BISC209/F13: Lab4
LAB 4: Community Level Testing for Functional Metabolic Diversity/ Structural Diversity in Cultured Isolates
Community Level EXOENYMES PREVALENCE con't:
In Lab 3 you started a quantitative assessment of the prevalence of microbial digesters of cellulose, starch, and for solubilizers of phosphates. Think about why we chose to test for your soil community's ability to process these particular nutrients. Why would it be efficient for a soil microbial community to include members that secrete enzymes that process starch, cellulose or phosphorus into other forms of nutrients? Would it be better if all of the community members possessed all of these abilities? Why or why not? What might be the cost? Would the microbial community be different if NONE of its members secreted these exoenzymes? If microbial community membership is always in flux, trying to maintain a balance where its members can all survive and thrive, do you think the prevalence of these exoenzyme producers is constant or variable? If variable, what are the conditions that might have an effect on prevalence?
Today you will complete the colony counts from the differential media that you inoculated with dilute soil extract last week. You and your group members will perform calculations that assess the prevalence of community members able to perform these valuable functions. Your data should provide evidence of co-operative behavior among members of this soil community.
Assessment of Digestive Exo-Enzymes
Examine the plates for evidence of digestion or processing of a particular nutrient (starch, cellulose or insoluble phosphates) in each of the differential culture medium. Remember that these differential media are not selective (they aren't designed to inhibit the growth of any groups of soil microorganisms), but they are differential media, in that they allow you to visibly SEE the difference in particular groups of microbes---in our case, between those that produce and secrete a functional exoenzyme and those that don't. You will count the number of individual colonies showing a clear zone (halo) around the colony (using the plate with 30-300 total colonies) and compare those numbers with the number at the same soil dilution that grew on NA- a general purpose, non-differential medium. Why does a halo indicate digestion of starch, cellulose, or processing of insoluble phosphate into a soluble form?
1. Colony Count on Nutrient Agar (NA) General Purpose Medium as a Control
(0.3% Beef extract, 0.5% Peptone, 1.5% Agar at pH 6.6- 7.0 at 25°C) is used to determine comparative number of total culturable bacteria:
ANALYSIS::
Choose the dilution that has between 30-300 colonies, and count the colonies on the two replicates. Record this number on the plate as well as in your lab notebook. Use the soil extract dilution of the plates counted to calculate CFU/gram of soil (wet weight) for each assessment medium. If you divide the number of colonies counted by the amount of inoculum plated times the dilution factor of that plate, you will obtain the number of cultivatable bacteria per gram of wet soil.
Number CFU/dilution plated*dilution factor = number of CFU/gram
For example, if you counted 150 colonies on the 10-3 plate the calculation is:
150/(0.1ml plated*1X10-3dilution)= 150X104 which in scientific notation is written as 1.5X106 CFU/gram
How did your count on this soil extract compare to the count made in Lab 2 from a different soil sample at the same location? Should they be relatively the same? Are they? You will use the calculation on the new soil sample in your calculations of the prevalence of starch and cellulose digestors, and for the phosphate solubilizers in the community.
2. Starch Medium (2.5%(wt/vol) soluble starch in Nutrient Agar) is used to determine the % of amalyase producing, starch digesting culturable microbes when compared to the total number counted in NA:
ANALYSIS::
Choose the dilution that shows 30-300 colonies. Flood the replicate plates with Grams Iodine. Let the plate sit for at least 10 minutes, longer is fine. The plate should turn dark blue as the iodine binds to the starch; Pour off the excess iodine (into paper towels in the biohazard bag on your bench) and wait until the remaining liquid is absorbed by the plate. Count the colonies that show a clear halo, indicating the starch in the medium was digested by amylase secreted from the bacteria in the colony. Count the number of colonies that are able to digest starch (clear zone around the colony). To calculate the % of culturable microbes able to digest starch/CFU in a gram of wet soil you will use the Average total number of colonies calculated from your NA plate count. Make sure that both counts were made on the same dilution. If not, you will have to do a conversion.
Reference: Beishir, Lois. 1996. Microbiology in Practice 6th ed. HarperCollins Publishers Inc. New York. Module 33: 301-306.
