BME100 f2016:Group2 W8AM L3
| Home People Lab Write-Up 1 | Lab Write-Up 2 | Lab Write-Up 3 Lab Write-Up 4 | Lab Write-Up 5 | Lab Write-Up 6 Course Logistics For Instructors Photos Wiki Editing Help | ||||||
|
OUR TEAM
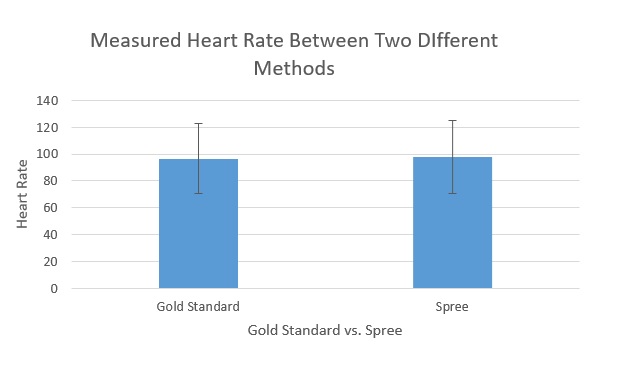
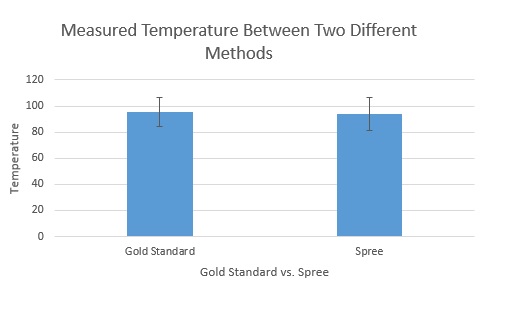
LAB 3 WRITE-UPDescriptive Stats and GraphHeart Rate Note: The "Gold Standard" heart rate was measured with a pulse oximeter. Gold Standard - Average: 96.4974026 Gold Standard - Standard Deviation: 25.99859918 Temperature Note: The "Gold Standard" temperature was measured with an oral thermometer. Gold Standard - Average: 95.48115152 Gold Standard - Standard Deviation: 10.76273631
Inferential StatsHeart Rate P-Value (Paired): 0.298225789 Temperature P-Value (Paired): 9.25843E-05
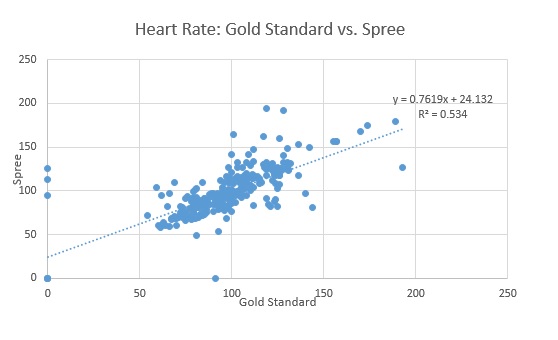
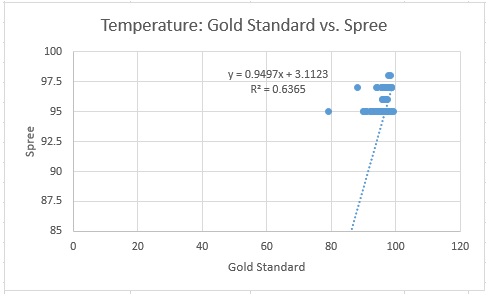
Design Flaws and RecommendationsWhen comparing the pulse ox heart rate measurements to those of the Spree Band, it is important to look at Pearson's R value. In this care, it is 0.730784539, meaning that there is a weak, positive correlation between the two devices. When comparing the P-Value (Paired) from the T-Test, 0.298225789, it is seen that it is greater than 0.05. This means that there is a significant difference between the measurements. When comparing the oral thermometer temperature measurements to those of the Spree Band, it is important to look at Pearson's R value. In this case, it is 0.797813158, meaning that there is a weak, positive correlation between the two devices. When comparing the P-Value (Paired) from the T-Test, 9.25843E-05 , it is seen that it is very close to zero. Based on the experimental results, it is determined that the Spree Band tracker is not effective for the intended purposes. The temperature and the heart rate were recorded from both the Spree (headband) and the Gold Standard Devices(an oral thermometer and a pulse oximeter). After the results of the Spree Band were recorded and compared to the Gold Standard measurements, it was determined that the Spree Band was not effective in its measurements. The device is flawed by providing inaccurate measurements, and is altogether unappealing to consumers.
Experimental Design of Own Device100 people will be tested using the device: 50 people who have been diagnosed with MS and 50 people who have tested negatively for MS. They will use the device as they would with a cholesterol tester. The patients will be blindly tested and the results from the device will be compared to the results of other traditional MS detecting tests. | ||||||