BME100 f2016:Group3 W8AM L2
| Home People Lab Write-Up 1 | Lab Write-Up 2 | Lab Write-Up 3 Lab Write-Up 4 | Lab Write-Up 5 | Lab Write-Up 6 Course Logistics For Instructors Photos Wiki Editing Help | ||||||
OUR TEAM
LAB 2 WRITE-UPDevice Image and DescriptionOur device is an implantable drug delivery device for Alzheimer's patients in the mouth. The implant will resemble that of a typical tooth implant with a porcelain cover to blend into the mouth to look natural and ensure safety for the patients. The removable top opens up to an injection spot where the drug will be refilled by medical professionals. Once injected, the medicine will travel into a reservoir located in screw of the implant. The screw, therefore, is not completely solid and has a hollow reservoir in order to maximize space and efficiency. The delivery process is powered by a small battery in the tooth portion of the device and powers a microprocessor that controls the dose of the medication. This occurs by the microprocessor sending a message to value sensor located towards the bottom of the medication reservoir. The valve then contracts to release the medication into micro needles that go into the main blood vessels.
The following is a prototype of our device. On top of this screw will be the molar tooth. Within the tooth there will be the battery and microprocessor chip. Within the screw there will be the medication reservoir, valve, and sensor controlling the valve to release the medication into a micro needle that attaches to the blood vessel in the gums.
Technical and Clinical FeasibilityTechnical Feasibility What are the challenges? The biggest challenge is creating small enough technologies to fit inside an implantable tooth. Another challenge is making sure the micro needles are connected to the blood vessel in the gums without hitting any nerves. What could go wrong? In the mouth there are a lot of nerves and permeant nerve damage is a scary possibility if the micro needles accidentally hit a nerve instead of the blood vessels. Another possible problem is if the micro needles are not secured in the blood vessels, it can cause it to move around causing either a rupture of the vessel or causing blood to flow out into the gums. Also, infection can occur due to the surgical implantation of the device in the patient. Clinical Feasibility What are the clinical risks? The clinical risks are possible nerve damage, possible bleeding of the gums, and/ or sensitivity to the materials of the product if someone is allergic. Another risk could be that the product does not distribute enough or distributes too much of the drug to the participants. Another risk is infection of participants disrupting the trial. Have similar products been in a clinical trial? Yes, the Intellidrug has been in a clinical trial, however, the results of the trial have not been released to the general public. How long was the trial?
Since the Intellidrug clinical trial results have either not been completed or the results just have not been released to the general public, the length of the trial is undetermined at this time.
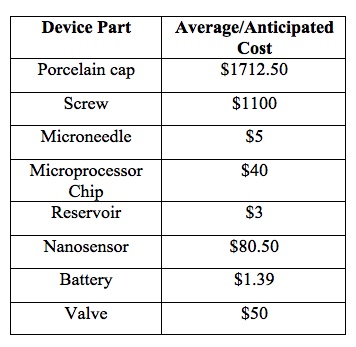
Market AnalysisValue Creation The implant our customer would receive has many advantages that advance their health. Dental implants tend to have a reputation of creating a comfortable and stable environment. This means that there is no need for adjustment after implantation other than the replacement of the drug that is being ejected by the microneedles. Tooth implants are known to be stronger than the natural tooth. Studies have shown that patients tend to not even realize that they have an implant. This is important for patients with Alzheimer's because as the disease progresses, they have a high tendency to get frightened when they cannot remember why something is the way that it is. This device would eliminate the cost of having a caretaker or someone to take off work in the early stages of the disease because the medication will be automatically administered on a timely basis. This greatly takes stress off the patient and the patient's family both financially and emotionally. Manufacturing Cost Our product consists of a porcelain tooth cover to disguise the implant and allow it to look more natural, a micro needle that injects the mediation directly into the blood stream, two sensors that allow the microchip to record how much medication is being released, a reservoir that stores the medication and a small battery that allows the implant to function. Overall, the cost to create our product is predicted to cost around $3000 to create. The justification of that price is outlined in the table below: Sales Price The average sale price of our product would be about $3750 per implant. Since our product is designed to save the patient and the patient's family money by replacing a caretaker in the early stages of the disease, we did not want to raise the price of the device too much. Another reason the sale price is not that much higher than the cost to make the device is that we want insurance companies to cover a procedure like this and by having a lower price, more insurance providers will be willing to do this. Thus, a greater number of individuals will be able to obtain this device which would ultimately increase our profit. Market Size There are currently over 5 million people affected by Alzheimer's disease. The expected amount of individuals with the disease in the year 2050 is over 13 million which means that the amount of people able to purchase the product will continue to increase based on the predictions made. It is stated that families that are affected by Alzheimer's spend over 5,000 dollars alone on annually caring for the patient affected by this disease. In the long run,the cost of purchasing the tooth implant and the administration of the drug could save a family thousands of dollars. If the amount of people obtaining Alzheimer's disease increases as projected, then more than 235,000 people will obtain the disease each year. Thus, our market size is about $881,000,000 per year. This gives us a score of 3 in the fundability worksheet because it falls between the range of 500+ million dollars per year.
Out of the categories that we gave scores for, our prototype would be given a score of 96. Thus, as long as the other scores are both a score of 2 or higher, our prototype should qualify to be funded at a total score of about 384. Since our product received scores of mostly 2, there is a stable amount of evidence that the product could be successful and should be funded. As nanotechnology continues to advance, the scores for technological feasibility and clinical feasibility should rise to scores of 3 which would most likely guarantee that the project would be funded.
Fundability DiscussionTechnological Feasibility: The technological feasibility of our device would fall under a score of 2 because although nanotechnology is not necessarily developed for this specific device yet, with time it can be developed without a doubt. Clinical Feasibility: The clinical feasibility of our device would fall under a score of 2 because our device definitely can be successful in a clinical trial, however, some research and/or expertise may be necessary to alter any mistakes or complications when running the device.
|
||||||