3. Cellulose Congo Red Agar Medium is used to determine the % of cellulolytic microbes (those producing cellulase and able to digest cellulose)
ANALYSIS::
Choose the dilution that shows 30-300 colonies. Count the colonies on the replicate plates that show a clear halo, indicating the cellulose in the medium was digested by cellulase secreted from the bacteria in the colony. The red color of the medium provides increased contrast between the cellulose containing medium and the halo or clear zone that indicates that cellulase has diffused out of bacterial producers and degraded the cellulose in the immediate vicinity. Count the number of colonies on the plate that are able to digest cellulose (clear zone around the colony). To calculate the % of cellulolytic microbes/CFU in a gram of wet soil you will use the total number of colonies calculated from your NA plate count. Make sure that both counts were made on the same dilution. If not, you will have to do a conversion.
Cellulose Congo Red Agar:
0.05% K2HPO4; 0.025% MgSo4; 0.188% ashed, acid washed cellulose powder; 0.02% Congo red, 1.0% Noble Agar, 0.2% gelatin, 10%(vol/vol) sterile soil extract(10.5% air-dried, sieved soil that's autoclaved twice, allowed to settle, and the supernatant filtered); final pH 7.0 - 8.0.
Reference: Hendricks, C.W., Doyle, J.D., Hugley, B. (1995) A new solid medium for enumerating cellulose-utilizing bacteria in soil. Applied and Environmental Microbiology 61, 2016-2010.
4.Phosphate Medium (Pidovskaya medium [PVK]) is used to determine the % phosphate solubilizing microbes (those producing phosphatases) in a soil community:
ANALYSIS::
Choose the dilution that shows 30-300 colonies. Count the colonies that show a clear halo on each of the replicate plates, indicating the phosphate in the medium was solubilized and processed by enzymes secreted from the bacteria in the colony. Count the number of colonies that are able to solubilize phosphate (clear zone around the colony). To calculate the % of culturable, phosphate solubilizing microbes /CFU in a gram of wet soil you will use the total number of colonies calculated from your NA plate count. Make sure that both counts were made on the same dilution. If not, you will have to do a conversion.
Pidovskaya Medium Modified (Nautiyal, 1999)
1% glucose; 0.5% Calcium Phosphate [Ca3(PO4)2; 1% MgCl2.6H2O; 0.25% Magnesium Sulfate (MgSO4.7H20);0.2% (NH4)2SO4; 0.25% KCL; 0.0025% BromoPhenol Blue; 1.5-2% agar
References: Pikovskaya, R.I. 1948. Mobilization of phosphorus in soil in connection with the vital activity of some microbial species. Microbiologia 17, 362-370>
Pranjal Baruah (2007)Isolation of phosphate solubilizing bacteria from soil and its activity. Available at: Biotechindia.files.wordpress.com/2007/12/isolation.pdf.
You will calculated the mean prevalence (in %) of each type of exoezyme secreting microbes out of the total in the culturable community using the formula (# positive colonies x dilution factor/total colony count x dilution factor on nutrient agar) X 100. The correction for dilution factor allows you to compare the CFUs counted from different dilutions on plates. If you are able to use control (NA) and test plates from the same dilution, you can omit the dilution factor. Be sure to show all your data and calculations in your notebook and enter your data in the lab Enumeration Google Doc. Follow your instructor's directions concerning analyzing these data.
Analysis of Carbon Source Utilization Data
You have been asked to calculate Community Metabolic Diversity (CMD). CMD is a simple way to represent the total number of substrates able to be effectively metabolized by the microbial community. It's a measurement of diversity in use of carbon sources. Clearly a quantitative evaluation of CMD is more meaningful in relationship to other soil communities or when compared to some standard or reference point. If you are only analyzing your own soil community and have no other communities to compare, the reference comparison that you should use to make this quantitative evidence meaningful is to calculate a percentage of substrates used by the community (CMD) out of the total number possible (the number of substrates on the plate).
