Angela A. Garibaldi Week 9
From OpenWetWare
Jump to navigationJump to search
HIV Structure Assignment
- Ho YS, Abecasis AB, Theys K, Deforche K, Dwyer DE, Charleston M, Vandamme AM, and Saksena NK. HIV-1 gp120 N-linked glycosylation differs between plasma and leukocyte compartments. Virol J. 2008 Jan 23;5:14. DOI:10.1186/1743-422X-5-14 |
Predicting the Secondary Structure of a Protein Sequence Exercises
- Prepare protein in FASTA formatted .txt file. You can find this on the NCBI Web Server NCBI
- Go to Protein Structure Prediction
- Click on Click here to access the server
- Copy your sequence and paste it into the input sequence window. Both FASTA and plain (with no header) formats are accepted.
- Keep the default pre-selected option in the Choose Prediction Method section
- Keep the default pre-selected option in the Filtering Options
- Enter your email address in the Submit Sequence section. There will also be a section where you can input a short name for your sequence for recognition.
- Click the Predict button.Results are sent my email in less than 30 minutes. It will also tell you how many jobs are currently in front of you in the processing queue. Server does not accept free email addresses.
Setting up the Experiment
- This time, we will be taking the same clones as we used from the HIV Evolution assignment from the same subjects and asking the same question, but in relation to the actual structure based on amino acid sequence of the clones. In order to narrow things down we will perform the following tasks:
- Upload clones' amino acid sequences from the first 5 visits that actually have clones from Subjects 5,6,7,10,13 to Biology WorkBench3.2
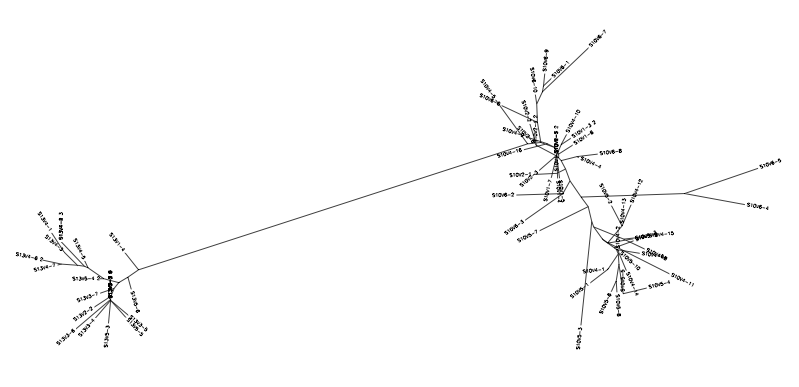
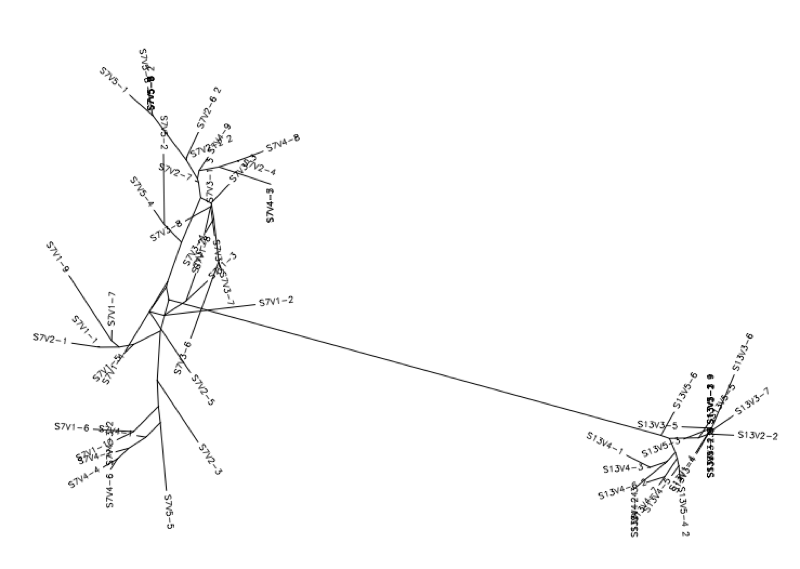
- Conduct a multiple sequence alignment between the following pairings used from the last experiment: 7-10,7-5,10-5; 6-7; 6-13,6-5,13-5 and the following as new comparisons: 13-10,6-10, 7-13.
- From here we will look at these results for any interesting alignments in relation to our past experimental results on the evolution.
