20.109(S14):DNA repair assays(Day6)
Introduction
The flow cytometry machine has revolutionized biology by allowing researchers to analyze and isolate cells based on their spectral qualities. Both genetic reporters and physical tags may be used to introduce fluorescence into cells. For example, if you have a fluorescently tagged antibody that preferentially binds to a unique cell receptor – or more likely, a set of antibodies that taken together identify a unique cell type – you can isolate a pure sample of this cell type from a complex mixture by using FACS: Fluorescence Activated Cell Sorting. Physical tags that are less specific than antibodies, such as propidium iodide, are also used in flow cytometry. Propidium iodide is a DNA intercalating agent that cannot pass through viable membranes and thus identifies dead cells. In addition to purification, the flow cytometer can count the number of cells that have a certain spectral quality. Technically, if the machine is used just for counting and not for separating subpopulations of cells, then the procedure is called flow cytometry, while cell sorting or isolation is called FACS. However, because FACS is shorter to say than flow cytometry, many people call anything to do with a flow cytometer FACS, much to the annoyance of pedantic fools like your instructor. So… let's start a revolution, and label flow cytometry FC from here on – even shorter than FACS!
Before there were flow cytometers, there were Coulter counters. Coulter counters are automated cell counting machines developed in the 1950s that count cells as they flow in a liquid stream. In an ingenious conceptual leap, Mack Fulwyler combined the technology of ink jet printers with that of Coulter counters to develop the first flow cytometer. The ink jet printer head works by vibrating a nozzle so that a spray of discrete droplets is formed. Similarly, in a flow cytometer, a liquid suspension of cells is forced at high pressure through a vibrating nozzle to create tiny charged droplets, each containing a single cell. The stream of droplets passes in front of a laser beam, and the scattered light is analyzed by a series of filters and photomultiplier tubes that convert the light signal into electrical impulses. Thus, each cell is "interrogated." In FACS, the spectral qualities of the cell are analyzed nearly instantaneously and compared to user-specified spectral qualities. For example, if you have a mixture of green fluorescent cells and non-fluorescent cells, you can ask the machine to isolate the green cells. If a cell registers as green, an electrical charge deflects the cell to make it fall into a collection chamber. In FC, each cells is interrogated, and documented as fluorescent or not, but then all the cells go into the same waste stream.
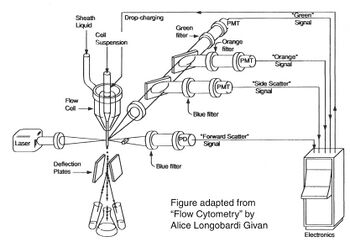
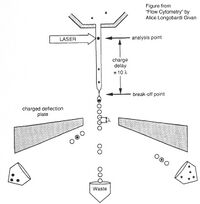
Cell sorting, or FACS, is technically challenging and most FACS machines are only run by experts. In contrast, regular old biologists and biological engineers are often trained to perform flow cytometry as graduate students. In preparation for an FC experiment, the user must appropriately set voltage, compensation, and gating using control samples; failure to set these parameters correctly will result in GIGO (garbage in, garbage out). The next step of an FC is experiment is to measure the experimental samples on the machine. The final step is to document and analyze the statistics output by the flow cytometer. In our case, this last step involves making an additional set of analysis gates. Although these three steps will primarily be done by the teaching faculty, we will describe them briefly below.
How does the machine "know" which cells are fluorescent or not? There is no magic here; the user must tell it. The first key control to include is a mock/negative control: the cells undergo the lipofection treatment, but without any DNA. The reason to mock transfect these cells is in case their viability or FC profile is affected by the lipofectamine itself. We view these cells first under side scatter versus forward scatter. Forward scatter (FSC) is proportional to size, while side scatter (SSC) tells us something about shape/roughness of the cell. The cells should be the largest objects in the solution, while dust and cell debris will tend to fall at very low FSC. Note that in the gate labeled "P1" below we do exclude very high FSC and very high SSC events, as these are likely aggregates of cells and dead cells, respectively. The voltages should be set such that few or no cells are lost on the SSC axis. Raising the voltage shifts cells up or right, while lowering the voltage shifts the cells down or left. Next we look at blue fluorescence (Pacific Blue) versus green fluorescence (FITC). We set the voltage to keep these negative cells in the lower left corner, while still keeping all of the cells visible, and set the gating such that 0-0.1% of negative cells show up as fluorescent.