Calculating Community Metabolic diversity (CMD) & Percent of Carbon Sources Utilized
Your group should have a week of daily measurements recorded on the Excel workbook we provided as a template. This template is pre-formatted for the first set of calculations you will do from these data. It includes the formulas to average replicate measurements each day and it will automatically subtract the background (readings in the water wells). There is a normalization for background that will be subtracted automatically (this threshold absorbance is provided by the manufacturer and was determined to be 0.25 absorbance units for each carbon source). These calculations are the first step in figuring out what you can learn from these data that provides evidence for one or more of our investigative goals.
CMD is calculated by summing the number of positive responses (wells with a positive A595nm value after all the corrections) at each reading day. On your worksheet enter for each carbon source either a 0 or a 1. One indicates a positive value for absorbance at A590nm after correction for background. (Remember that this correction is built into the formulas embedded in the template spreadsheet.) Zero should be entered for carbon sources that have either a negative value or a zero (0.0) absorbance after the built-in correction. Zero means that the community was unable to use that particular carbon source for its metabolic needs. Once you have entered a 1 or a 0 for each substrate, the template will sum the positive values. This number is your CMD for that day. It indicates the number of substrates usable by the community. Enter your daily CMD values into indicated column on the final page (CMD graph) of your worksheet template. A graph should be automatically generated showing average CMD on the y axis versus time (day) post-inoculation. Use the peak CMD value to calculate % of carbon sources utilized [number utilized divided by the total number available(31) x 100].
What to do with your data:
In lab today brain storm with your partners about how you will use these results in a figure/table and in the results narrative to help the hypothetical reader of your research report on this investigation to understand what your data mean in terms of our experimental questions. You will analyze these data and think of creative ways to make effective tables/figures with appropriate legends.
More information about how to analyze these data and on how to use your data as evidence for conclusions to our experimental questions can be found in the description of Assignment 4 .
Isolation of Interesting and Diverse Bacteria
Continue to isolate to pure culture interesting bacterial members of your soil community.
If you are interested in one of the organisms that produces an exoenzyme you might choose one of these colonies today and start isolation to pure culture. You can use the colony selected for the simple staining procedure even though you cannot consider this a pure culture yet. You will each eventually study two cultures but some cultures may die over time or might not be diverse enough for your site. You will need to isolation streak at least 3-4 times to make sure you have a pure culture of any new organisms selected today before performing the structural tests included in future labs.
Directions for Streaking for Isolation onto new solid media is found at Streaking for Isolation
Your goal is for each student to end up with ~2 pure cultures of genera of bacteria that are different from each other and different from those of your teammates.
Once you believe you have pure isolates, continue to subculture to fresh NA plates each week (isolation streak a colony onto a fresh plate).
Assessing Bacterial Morphology by Simple Stain
Today you will make smear slides (explained here and in Protocols as Smear Slide Preparation) and perform a simple stain (found here and in protocols as Stains: Simple Stain). You will visually inspect your isolates to see if they are bacteria, not yeast, and to see if they appear to be pure based solely on morphology.
Background on Using Stains in Bacteriology
The first of the dyes most useful to bacteriologists was a reddish violet dye, mauvein, synthesized in England by William. H. Perkin, and patented by him in 1856. This synthetic dye and others were immediately appreciated by histologists, but were not applied to bacterial cells until Carl Weigert (a cousin of Paul Ehrlich) used methyl violet to stain cocci in preparations of diseased tissue in 1875. Subsequently, the use of various synthetic dyes for bacteriological preparations developed rapidly when they were promoted through the publications of Robert Koch and Paul Ehrlich.
The synthetic dyes are classified as acid dyes, or basic dyes, depending on whether the molecule is a cation or an anion. The introduction of the terms acidic and basic was unfortunate because it would be more revealing to refer to them as cationic or anionic dyes. A look at the structural formula reveals the nature of the dye.