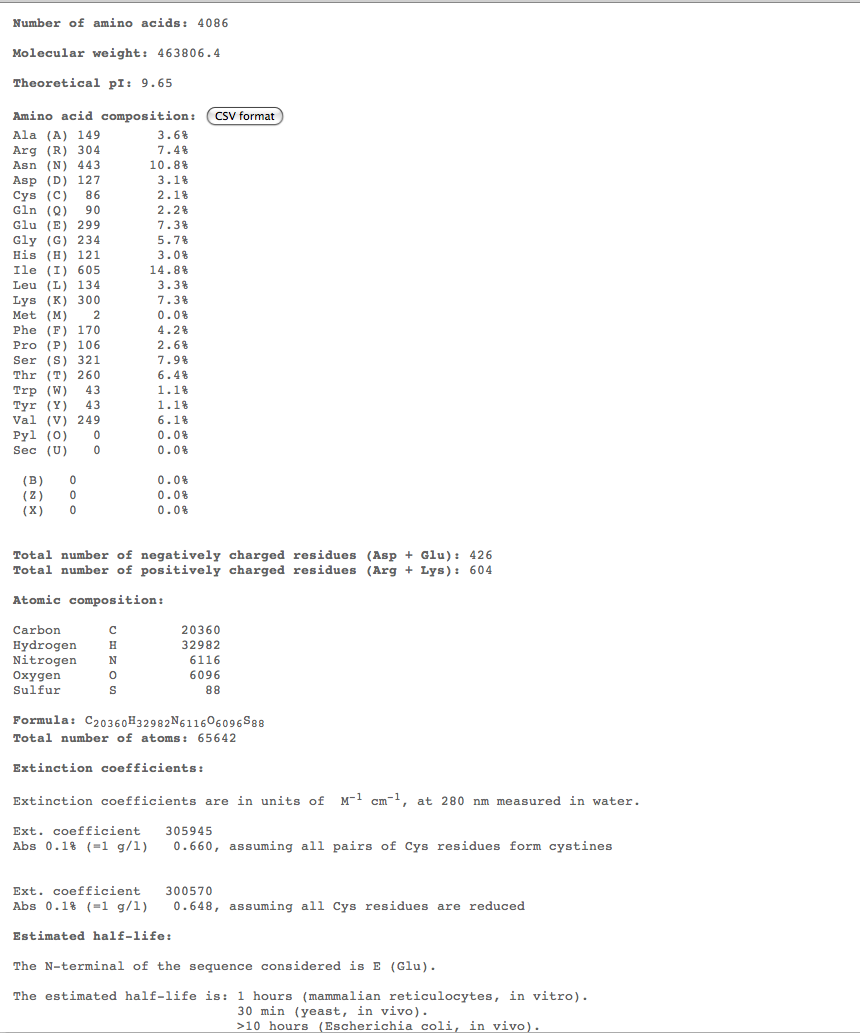
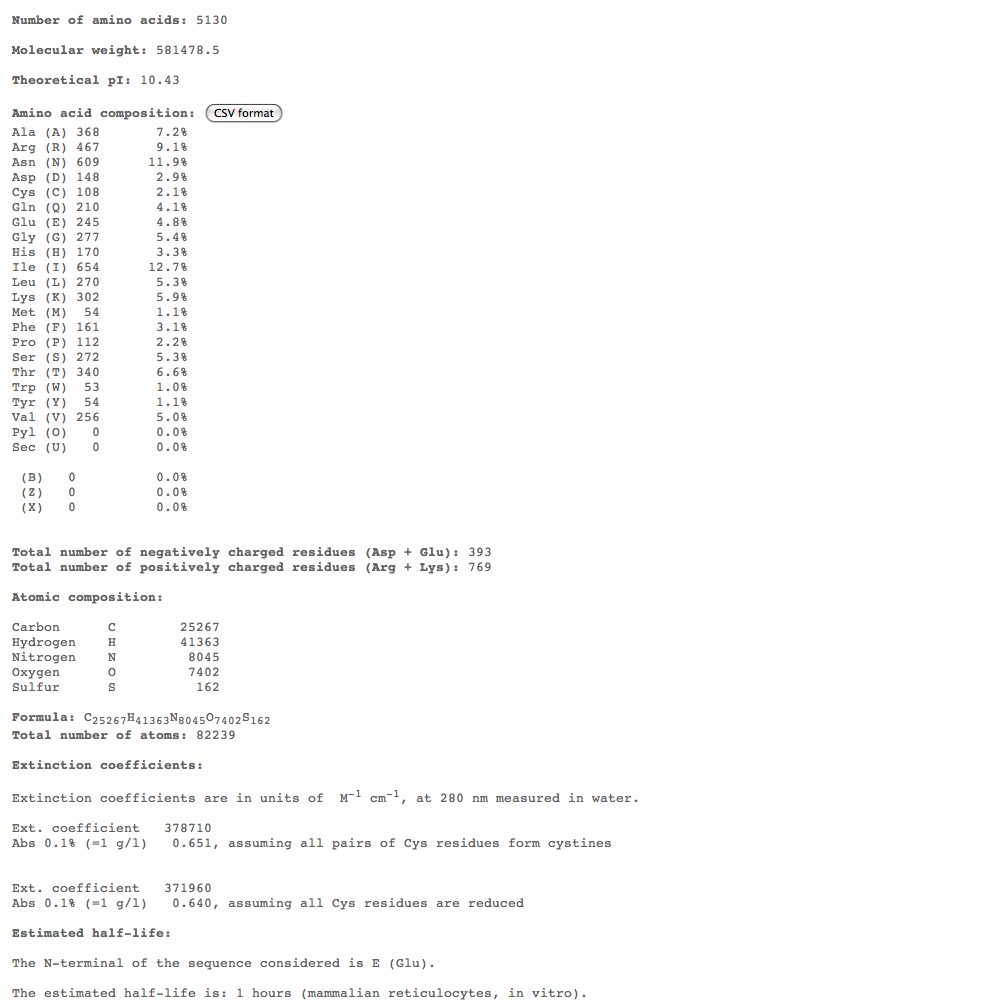
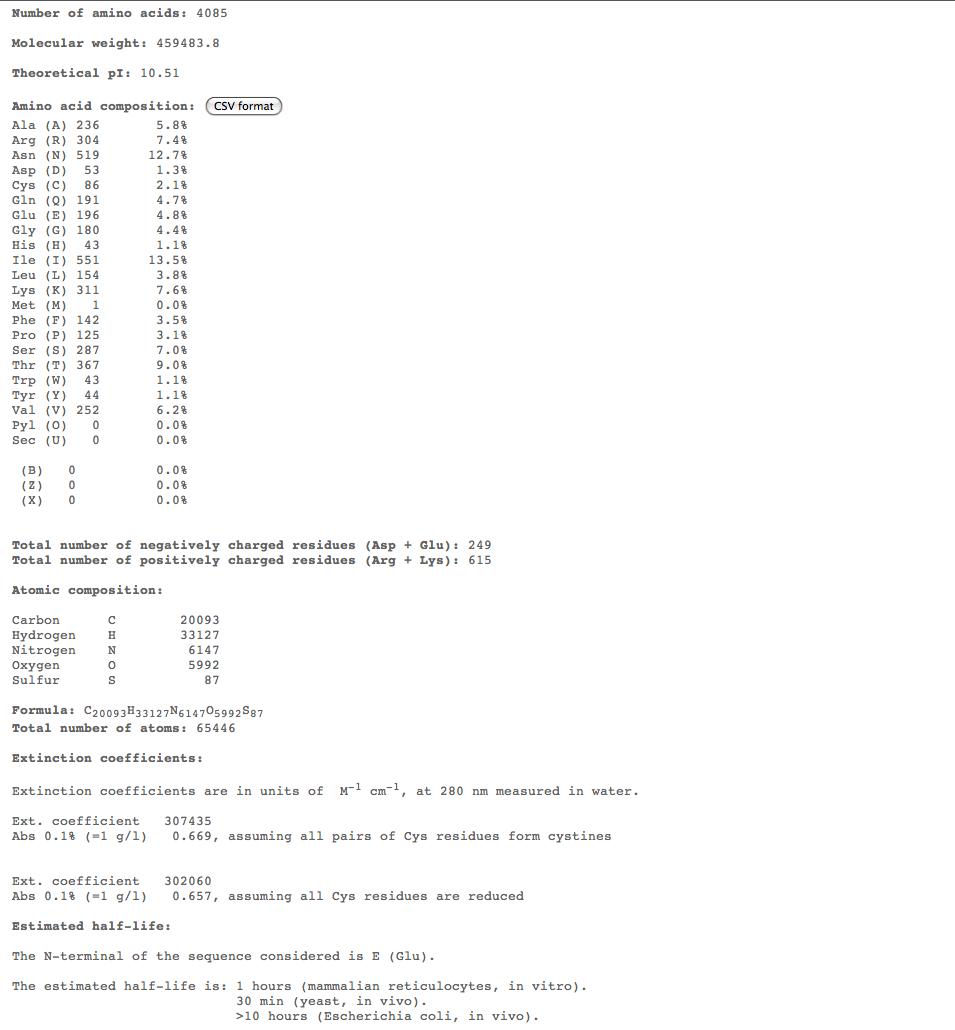
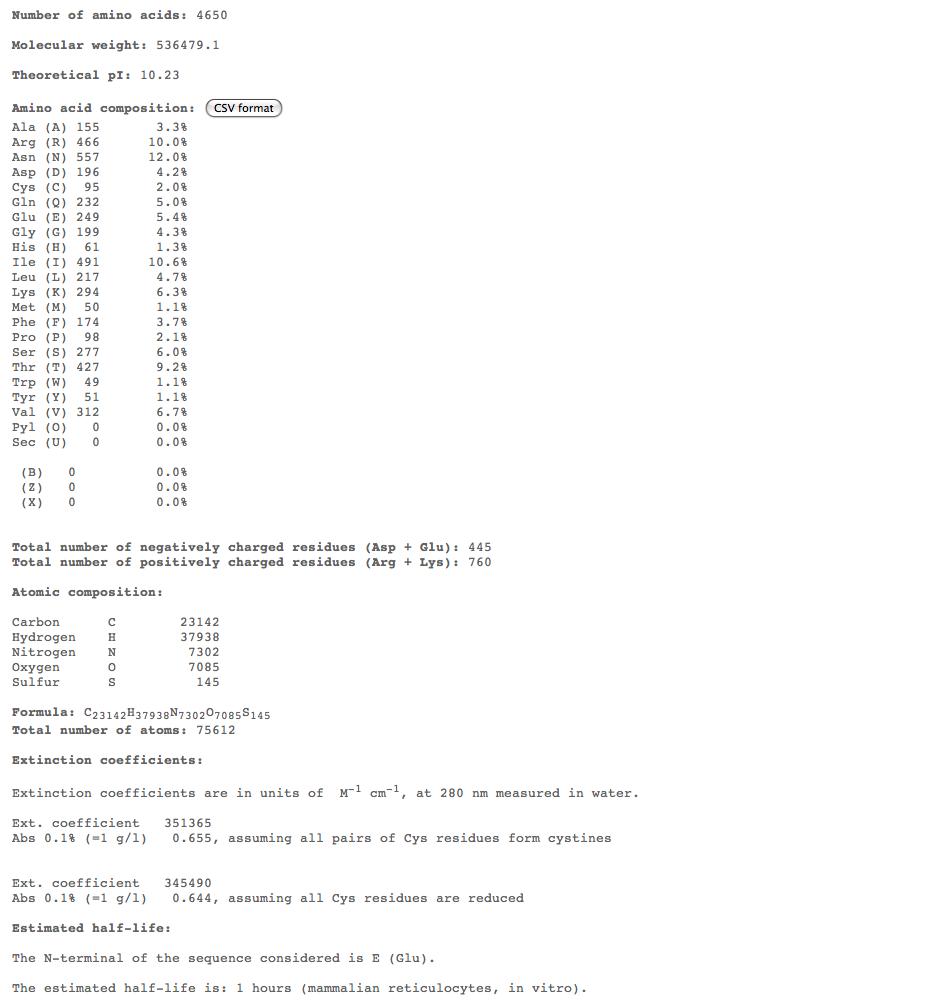
- For the more interesting alignments, we plan on conducting multiple comparisons of structure over 3 time points to compare the structures of these subjects over time. For this we could use ProtParam ProtParam to compare physical-chemical properties for these time points
- 3d structure and Post-Translational Modifications to predict modifications of the protein via Scan Prosite.
- If results look promising between a trial comparison between two subjects and their time points, we will take a look at the transmembrane segments using either ProtScale or TMHMM TMHMM which is more precise but does not predict segments in the middle; only those outside or inside the cell.