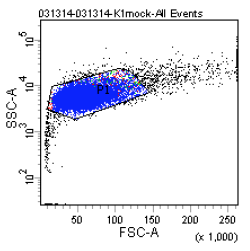
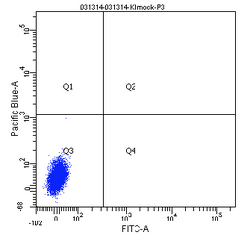
We then perform similar adjustments using single-color controls: cells that have been transfected only with GFP or only with BFP. The BFP spectrum is quite nicely contained, and doesn't "bleed into" the GFP spectrum at all. In other words, blue fluorescent cells won't also (spuriously) appear green. In contrast, the GFP spectrum does bleed into the BFP spectrum. Normally, we could use mathematical compensation to fix this issue: simply subtract a fixed percentage of the GFP fluorescence signal from the BFP signal! However, lipofection in particular leads to a small population of highly fluorescent cells that are not well-adjusted by compensation, and instead must be gated out.
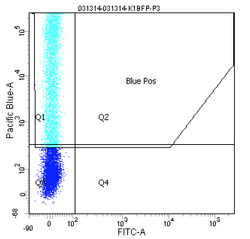
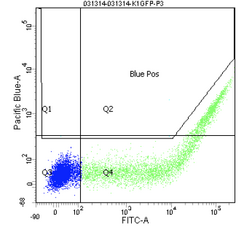
Finally, the experimental samples, which are co-transfected with GFP and either intact or damaged BFP, may be observed, and will look similar to the plots below. By measuring the percentage of cells that fluoresce blue, and normalizing according to the equation presented on Day 5, you will have some measure of the frequency of non-homologous end-joining within CHO cells.
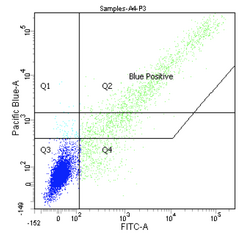
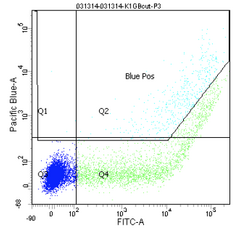
Flow cytometers have been around since the 1970s, and while the details have gotten fancier (more colors, faster measurements, etc.), the general principles have remained the same. It is truly a testament to the versatility and utility of flow cytometry that the technology does not look to be supplanted by another approach any time soon.
After preparing your samples to be measured by flow cytometry, you will prepare one final validation experiment: a dose response curve for the C401 DNA-PK inhibitor. Six flasks of cells have been pre-treated with different amounts of C401. Working together as a class, you will count the cell abundance in each trypsinized flask, while a member of Samson lab takes the remaining cells away and irradiates them. On his/her return, you will follow Part 2 of the protocol below. We will discuss the background of this experiment further in the Day 7 introduction and pre-lab.
Protocols
Part 1: Prepare cells for flow cytometry
- Begin by briefly looking at your cells under the microscope. Do the cells in any wells appear less dense or less healthy than in others? Note down any such observations.
- Aspirate the media from each well according to the protocol below.
- As you work, tip the plate down a little to pool the media at the bottom of each well.
- Place your aspirator at the bottom of a well, and suck up the media. Remove all the liquid, but remove the aspirator promptly after that or you may damage/aspirate some cells.
- Before moving to the next (non-duplicate) well, dip your aspirator briefly (less than a second!) in ethanol. Then hold the Pasteur pipet up to dry and count to 3.
