BIO254:LTP
What is Long Term Potentiation?
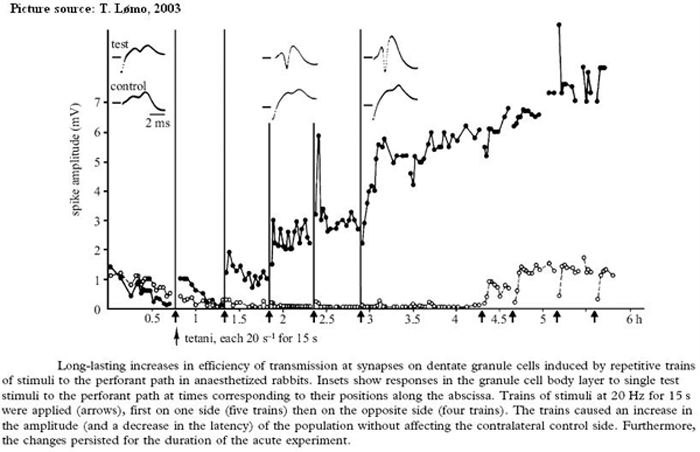
A short high-frequency train of stimuli to any of the three major synaptic pathways in the hippocampus increases the amplitude of the excitatory postsynaptic potentials in the target hippocampal neurons. This facilitation is termed long term potentiation (LTP). LTP modifies synaptic effectiveness, which contributes to neural plasticity and memory.
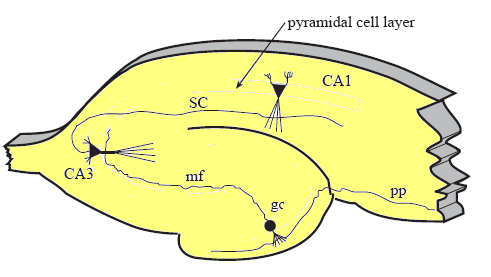
The three major synaptic pathways in the hippocampus where LTP is observed are:
- The perforant path (pp): from entorhinal cortex to the granule cells (gc) in the dentate
- Mossy fibers (mf): from granule cells in dentate to CA3 pyramidal neurons
- Schaffer collateral fibers (SC): from CA3 pyramidal neurons to CA1 pyramidal neurons
Although most of the research thus far has focused on LTP in the hippocampus, this process in known to occur in other areas of the mamallian nervous system.
[Cross section of mamallian hippocampus-- Personal communication with Robert Malenka]
Triggering of LTP
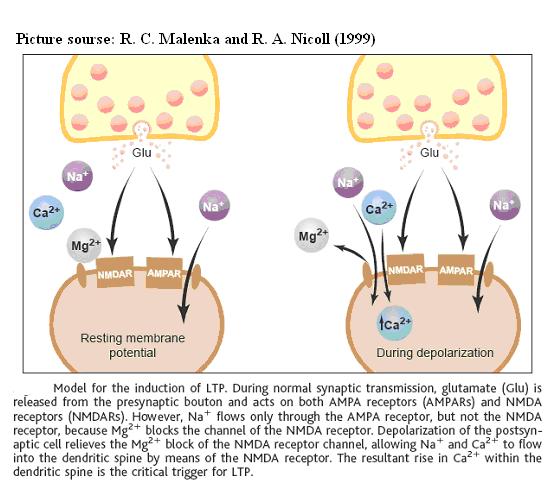
The triggering of LTP in the CA1 region of the hippocampus depends on an influx of Ca2+ through postsynaptic NMDA receptors, which are both ligand-gated and voltage gated. The binding of glutamate, the most prevalant excitatory neurotransmitter in the brain, induces the channel to adopt an open configuration; if the cell is at resting potential, however, ionic transfer is still prevented due to the presence of a magnesium ion in the channel pore. The Ca2+ influx is permitted only when the cell is sufficiently depolarized to relieve the Mg2+ block. The high concentration of Ca2+ then triggers a series of biochemical reactions which eventually modify the synaptic effectiveness. These enhancing changes can include an increase in the number postsynaptic receptors; an increase in the activity of existing receptors (via phosphorylation); a restructuring of the shape of the dendritic spine; and probably some kind of retrograde signaling to the presynaptic cell, increasing the efficacy of its signal transmission. However, whether Ca2+ alone is sufficient for the induction of LTP remains unclear.
