3D Printing in the Vascularization of Microfluidics - Daniel Bell
Introduction
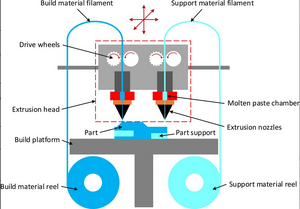
The vascularization of microfluidic devices is the creation of channels within a device capable of transporting blood in one end and completely out the other. As blood vessels range in size from the smallest capillaries of 5 micrometers to the aorta of 25 mm, microfluidic devices are excellent tools in order to provide the proper channels for blood flow [2]. The first generation of these devices relied on 2D manufactured devices stacked on top of one another in order to create a 3D vascularized device [3]. Now technology allows for the creation of detailed 3D vascularized microfluidic devices with the proper channel size for the type of blood vessel being recreated and type of tissue being replaced.
The vascularization of microfluidic devices has a vast number of applications as the ability to successfully move blood in an artificial tissue construct is important in the effectiveness of the device. Vascularized microfluidic devices can be used in both the medical field as a tissue implant in a number of areas of the body or for more research applications as a way of studying blood flow in vitro [1]. A number of considerations must be taken into account when creating these devices because of their use within the body. Biocompatibility of the materials used in these devices is very important as cells must adhere to the surface of the device and each device should accurately model the body [2]. Over the past few decades, several different techniques for the construction of vascularized microfluidic devices have been developed including traditional methods of construction using photolithography[1] and soft lithography[2] as well as emerging technologies of 3D printing through both dissolvable sacrificial moldings and hybrid method bioprinting. As these technologies continue to develop, vascularized microfluidic devices will continue to grow in importance for organ and tissue repair.
Technologies
Current 3D printing for microfluidic devices [3] has significant research investment and specifically for vascularization, the typical technologies used can be split into 2 main categories: sacrificial molding where a dissolvable material is 3D printed into the desired shape of the channel and later removed leaving the channels and bioprinting which takes advantage of printing with many materials at once including inks containing cells [2] [1] [4].
Sacrificial Molding

In order to create complex networks of channels in 3D devices, 3D printing using a dissolvable substance such as collagen can be used [2]. First, the desired channel is created using traditional 3D printing techniques, followed by encapsulation in the desired 3D hydrogel, and finally the 3D printed dissolvable portion can be dissolved leaving a network of channels of any desired size. A typical example of the creation of a 3D device using a dissolvable substance is the creation of gelatin channels and printing the non-dissolvable scaffold in collagen or fibrin [2]. Another example is the use of a fugitive ink composed of an coblock organic polymer that can be melted and removed under a vacuum [5]. This method provides additional structure when initially printing as the fugitive ink has more stability than gelatin, but it is more difficult to remove and complete the sacrificial molding process. Materials and processes for sacrificial molding must be kept under control as cells living within the additional portions of the 3D device must remain healthy [5]. While the hydrogels required for the necessary mechanical properties in tissue and organs in the body are not very easy to 3D print in traditional ways, creating the channels as dissolvable structures offers a method to take advantage of affordable 3D printing for creation of vascularized devices [6]. A new technology called omnidirectional printing offers additional improvements to the complexity of 3D printed devices by removing the need to create one layer of the material at a time [2]. Dissolvable sacrificial technology has the potential to be very useful in creating vascularized microfluidic scale products through the use of 3D printing technology.
Hybrid Method Bioprinting
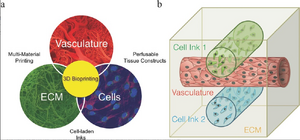
The use of bioprinting in order to create vascularized devices is a fairly new technology that uses the ability for 3D printers to extrude a number of components at once and extrudes an ink containing cells [1]. Multiple extrudates containing materials such as poly(dimethyl siloxane) (PDMS) and an aqueous mixture of various polymers that allow the cells to survive for the short time necessary for injection. One commonly used polymer is a triblock polymer composed of one part poly(propylene oxide) and two parts poly(ethylene oxide) called Pluronic F127 [1]. This material is inert to cells and can be washed away, leaving the healthy cells within the channels now created in the microfluidic device. One of the difficulties in 3D printed vascularized systems is the integration of cells after the printing, but the integration with the printing itself provides an exciting new method for successful vascularization of microfluidic systems [7]. The potential limitation with this technology is the size of the channels, since expensive printers with advanced crosslinking capabilities can produce channels close to the smallest blood vessels, but slightly less expensive printers with less options for materials or crosslinking will only be able to study a smaller range of blood vessel types.

