BME100 f2016:Group3 W1030AM L2
| Home People Lab Write-Up 1 | Lab Write-Up 2 | Lab Write-Up 3 Lab Write-Up 4 | Lab Write-Up 5 | Lab Write-Up 6 Course Logistics For Instructors Photos Wiki Editing Help | ||||||
OUR TEAM
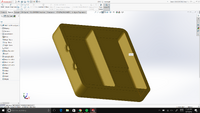
LAB 2 WRITE-UPDevice Image and Description
Technical and Clinical FeasibilityTechnical Feasibility a. What are the technologies needed? •The implanted device needs a means to relay information/signals to the external device; much like BlueTooth.
•Ensuring that the internal temperature of the body does not shorten the shelf-life of the epinephrine
c. What could go wrong? •System could malfunction
Clinical Feasibility a. Will it work in the clinic? Clinical trials •https://clinicaltrials.gov/ct2/show/NCT00828542?term=implanon&rank=2 Clinical trial for Implanon birth control (Implanon is an implantable birth control device that releases hormones) consisted of two groups one group that was given oral hormonal birth control and another that received the implanted device. To monitor the success of the device in comparison to the pill, blood samples were taken periodically overtime and certain metabolic variable associated with birth control were monitored. •https://clinicaltrials.gov/ct2/show/NCT00636493?term=implantable+drug+delivery+device&rank=1 This study assessed the efficacy of a device inserted in the eye that releases steroids to combat retinal vein occlusion. This device was deemed safe and effective. •https://clinicaltrials.gov/ct2/show/NCT01432522?term=epi+pen&rank=2 This study compared the absorption and circulation of epinephrine administered through intramuscular injection and that of epinephrine administered by intranasal spray.
-Since this is a life saving device, people’s lives have to be put in danger in order to test if this device is effective. One way to test the effectiveness of the device, would be to test it thoroughly on animals. Also, one of the clinical trials did not induce an allergic reaction to test the effectiveness of their drug (epinephrine delivered intranasally). They instead compared the absorption of epinephrine into circulation of both their device(the intranasal spray) and an epi pen. - Subject age, many children who would be using the device are ineligible for a clinical trial.
-Intranasal epinephrine delivery spray (another epi pen alternative): The clinical trial went from July 2010- September 2011. -Implanon device (Implanon is an implantable birth control device that releases hormones.): The clinical trial lasted from July 2007 to February 2008. -Fluocinolone Acetonide Implant for Retinal Vein Occlusion (This device is inserted into the eye and releases steroids to combat retinal vein occlusion) : The study lasted from October 2002-May 2009.
Market AnalysisValue Creation Manufacturing Cost -The life-saving dose of epinephrine contained in an Epi-pen is worth approximately $1 http://www.bloomberg.com/news/articles/2015-09-23/how-marketing-turned-the-epipen-into-a-billion-dollar-business -The cost of the Implanon birth control implant varies widely from $5-$25 to $400+ depending on insurance, which suggests that the cost to produce the implant is relatively low. https://www.getthefacts.health.wa.gov.au/condoms-contraception/types-of-contraception/implanonreg https://www.plannedparenthood.org/learn/birth-control/birth-control-implant-implanon Sales Price Market Size
Fundability DiscussionCustomer validation - 2 Market size - 3 Competition - 2 IP position - 2 Technical Feasibility--2 Clinical Feasibility--1 Our fundability score is 48. Our prototype should be funded because there are many people who are looking for an alternative to Epi Pens. Our product offers many benefits that Epi Pen does not offer such as increased convenience (through its unique delivery system) and peace of mind (from the fact that it’s always with the customer).
|
||||||