Basic Properties of LTP
The four basic properties of LTP (frequency, input-specificity, associativity, and cooperativity) collectively state that in order for potentiation to occur, both the synapse and the postsynaptic neuron must simultanously be depolarized beyond a set threshold.
- LTP is input specific, which means that LTP enhances the synaptic effectiveness of certain synapses without affecting other synapses in the cell. Due largely to the compartmentalization conferred by the dendritic spine structure, this property greatly increases the storage capacity of individual neurons.
- LTP is cooperative, which means that weak stimulations in a single pathway can cooperatively induce LTP.
- LTP is associative, which means that weak stimulation in pathways, when coupled with strong stimulation in another, can induce LTP. Assoiativivity of the LTP can be viewed as a cellular analog of classical conditioning.
- Stimuli must be delivered at high frequency because cells must be depolarized past a certain threshold in order for LTP to occur
Two phases of LTP
LTP has a transient early (lasting 1-3 hours) and a consolidated later phase (at least 24 hours). The transient early phase does not require new protein synthesis. For example, in the early phase the conductance of the postsynaptic receptors is enhanced. The later phase requires new protein and RNA synthesis. For example, new presynaptic active zones and postsynaptic receptors can grow in the late phase.
Bolshakov and his colleagues show that the early and late phases of LTP are evident in the synaptic transmission between a single CA3 cell and a single CA1 cell. (Images and captions are adaped from Ref 2.)
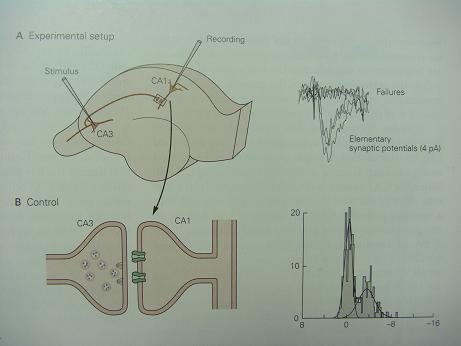
Experimental Setup: a single CA3 cell can be stimulated selectively to produce a single elementary synaptic potential in a CA1 cell. When the CA3 cell is stimulated repeatedly at low frequency, it gives rise either to an elementary response of the size of a miniature synaptic potential or to a failure. In control cells there are many failures, and the synapse has a low probability of releasing vesicles. The distribution of the amplitudes of many responses can be approximated by two Gaussian curves, one centered on zero (the failures) and the other centered on 4 pA (the successful responses.) These histograms are consistent with the type of synapse illustrated here, in which a single CA3 cell makes a single connection on a CA1 cell. This connection has a single active zone from which it releases a single vesicle in an all-or-none manner (failures or successes).
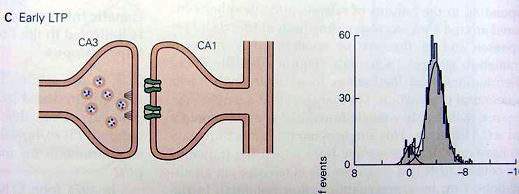
With the early phase of LTP the probability of release rises significantly, but the two Gaussian curves in the distribution of responses is consistent with the view that a single release site still releases only a vesicle but now with a higher probability of release.
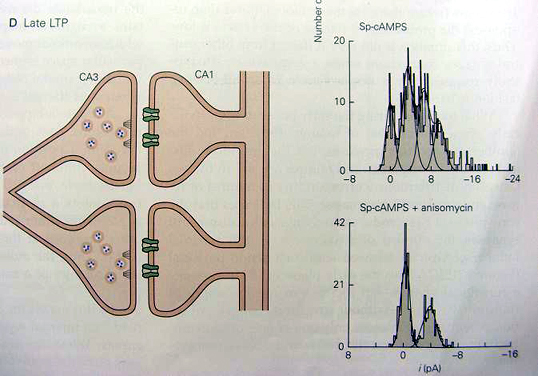
When the late phases of LTP is induced by a cAMP analog, the distribution of responses no longer fits two Gaussian curves but instead requires three or four Gaussian curves, suggesting the possibility that new presynaptic active zones and postsynaptic receptors have grown These effects are blocked by anisomycin, an inhibitor of protein synthesis.