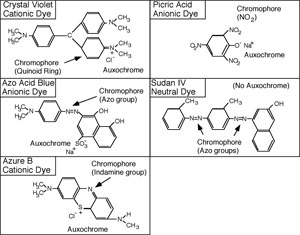
Each dye molecule has at least two functional chemical groupings. The auxochrome ionizes and gives the molecule the ability to react with the substrate, while the unsaturated chromophore absorbs specific wavelengths of light. The color of the solution obtained is that of the unabsorbed (transmitted) light. To be a dye, the molecule must have both auxochrome and chromophore groups. The auxochrome is usually an ionized carboxyl, hydroxyl, or pentavalent nitrogen group. The chromophore may have unsaturated nitrogen bonds such as azo (-N=N-) indamine (-N=), nitroso (-N=O) or nitro (O-N=O), groups; or unsaturated carbon to carbon, carbon to oxygen, or carbon to sulfur bonds, such as ethenyl (C=C), carbonyl (C=O), C=S, or the quinoid ring (= = =).
Cationic dyes will react with substrate groups that ionize to produce a negative charge, such as carboxyl, phenolic, or sulfhydryl groups. Anionic dyes will react with substrate groups which ionize to produce positive charges, such as the ammonium ion. Any substrate with such ionized groups should have an ability to combine with cationic or anionic dyes. Generally, the most important staining substrates in bacterial cells are proteins, especially the cytoplasmic proteins; however, other substances also have dye affinity. These include amino sugars, organic acids, nucleic acids, and certain polysaccharides.
Resonance is also important to color. In crystal violet, the primary stain in a Gram stain, an electron resonates between the three benzene rings. As the pH of the solution is lowered, the resonance becomes more and more restricted. When the resonance is restricted from three to only two benzene rings, the solution turns from violet to green, and then to red when resonance between the two rings ceases.
Activity: Preparing a bacterial smear slide
1. Use a graphite pencil to label on the far left side of a clean, glass slide the code ID of three of your isolates. Make enough slides, with no more than 3 organisms/slide so that all your isolates are included and there is one space for a "control" organism EC (Escherichia coli).
2. Prepare the slide with the "control" first by using your (unsterile is OK) loop to apply three tiny drops of deionized water to different parts of this pre-labeled slide. Flame sterilize the loop, allow it to cool for a few seconds and touch the cooled loop to a single colony of the control culture plate of E. coli. Pick up a VERY TINY bit of growth from a single bacterial colony.
3. Place the loop with the bacterial growth into the drop of water. Use a circular motion with the loop to make a smooth suspension of the bacteria in the water. Stop when you have spread out the circle of emulsified bacteria as much as possible to cover a space about the size of a quarter, while making sure to leave room to make two other smears on the slide.
4. Now you are ready to apply your isolates. Using your best aseptic technique, flame sterilize your loop. Apply a very small loopful of deionized water (bottle on your bench) to the slide and then add a tiny amount of bacteria from a fresh culture growing on solid medium of one your isolates. Create the smear as you did for the E. coli. Move from left to right spacing the smears so that isolates fit on the slides. (NOTE: Broths can also be used to make a smear. In this case, be sure to mix the broth, carefully vortex or tap the bottom of the tube. Remove the culture tube top (NOT placing it on the bench) and pass the lip of the culture tube rapidly through the flame. Dip your loop into the broth culture and place a small drop of broth culture of that isolate on the appropriate place on the labeled slide. (The labeling from top to bottom should refer to isolates placed left to right.) Mix the drop with your loop to disperse the broth over an area that will not touch the next culture but is large enough to allow your slide to dry quickly.
5. Allow the smears to air dry completely! Be sure all the water on the slide has evaporated before proceeding to heat fixation!!! This drying step is crucially important. If you are impatient, you will "explode" the cells in the next step .
6. Heat fix (to kill and attach organisms to the slide) by passing the slide (smear side up) quickly through a flame 3 times. Use a clothes pin or slide holder to avoid contact with hot glass.
An example of a multiple smear labeled slide:

Simple Stain Procedure:
To simple stain the previously prepared heat fixed, bacterial smear slides, perform the staining protocol from start to finish on one slide at a time. You must be careful to apply the staining reagents liberally so all the smears are evenly and completely covered and you must be sure to expose each smear to each reagent for the same amount of time.
1. Place a heat-fixed bacterial smear slide on the staining tray. It is important that the slide is level during staining; therefore, you should use paper towels under the tray to level the slide. If you do this, it is much easier to ensure that your smears will be covered evenly with each reagent with as little waste of the dye as possible (a crucial aspect of proper staining results).