Applications
A huge number of applications are available for 3D printing vascularized microfluidic devices in both in vivo and in vitro. As a tool in the laboratory, accurately recreating blood vessels on 3D printed devices would allow for important research in vitro before transitions are made to in vivo experiments [3]. Current technologies are fairly expensive when attempting to simulate systems like blood flow in small categories, but the low cost of 3D printing could help make more in vitro studies possible.
Microfluidic vascularized medical devices have major importance moving forward in organ and tissue engineering because of the need for blood to flow through implanted systems. These 3D printed devices need to have oxygen diffuse through them and creating blood vessels from the beginning throughout the chip can help with the integration of artificial tissue [1]. PDMS has high gas diffusion and vascularized microfluidic devices made of PDMS have enough oxygen diffusion to support cell growth. Studies looking to research the effects of hypoxic environments can use materials such as poly(methacrylic acid) which do not allow oxygen diffusion [8]. As this technology progresses and becomes cheaper and more accessible, 3D printed vascularized microfluidic devices will continue to grow in popularity and importance for these applications and more.
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 Kolesky, D.B.; Truby, R.L.; Gladman, A.S.; Busbee, T.A.; Homan, K.A.; Lewis, J.A. 3D bioprinting of vascularized, heterogeneous cell‐laden tissue constructs. Advanced materials, 2014, 26(19), 3124-3130. https://doi.org/10.1002/adma.201305506
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 2.7 Hasan, A.; Paul, A.; Vrana, N.E.; Zhao, X.; Memic, A.; Hwang, Y.S.; Dokmeci, M.R.; Khademhosseini, A. Microfluidic techniques for development of 3D vascularized tissue. Biomaterials, 2014, 35(26), 7308-7325. https://doi.org/10.1016/j.biomaterials.2014.04.091
- ↑ 3.0 3.1 He, Y.; Wu, Y.; Fu, J.Z.; Gao, Q.; Qiu, J.J. Developments of 3D printing microfluidics and applications in chemistry and biology: a review. Electroanalysis, 2016, 28(8), 1658-1678. https://doi.org/10.1002/elan.201600043
- ↑ Phillippi, J.A.; Miller, E.; Weiss, L.; Huard, J.; Waggoner, A.; Campbell, P. Microenvironments engineered by inkjet bioprinting spatially direct adult stem cells toward muscle‐and bone‐like subpopulations. Stem cells, 2008, 26(1), 127-134. https://doi.org/10.1634/stemcells.2007-0520
- ↑ 5.0 5.1 5.2 Wu, W.; DeConinck, A.; Lewis, J.A. Omnidirectional printing of 3D microvascular networks. Advanced materials, 2011, 23(24), H178-H183. DOI: https://doi.org/10.1002/adma.201004625
- ↑ Miller, J.S.; Stevens, K.R.; Yang, M.T.; Baker, B.M.; Nguyen, D.H.T.; Cohen, D.M.; Toro, E.; Chen, A.A.; Galie, P.A.; Yu, X.; Chaturvedi, R. Rapid casting of patterned vascular networks for perfusable engineered three-dimensional tissues. Nature materials, 2012, 11(9), 768. https://doi.org/10.1038/nmat3357
- ↑ Smith, C.M.; Stone, A.L.; Parkhill, R.L.; Stewart, R.L.; Simpkins, M.W.; Kachurin, A.M.; Warren, W.L.; Williams, S.K. Three-dimensional bioassembly tool for generating viable tissue-engineered constructs. Tissue engineering, 2004, 10(9-10), 1566-1576. https://doi.org/10.1089/ten.2004.10.1566
- ↑ Ochs, C.J.; Kasuya, J.; Pavesi, A.; Kamm, R.D. Oxygen levels in thermoplastic microfluidic devices during cell culture. Lab on a Chip, 2014, 14(3), 459-462. DOI: https://dx.doi.org/10.1039/c3lc51160j
