BME100 f2016:Group11 W8AM L5
| Home People Lab Write-Up 1 | Lab Write-Up 2 | Lab Write-Up 3 Lab Write-Up 4 | Lab Write-Up 5 | Lab Write-Up 6 Course Logistics For Instructors Photos Wiki Editing Help | ||||||
OUR TEAM
LAB 5 WRITE-UPPCR Reaction ReportIn the beginning, we had a set of micropipette tips along with a micropipette. We had PCR samples to mix with the lab patients data and a multitude of tubes. The method into labeling the test tube with PCR was based on two different groups. We cut the eight tubes given into two sets of four tubes so that they could fit into the cycler. Putting the group number first(G11), we had a positive and negative control tube, three tubes with DNA samples of the corresponding patient. There was no change needed in labeling, most of the labeling was just a conscious understanding of our team about what the labels meant for each test tube.
After the tubes were labeled, the next step was combining the necessary elements of the PCR, such as the DNA samples from each of the patients, the PCR mix with the enzymes and Taq plymerase, and the positives and negatives. Of the PCR mix, in every labeled tube 50 microliters were added. Then, into the positive and negative tubes, 50 microliters of the positive and negative samples were added, respectively. In the tubes of DNA of the patients, 50 microliters of each patent's sample was inserted. This way, in tubes 1.1, 1.2, and 1.3, the DNA of patient 56439 was placed, and in tubes 2.1, 2.2, and 2.3, the DNA of patient 58174 was inserted. For each addition of liquid the following steps were followed: First, the pipette was set to the desired amount. A tube with liquid that will be transferred was opened. Next, a pipette tip was attached to pipette by firmly pressing on it. Before placing the pipette tip into the liquid, the button was pressed to the first stop. The tip was then inserted and the stopper was slowly released to suck the amount of liquid needed. Next, the tip was inserted into the new tube (with or without liquid, depending on the sequence of the addition of the liquids), and the stopper was pressed to the second stop to get out the exact amount of liquid. Finally, the tip was ejected into the cup. After everything was added to the tube, the tube lid was closed. After filling all the tubes with the necessary fluids, they were taken to the thermal cycler which was programmed to the specific cycling program with specific temperatures and times. The lid was closed, and the cycler started working. After about one hour, the tubes were placed in a freezer for the next lab. The pre-lab reading did help, especially with understanding proper micro-pipetting techniques and the concepts behind thermal cycling as well as knowing which mixes were needed for the final samples in the thermal cycler. This also means we had a solid understanding of the difference between the first and second stop. The first was used to collect a correct volume of liquid while going down to the second stop would have a miscalibrated suction power, collecting an incorrect volume of liquid. Pushing down to the second stop is only appropriate when dispensing collected liquid. The final liquids unfortunately did vary slightly in volume as visibly viewed at the side of the tubes. On one particular PCR reaction mix tube, there was significantly more left than in the other 7 tubes, indicating a break from proper micro-pipetting technique when it was being collected. Fluorimeter ProcedureImaging set-up
Placing Samples onto the Fluorimeter
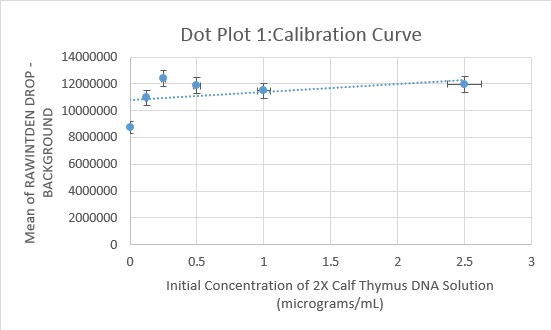
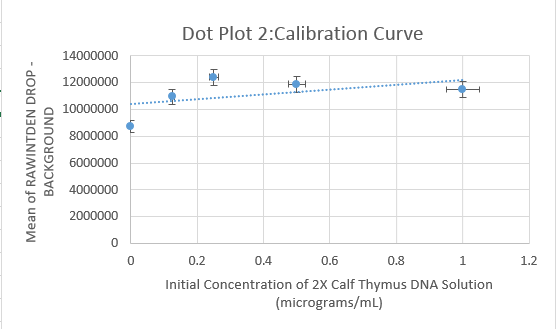
Data Collection and AnalysisImages of High, Low, and Zero Calf Thymus DNA
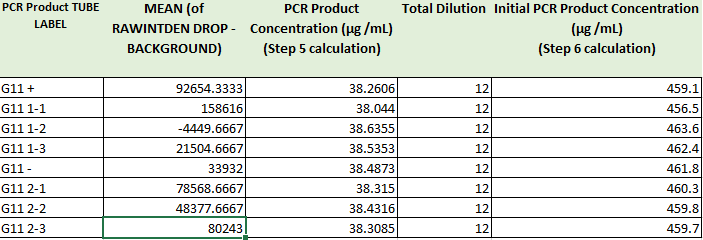
Images of Our PCR Negative and Positive Controls Positive Control Negative Control PCR Results: PCR concentrations solved
| ||||||