2. Dispense just enough of our red simple stain, safranin solution, to cover all the smears on your slide and let it sit for 1 minute.
3. Rinse the slide by lifting the slide at a 45 degree angle (using gloves or a clothes pin or slide holder) and use a squirt bottle to direct a very gentle stream of water slightly above the top smear. Rinse until the waste water coming off at the bottom is relatively clear. Drain off excess water by touching the edge of the slide to a paper towel.
4. Blot dry using the bibulous paper package found in your orange drawer. Do not tear out the pages, just insert your slide and pat it dry.
5. Clean up your area: Use paper towels to absorb the water and stain in your staining tray before throwing those paper towels in the trash can. Rinse your staining tray in the sink and leave it to drain upside down on paper towels.
Use of the Compound Light Microscope
Activity: View your stained bacteria and assess the purity of your cultured isolates
Refer to the directions for using your compound brightfield microscope BISC209/F13:_Microscopy found in the Protocols section. Today you will use only the 10x and 100x objectives. Remember also to read and follow the directions for care of this precision instrument (particularly on how to avoid getting immersion oil on any objective other than the 100x oil immersion lens).
CLEAN UP
1. All culture plates that you are finished with should be discarded in the big orange autoclave bag near the sink next to the instructor table. Ask your instructor whether or not to save stock cultures and plates with organisms that are provided.
2. Culture plates, stocks, etc. that you are not finished with should be labeled on a piece of your your team color tape. Place the labeled cultures in your lab section's designated area in the incubator, the walk-in cold room, or at room temp. in a labeled rack. If you have a stack of plates, wrap a piece of your team color tape around the whole stack.
3. Remove tape from all liquid cultures in glass tubes. Then place the glass tubes with caps in racks by the sink near the instructor's table. Do not discard the contents of the tubes.
4. Glass slides or disposable glass tubes can be discarded in the glass disposal box.
5. Make sure all contaminated, plastic, disposable, serologic pipets and used contaminated micropipet tips are in the small orange autoclave bag sitting in the plastic container on your bench.
6. If you used the microscope, clean the lenses of the microscope with lens paper, being very careful NOT to get oil residue on any of the objectives other than the oil immersion 100x objective. Move the lowest power objective into the locked viewing position, turn off the light source, wind the power cord, and cover the microscope with its dust cover before replacing the microscope in the cabinet.
7. If you used it, rinse your staining tray and leave it upside down on paper towels next to your sink.
8. Turn off the gas and remove the tube from the nozzle. Place your bunsen burner and tube in your large drawer.
9. Place all your equipment (loop, striker, sharpie, etc) including your microfuge rack, your micropipets and your micropipet tips in your small or large drawer.
10. Move your notebook and lab manual so that you can disinfect your bench thoroughly.
11. Take off your lab coat and store it in the blue cabinet with your microscope.
12. Wash your hands.
Assignment
Graded Assignment:
Assignment (20 points): Community Functional Metabolic Diversity/Co-operation in a Soil Community Results: Design and Analysis . Instructions: Evidence for Functional Metabolic Diversity/Co-operation in a Soil Community
To Do Before the Next Lab:
Someone in your sampling group must return to your soil sampling site on the morning of lab to obtain 2 small microfuge tubes full of fresh soil to do genomic soil DNA isolation. Because we only want SOIL microorganisms in this sample and NOT your body flora, you will have to be particularly careful not to contaminate the inside of the microfuge tube lid or the soil. You will find a "kit" of gloves, sterile microfuge tubes, sterile spoon or spatula in the lab. Please pick of these kits up before you go and be careful to select soil from the same sampling site as you collected previously for your other tests. Avoid the top few millimeters and fill your tube about 3/4 full. Bring this soil sample to lab with you.
Continue monitoring and following the appropriate protocols to isolate our targeted bacteria to pure culture. Please make a fresh plate of each of your isolates at least once (twice would be preferable) before your next lab. You must have pure cultures of your bacteria of interest by Lab 6.