BME100 f2014:Group22 L2
| Home People Lab Write-Up 1 | Lab Write-Up 2 | Lab Write-Up 3 Lab Write-Up 4 | Lab Write-Up 5 | Lab Write-Up 6 Course Logistics For Instructors Photos Wiki Editing Help | |||||||||||||||||||||||||||||||||||||||
|
OUR TEAM
LAB WRITE UP 2Descriptive StatisticsHuman Trial: Average Inflammotin Levels
Rat Trial: Average Inflammotin Levels
Results (Graphs)Human TrialRat Trial
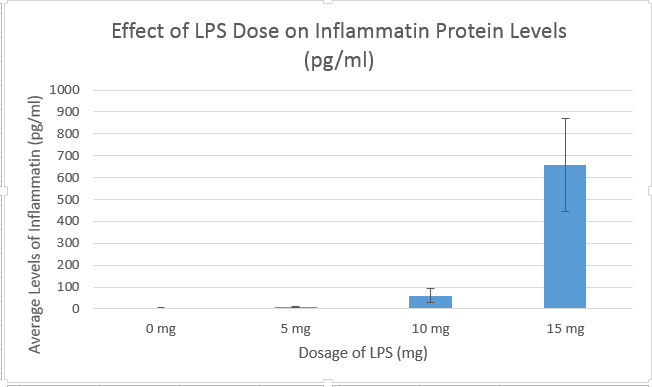
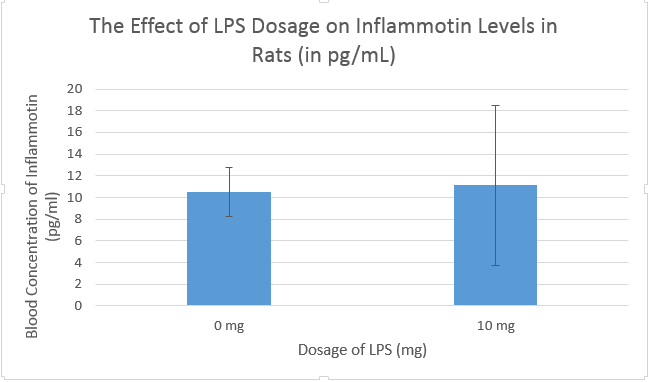
Analysis (Inferential Statistics)Human TrialA one-way ANOVA test was run for the human trial in order to compare between-group variation of four doses of LPS. Post-hoc, a Student's T Test was run on the human trial data in order to compare the average inflammotin levels for each dose to the average of each other trial (i.e., 0 mg vs. 5 mg, 0 mg vs 10 mg, etc.). The ANOVA test returned a p-value of 1.40 x 10-16, far below the p=0.05 maximum uncertainty margin for statistical inter-group significance. Therefore, based on the data, it can be concluded that there is a statistically significant difference between the protein levels of each of our experimental groups. Post-hoc, a Bonferroni Significance Test was performed, and the adjusted T Test value to achieve significance was 0.00833. Between each pair of trials (a total of 6 comparisons), the T Test values were well below the 0.00833 margin, peaking at 1.51 x 10-5. Therefore, it is statistically supported that LPS is capable of increasing the levels of inflammotin in the blood of human subjects. Rat TrialFor the Rat trial, there were only two trials done, a 0 mg of LPS trial and a 10 mg of LPS trial. A T Test was performed in order to determine whether the difference in means between the two trials was statistically significant. The T Test returned a p-value of 0.87, far above the acceptable maximum p=0.05 margin for statistical significance. Therefore, it can be concluded that any differences between the 0 mg dose of LPS and the 10 mg dose of LPS in rats is very likely due to random chance.
Summary and DiscussionThe Human Study data shows how different dosages of LPS will have different effects on inflammotin levels. The ANOVA test resulted in a p-value of 3.77*10-14, which is lower than the upper-margin uncertainty value of p=0.05. Because of the low p-value, the ANOVA test concluded that the variation between each trial was statistically significant. Therefore, the human study data was showed a conclusive relationship between the dosage of LPS and the levels of inflammotin in human subjects. This result is shown in the Human Trial bar graph, in which the error bars of each bar do not overlap, implying that the difference between the trials cannot be attributed to random variation. This conclusion was supported by by the Bonferroni Significance Test, which further concluded that, when compared individually, the difference between each trial and each other trial was statistically significant as well. The rat study was inconclusive. After performing a T Test, the test returned a p-value of 0.87, a result of the extremely large within-group variation in both trials. This p-value carries the implication that, because the measurements or experiment as a whole was imprecise, there is an 87% chance that the difference between the two is due to random chance. The 0 mg dose sample has a standard deviation of 2.225 pg/ml, and the 10 mg sample has a standard deviation of 7.40 pg/ml. The difference between the average inflammotin level as a result of each LPS dose was only 0.596 pg/ml, while both standard deviations were well above this value. This particular result, as also displayed by the rat trial bar graph, shows that the standard deviation within each group drowns out the small difference between the average of each trial. In conclusion, the rat trial of the effects of LPS on inflammotin levels was inconclusive. Between the two tests, only the human trial showed any statistically significant result linking the dose of LPS given to patients and the levels of inflammotin in their blood. This result is likely due to the biological differences between rats and humans; LPS may have no effect on rats, but it conclusively raises inflammotin levels in human subjects. This also may be a result of a flaw in the rat trial, perhaps involving difficulties in measuring blood inflammotin levels in such small mammals, or involving the very small sample size included in the experiment. In context with the potential uses of LPS treatment, the successful human trial opens up the possibility of more extensive clinical research into the medicinal uses of LPS, perhaps itself leading to the eventual development of a useful new drug.
|
|||||||||||||||||||||||||||||||||||||||