves Images of Our PCR Negative and Positive Controls PCR Positive Control PCR Negative Control
PCR Results: Summary
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||