- Alternatively, you may use a fresh yellow tip on the end of the Pasteur pipet when changing samples.
- Gently distribute 0.5 mL of warm PBS to each well, using a 2 mL serological pipet.
- In other words, don't blast the liquid right at your cells.
- Repeat the aspiration step, again using the ethanol to clear the pipet between samples.
- Add 0.25 mL of phenol-red-free trypsin/EDTA to each well, using a P1000.
- Incubate for about 3 min, until cells are rounding up and starting to come off.
- Meanwhile, label your pre-chilled flow cytometry tubes according to the tables below.
- Distribute 0.25 mL of media to each well. You are welcome to use the same pipet tip for each well, as long as you don't touch the tip down into the trypsin/well!
- One well at a time, follow the protocol below to transfer a uniform cell suspension to the appropriately labeled flow cytometry tube.
- Pipet the full 0.5 mL of solution up and down about four times. Make sure to vary where your tip is in the well, concentrating on the four "edges" of the circle (if the circle were, you know, a square) to more fully resuspend the sticky cells. Check the first well or two under the microscope to be confident of your technique. You should see very few cells.
- On the final resuspension, have your partner hand you the filter-topped flow cytometry tube. Press the blue tip gently against the top of the filter, and expel the cells. You should see the liquid go right through; if you feel resistance, ask for help.
- Each tube should be immediately returned to your ice bucket.
Sample order for each team: K1 intact, K1 intact duplicate, C401 intact, C401 intact duplicate, xrs6 intact, xrs6 intact duplicate, K1 damaged, K1 damaged duplicate, C401 damaged, C401 damaged duplicate, xrs6 damaged, xrs6 damaged duplicate.
The original table has been revised to correspond with your file numbers! (A few teaching faculty samples will be evaluated first.)
| Team Color | Tube numbers assigned T/R | Tube numbers assigned W/F |
|---|---|---|
| Red | 16-27 | 10-21 |
| Orange | 28-39 | 22-33 |
| Yellow | 40-51 | 34-45 |
| Green | 52-63 | 46-57 |
| Blue | 64-75 | 58-69 |
| Pink | 76-87 | 70-81 |
| Purple | 82-93 | |
| Silver | 94-105 | |
| White | 106-117 |
Part 2: Plate irradiated cells for inhibitor dose response
Today you will do a bit of teamwork to avoid duplicating effort, for both you and for the teaching faculty. In short, as a class you will split up the counting of six distinct cell populations, and then everyone will plate from the same six pools of cells. You can also brainstorm approaches to the protocol and double check each others' calculations as you go!
- Among the class, please prepare six 15 mL conicals, labeled 0, 0.1, 0.5, 1, 5, and 10 μM, and six similarly labeled eppendorf tubes.
- The following steps will most likely be done by the teaching faculty:
- Each T25 flask of cells will be trypsinized according to the M2D1 procedure.
- As the cells are triturated, the teaching faculty will note down the precise volume in each conical, subtracting off the 90 μL of cells that are taken away for counting.
- The six conicals of cells will then be picked up and irradiated at 4 Gy by a member of the Samson lab. Meanwhile…
- Add 10 μL of Trypan blue to your 90 μL aliquot of cells, and proceed to count them. We will have one aliquot per team on T/R, and a different distribution of work or duplicate counting on W/F. Some reminders:
- Count at least two 4x4 squares, and all four 4x4 squares if the difference between the first two is >10-20%.
- Have a consistent counting method to avoid under- or over-counting. For example, always count cells that fall precisely on the top line of a square, but never count cells that fall right on the bottom line.
- Multiply by the dilution factor first. Then, multiply by 10,000 to calculate cells/mL. You can also multiply by the total volume recorded by the teaching faculty to get an absolute cell count and check whether we have enough cells to proceed.
- Once you have a concentration, figure out how many cells total to take, and how much media to dilute them in. Specifically, note that:
- Each team needs 1000 cells from each cell population (each C401 concentration), plated in 3 mL of media. Don't forget to have an overall excess of 10-15%!
