BME100 f2016:Group11 W8AM L6
| Home People Lab Write-Up 1 | Lab Write-Up 2 | Lab Write-Up 3 Lab Write-Up 4 | Lab Write-Up 5 | Lab Write-Up 6 Course Logistics For Instructors Photos Wiki Editing Help | |||||||
|
P.A.N.D.A.
Our Brand Name LAB 6 WRITE-UPBayesian StatisticsOverview of the Original Diagnosis System
There were a couple of error precautions that we did. For each patient there were three runs that were done so that there will be less errors. Then the PCR controls were all done by a machine, so that excluded human error. The ImageJ calibration controls were done on the computer; there was little human error present there - only drawing the area of the drop as accurately as possible. Three images per each drop were taken so that the results would be an accurate representation as to whether the drop actually fluoresced or not. Of our results we had two patients with blank data. Then, there were 6 slots with inconclusive results. We had 13 positive and 15 negative test results. There were 7 results that were completely wrong out of 30 tested.
What Bayes Statistics Imply about This Diagnostic Approach
Intro to Computer-Aided Design3D Modeling
Feature 1: ConsumablesVery important consumables are ones that are required to do one experiment. Without them, the experiment cannot be done.
Instead, we will be using a spectrometer nanodrop device which is built into the device and so eliminates any need for SYBR green dye, for example. The complete and total automatic nature of the device also eliminates the need for micropippettes which are reusable within the internal mechanisms of the machine.
These of course will be required to build the many replications of a diseased section of DNA and will be included and place in its specific cartrage during testing. This will likely end up being the limiting reactant in each experiment and so a suitable amount will be placed according to the needs of our client.
This will include the taq polymerase required to build the extra strands of DNA during the thermocyling with a new batch required for every new test. This will be a critical component of the reaction and will certainly come with every kit of the device in adequate supply.
Disease specific primers will be included according to the individual needs of each client. This will depend on what disease is primarily being tested, though it is expected that a large amount of hospitals will purchase our product. For this, we will have a special edition kit that includes a broad spectrum of primers in order to appropriately meet the unpredictable demand of an emergency room or several hundred long term patients.
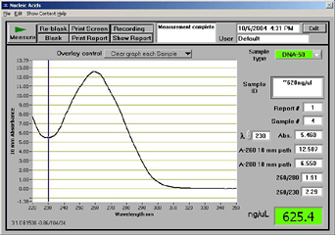
This will also be included as a courtesy for the step that will require a sample of a patient's DNA to be inserted into the PCR Mix. We will also include the option of not ordering this product along with pippette tips as these will be likely already owned by the hospitals and research institutions that will purchase our product. Feature 2: Hardware - PCR Machine & FluorimeterA major weakness that our device addresses is the slow time and tedious nature of receiving results primarily caused by the fluorimeter method. We will be excluding it altogether and instead incorporate a nanodrop spectrometer into the device with results that can be transmitted by Bluetooth onto an app or computer. This will reduce the number of thermocycles required for testing as the nanodrop device can detecty far lower concentrations than can be using the fluorimeter in the original method with the vanilla OpenPCR. We will also augment the device by creating a tray that will work similarly to a printer with trays for PCR mix, primer solutions, etc. with it all being mixed in precise amounts internally with mechanized micropipettes. Once the mix is prepared a vacuum tube will send it to the thermocyler and will then afterwards be delivered to the nanodrop machine for final analysis. The color will be changed to black. Other than this, the design of the OpenPCR will reamain unchanged. In order to accomadate all the extra hardware, it is worth noting that the device will likely be larger and heavier than its predecessor. Nanodrop Machine: Source: U. (2014, April 23). NanoDrop. Retrieved November 21, 2016, from http://gcf.uta.edu/Nanodrop.html Results Example: Source: Nanodrop Spectrophotometer. (2010, March 14). Retrieved November 21, 2016, from http://dnatech.genomecenter.ucdavis.edu/nanodrop-spectrophotometer/
| |||||||