LTP and Memory
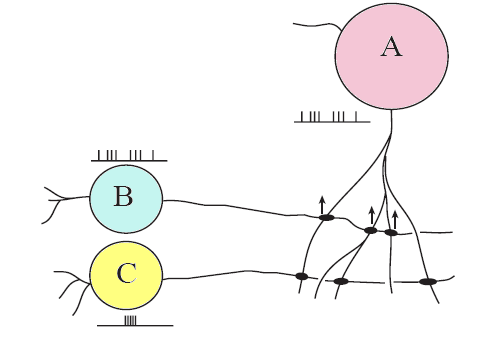
When neurons fire asynchronously, no changes in their synaptic connections are observed. However, when neurons fire in sync, the synapses between them are strengthened. These synaptic connections are called Hebbian synapses. The activity induced in synaptic efficiency at these synapses in the hippocampus is a result of LTP. Therefore, LTP is believed to play a role in the formation and storage of cognitive memory.
[Arrows show increased synaptic strength between neurons with identical firing patterns (A and B). No cahnge in synaptic strength is observed when the firing pattern of neurons is different (A and C).--Personal communication with Robert Malenka]
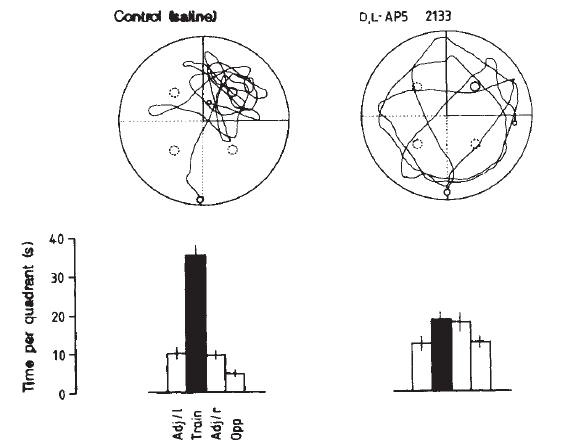
The strongest evidence supporting the role of LTP in memory formation comes from pharmacological and genetic studies. Moriss et. al studied the effects of NMDA receptor antagonists on memory formation in rats. They found that rats treated with AP5 (a NMDA receptor antagonist, and strong inhibitor of LTP) showed deficits in memory formation, as compared to controls, during a water maze task.
[Rats were treated with either AP5 or saline and trained to find a platform in a pool of opaque water. Swimming paths taken by rats in control and AP5 treatment group are shown. Computerized tracking systems compared the amount of time each animal spent in each quadrant of the tank and scores for each group were tallied. The mean time spent in the training quadrant were: Control- 35.4 + 2,2s (59%); AP5- 18.2 + 2.0 s (30%). Morris et. al, 1986]
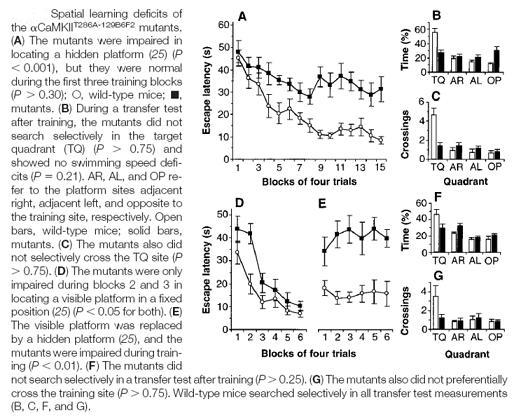
Similarly, Giesse et. al showed that mice lacking the CaM kinase gene also showed similar deficits in spatial memory. CaM kinase has been associated with induction of LTP.