- If you are transferring a very small volume of cells, it may be better to prepare a 1:10 intermediate cell dilution before making the final plating solution.
- Try these calculations manually first, and then double-check them by adding your raw data to the Excel file on the instructor's computer.
- Each team should end up with each cell population in one well of their own personal 6-well plate. Some possible strategies:
- Pass the conical tubes around the hoods. Each team carefully keeps track of which C401 concentration they are currently working with, plating according to the diagram below.
- All the conicals and plates are kept in one hood. Each team takes a turn pipeting one of the six cell populations into all six (TR) or nine (WF) teams' plates, always in the same well. Then the next team pipets the next of the six cell populations in the next well.
- Your fun/efficient/etc. idea here!
Shannon and Su will be around to help both days, while Agi and Aneesh will be at the flow cytometry facility. Please don't stare at this protocol too long willing it to make sense :)
For next time
Due on M2D7
The major assessment for this module will be a research article describing your NHEJ system work. For this assignment, you will write a first draft of the introduction to your report. Recall that the introduction provides a framework for the story you are about to tell, with respect to both background and motivation. You are encouraged to revisit the scientific writing guidelines before you begin.
- What might the big picture section consist of? How will you motivate measuring DNA repair and place this study in a broader context?
- What background would the reader like to have as you zoom in? What biology topic(s) and what engineering topic(s) are relevant? Be sure to cite previous work to establish credibility.
- When you define your specific study, include preliminary hypotheses about your findings.
- Begin by doing some advance planning and writing an outline. There is no one right format, but we strongly suggest submitting: A topic sentence for each paragraph and a transition statement/phrase showing how the content in that paragraph logically leads to the main topic covered in the next paragraph. You can include a few additional notes if you wish, but please don't submit pages and pages of your thought process or of every sub-point you plan to make! We suspect that you will find the BE Writing Lab an outstanding resource for assisting with the planning/outlining stage. This part of the assignment is for bonus credit only; it is not required.
- From your outline, write a full draft of the introduction to your Module 2 report.
Note: the full research article assignment description will be updated soon!
Due on M3D1, but you may want to start sooner…
- Revise your earlier draft of the Methods section, just through M2D2, applying the feedback you received.
- Prepare the rest of your Methods section (through M2D7) in outline form. Start by considering what methods may be logically grouped together. At a minimum, you should turn in
- sub-section titles,
- topic sentences for each sub-section,
- and a few short phrases indicating what content will be included in that sub-section.
- The phrases do not need to include every single material/concentration/etc. that you will use, but they should convey the scope of that information in very abbreviated form.
- For example, phrases for the first half of a Western sub-section could look like: cell lysis method (RIPA and inhibitors from BBP, scraping, ice 15 min and spin 15 min); Precision Red to measure protein in supernatant; preparation step (add Laemlli, boil equal amounts of protein). The second half would include similar types of phrases to cover the PAGE and transfer steps.
Reagent list
- 0.5% Trypsin/4.8 mM EDTA diluted 2-fold in PBS (Life Technologies)
- OptiMEM as on Day 5
- Compound 401 at designated concentrations (Tocris/R&D Systems)
- prepared from 5 mM stock solution in ethanol
- Media and trypsinization reagents as on M2D1
- BD LSR II flow cytometer (BD Biosciences)
- Green excitation: 488 nm
- Blue excitation: 405 nm
- Green emission filter: FITC -- 530 nm emission peak, 30 nm width
- Blue emission filter: Pacific Blue -- 450 nm emission peak, 50 nm width
- Cells for irradiation experiment
- were plated at 400,000 2 days in advance (T/R) or 200,000 3 days in advance (W/F) in T25s
- the day before use, C401/ethanol mixtures (with ethanol volume kept constant) were added to the cells at 5 pm
Navigation Links
Next Day: Data analysis Previous Day: Cell preparation for DNA repair assays