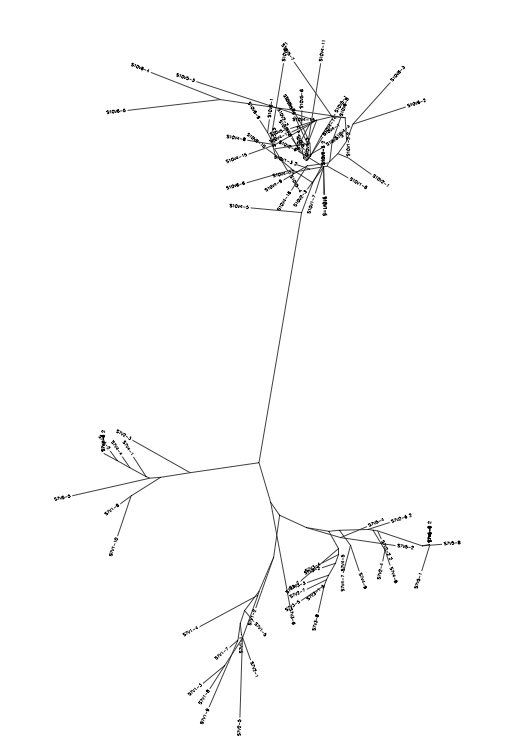
Phylogenetic Tree Results
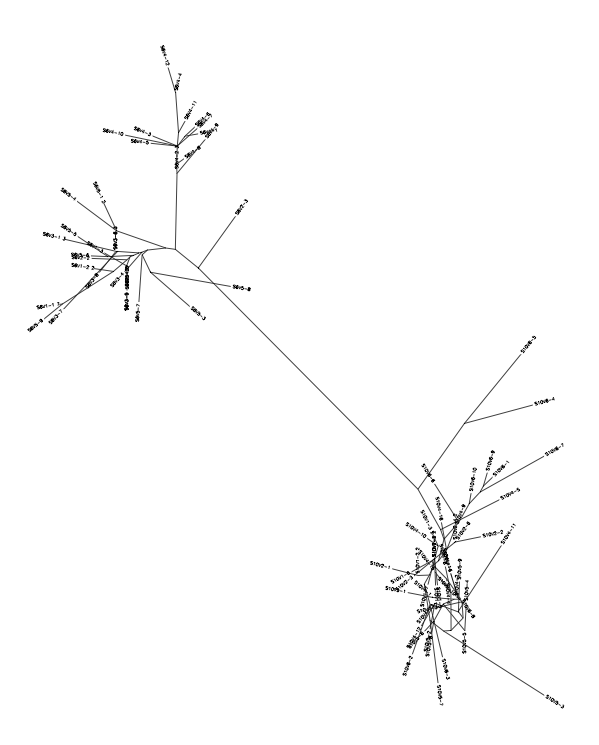
- From the subject 7 comparisons, it is clear that overall distance from 7vs5 is identical to 5vs 10, hinting that 7 is similar to 10 when compared to 5 (the control Moderate).
10 vs 7
Subject 6 vs 13
Subject 6 vs 5
Subject 13 vs 5
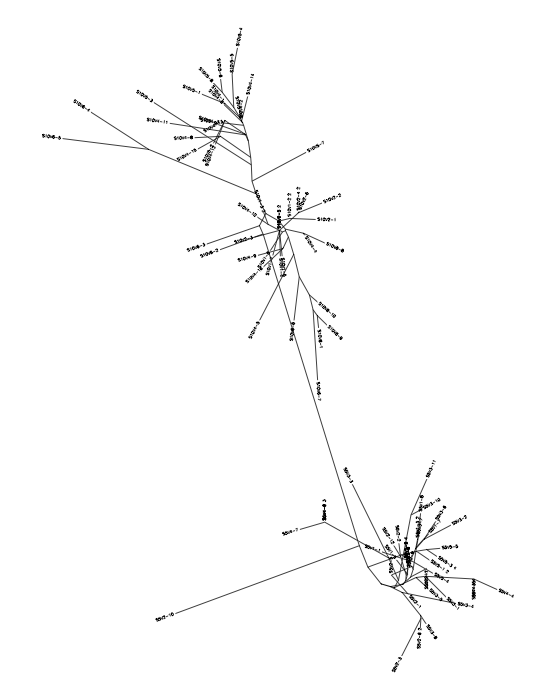
Comparisons not Previously Made
- 13 (non progressor) vs 10 (Standard Rapid Progressor)
- 6(Test Moderate progressor- Non Progressor) vs 10 (Standard Rapid Progressor)
- 7(Test Moderate Progressor-Rapid Progressor) vs 13 (Standard Non-Progressor)
File:Hivproteinstructure.ppt Powerpoint Presentation
Results
Subject 5
Subject 7
Subject 10
Navigation
Journal Links
Personal Journal
- Angela A. Garibaldi Week 2
- Angela A. Garibaldi Week 3
- Angela A. Garibaldi Week 4
- Angela A. Garibaldi Week 5
- Angela A. Garibaldi Week 6
- Angela A. Garibaldi Week 7
- Angela A. Garibaldi Week 8
- Angela A. Garibaldi Week 9
- Angela A. Garibaldi Week 10
- Angela A. Garibaldi Week 11
- Angela A. Garibaldi Week 12
- Angela A. Garibaldi Week 13
- Angela A. Garibaldi Week 14
- Angela A. Garibaldi GS Papers 1