[Mutant mice had a point mutation introduced into their CaMKII gene at Thr 286 and showed no NMDA-mediated LTP during spatial learning tasks in the Morris water maze. Giese et. al, 1998]
Signal Transduction in LTP
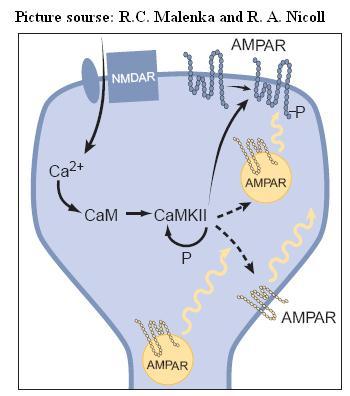
Signal transduction in LTP is very complicated and many candidate molecules have been identified. However, only a few molecules play a mandatory role in signal transduction in LTP. One of them is α-calcium-calmodulin– dependent proteinkinase II (CaMKII). Postsynaptic inhibition of CaMKII or a genetic deletion of a critical subunit of CaMKII stops the induction of LTP. During the transient phase of LTP, CaMKII is activated by calcium-camodulin complex, and it enhaces the activity of existing AMPA receptors, phosphorylates intracellular AMPARs and activate Syn GAP and the MAPK cascade, which facilitates the insertion of new AMPARs into the postsynaptic membrane. Several other protein kinases, including protein kinase C (PKC), cyclic adeonosine 39,59-monophosphate (cAMP)–dependent protein kinase (PKA), the tyrosine kinase Src, and mitogen-activated protein kinase (MAPK), have also been suggested to contribute to LTP.
Current Debate
Whether the increase in synaptic strength is primarily due to pre or postsynaptic effects is currently under debate. Current experimental data supports both. For instance, in the transient phase of LTP, there is an increase in the sensitivity and number of postsynaptic non-NMDA (AMPA) receptors to glutamate; also there is an increase in the probability of neurotransmitter release from the presynaptic side.
Reference
1. Terje Lømo (2003). "The discovery of long-term potentiation". Philos Trans R Soc Lond B Biol Sci 358 (1432): 617-20. PMID 12740104.
2. E.R. Kandel, J.H. Schwartz, T.M. Jessell, "Principles of Neural Science." 4th edition.
3. R. C. Malenka and a. R. A. Nicoll. "Long-Term Potentiation--A Decade of Progress?" Science, September 17, 1999; 285(5435): 1870 - 1874.
4. Lynch, MA. Long-Term Potentiation and Memory. Physiol Rev 84: 87–136, 2004; 10.1152/physrev.00014.2003
5. Giese KP, Fedorov NB, Filipkowski RK, Silva AJ. Autophosphorylation at Thr286 of the alpha calcium-calmodulin kinase II in LTP and learning.
Science 1998; 279: 870–3.
6. Morris RG, Anderson E, Lynch GS, Baudry M. Selective impairment of learning and blockade of long-term potentiation by an N-methyl-
D-aspartate receptor antagonist, AP5. Nature 1986; 319: 774–6.
List of abbreviations:
- N
- This edit created a new page (also see list of new pages)
- m
- This is a minor edit
- b
- This edit was performed by a bot
- (±123)
- The page size changed by this number of bytes
5 June 2026
4 June 2026
3 June 2026
| 21:28 | CHIP:Talks diffhist +545 Gabor Balazsi talk contribs | ||||
|
|
15:53 | User:Johannes W. Dietrich 3 changes history −220 [Johannes W. Dietrich (3×)] | |||
|
|
15:53 (cur | prev) +44 Johannes W. Dietrich talk contribs (→Research interests) | ||||
|
|
15:50 (cur | prev) +83 Johannes W. Dietrich talk contribs (→Contact Info) | ||||
|
|
15:47 (cur | prev) −347 Johannes W. Dietrich talk contribs (→Contact Info) | ||||
2 June 2026
|
|
21:32 | Hu:Members 2 changes history +439 [Hugangqing (2×)] | |||
|
|
21:32 (cur | prev) +389 Hugangqing talk contribs | ||||
|
|
21:31 (cur | prev) +50 Hugangqing talk contribs | ||||
| m 16:48 | Gerber:Publications diffhist +56 Andre P. Gerber talk contribs | ||||
Briefs of Long Term Potentiation.